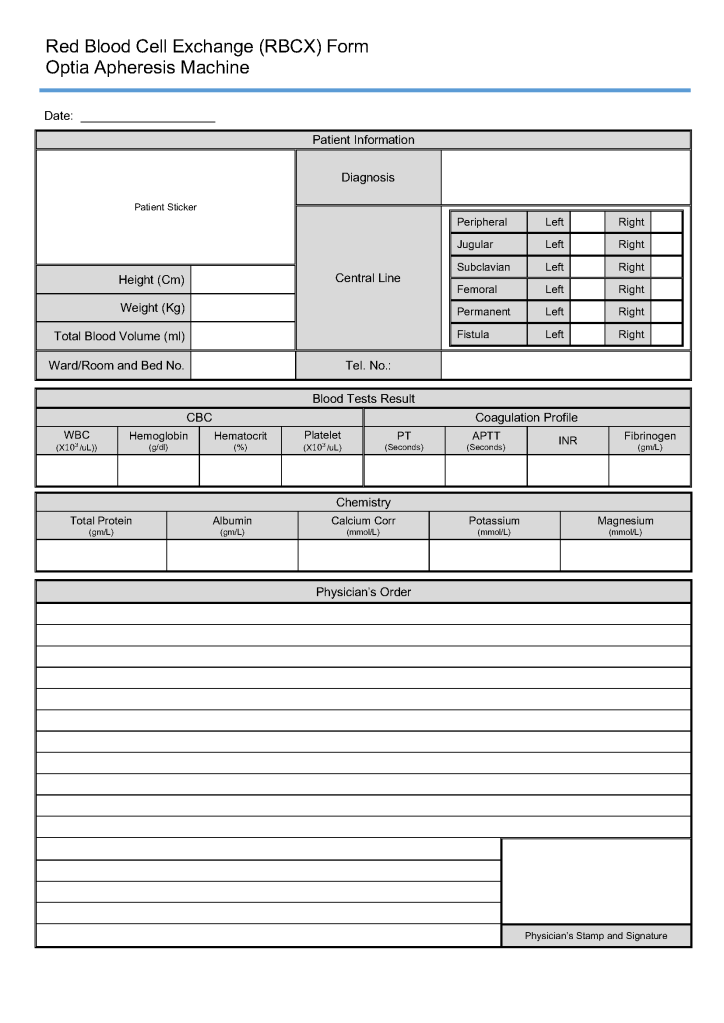
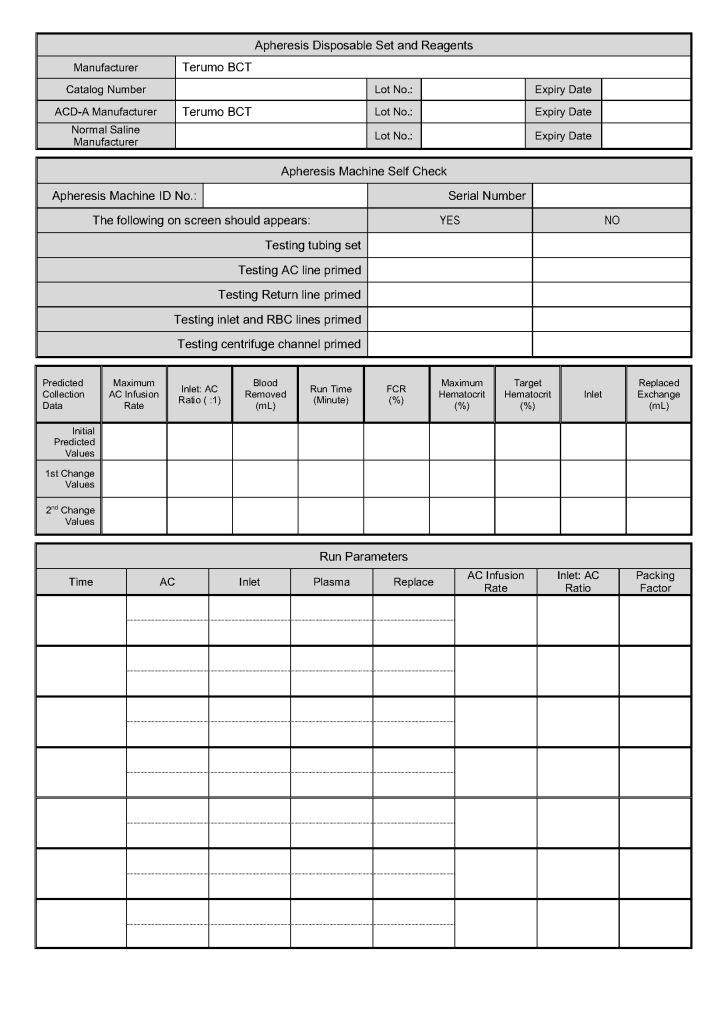
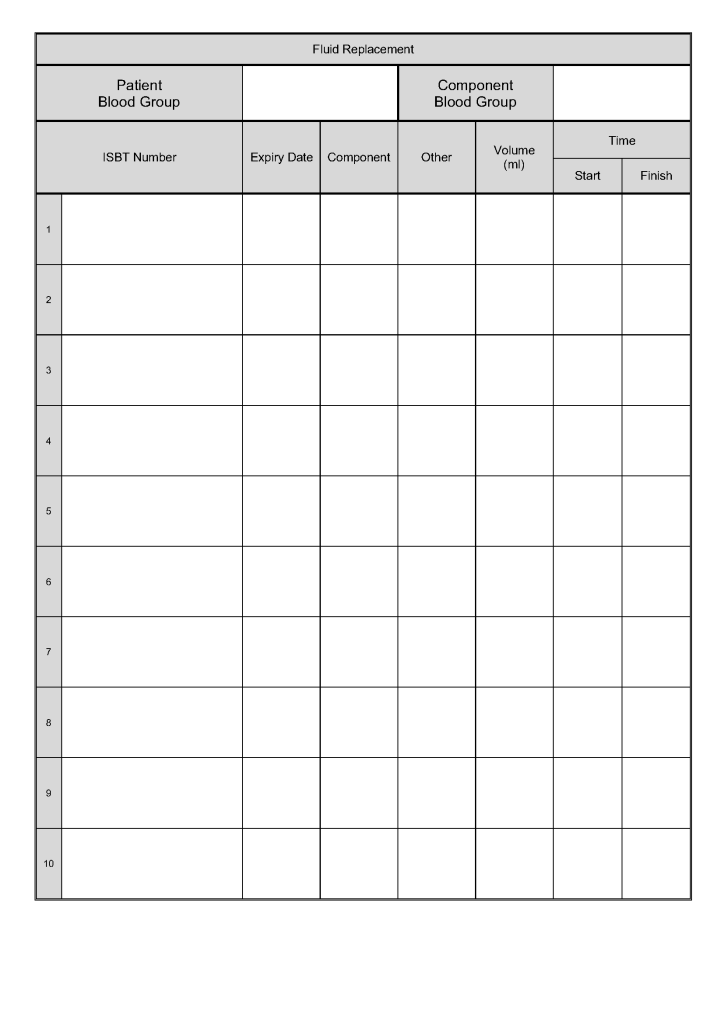
