In my long career, there have been cycles in transfusion practice. Today’s dogma becomes yesterday’s heresy and then later again the dogma. Just consider the selection of blood components before the introduction of cyclosporine for intended renal transplant recipients.
In training, I was told NEVER, NEVER use the intended donor’s blood for the renal recipient. This would immunize him against the donor tissue antigens and cause the transplant to fail.
Several years later after cyclosporine, we were doing a booming business of directed RBC transfusions from the donor to his/her recipient.
Just a few years ago, I used blood component therapy for all, especially trauma patients. Give the victim what he lacks: for oxygen-carrying capacity, RBCs; for volume crystalloid; for low protein albumin; for coagulopathy FFP, factor concentrates, cryoprecipitate; for thrombocytopenia, platelets.
Most recent studies now mention the danger of giving too much crystalloid, etc. It talks about using fresh whole blood to provide all of the above in less volume. Results from trauma and military studies are encouraging and may be better than individual component therapy.
There are special considerations for whole blood:
- 21-day outdate for the RBCs
- Platelet functionality limited after 7 days
- Use of group O, low-ABO-titer
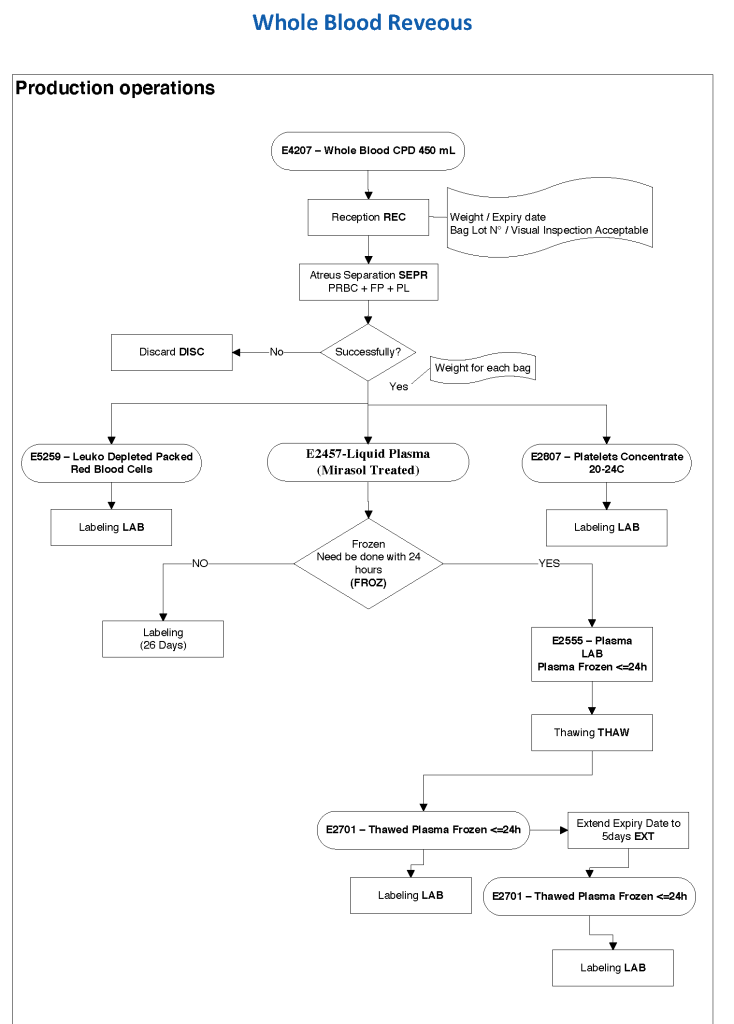
Pathogen-inactivation of whole blood is CE-approved by riboflavin (Mirasol). Terumo BCT is developing an exciting technology to first use Mirasol and then make components using the Reveos automated component system—RBCs, plasma, and platelets. This is an ongoing project so for now the only CE-approved project is use as whole blood. Such Mirasol-treated whole blood has been shown to prevent malaria transmission in Ghana.
From my review of the literature, these are my specifications as of this date 18/8/20:
- Use/ordering restricted to trauma and selected ICU/surgery suites
- Only male donors
- 7 day outdate
- Group O, Anti-A and anti-B IgM titers, both <= 1/256
- Leukodepleted < 1E6 residual WBCs
What level of anti-A and anti-B titers is acceptable? The titer was set as low as 1:32 but at recent THOR meeting 1:256 has been used. In Qatar in a pilot study, I found that about 50% of our donors had titers < 1:256. The issue is that each time the donor presents himself/herself, we must repeat the titer—it is not stable.
Of course, performing even just a saline-titer is time consuming. The only practical way for us in Doha would have been to use an automated titration option on an immunohematology analyzer—in our case, the Ortho Vision MAX, which could perform 1 titration run in about 30 minutes, and the instrument cannot be used for any other testing during the process.
I personally would perform leukodepletion to conform to CE, but you need a special whole blood filter that removes WBCs but spares the platelets. Terumo BCT has such a filter that achieves <1E6 residual WBCs. Never use a standard RBC leukodepletion filter since it will remove BOTH platelets and WBCs—this would defeat the purpose of using whole blood.
At HMC Doha, female donors were only used for packed RBC production—all plasma and platelets were discarded. Some centers do HLA antibody screens and allow negative females to donate.
Finally, many groups do not leukodeplete at all. I am concerned about the risk of adverse reactions and TRALI so I would conform to CE and do it.
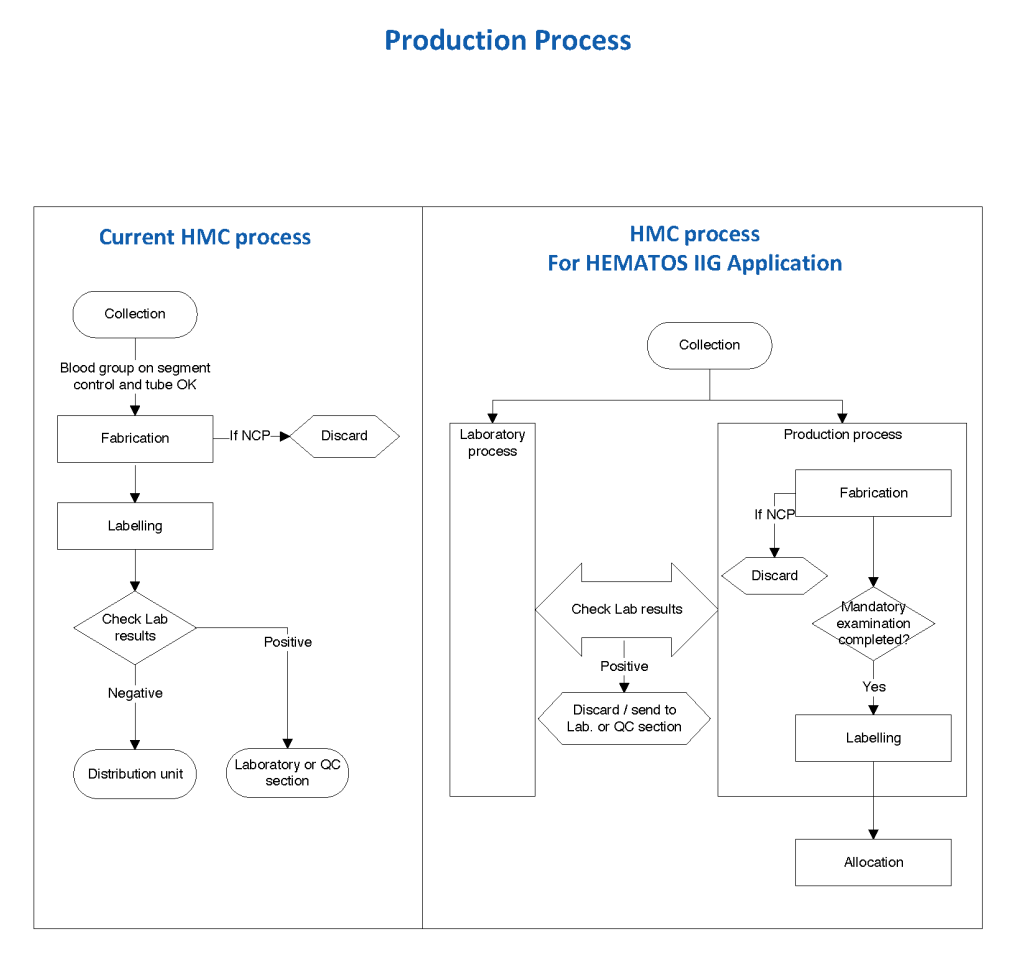
Whichever conditions you stipulate, it is easy to create the process in Medinfo. The most important thing is to know what you want to specify.
19/8/20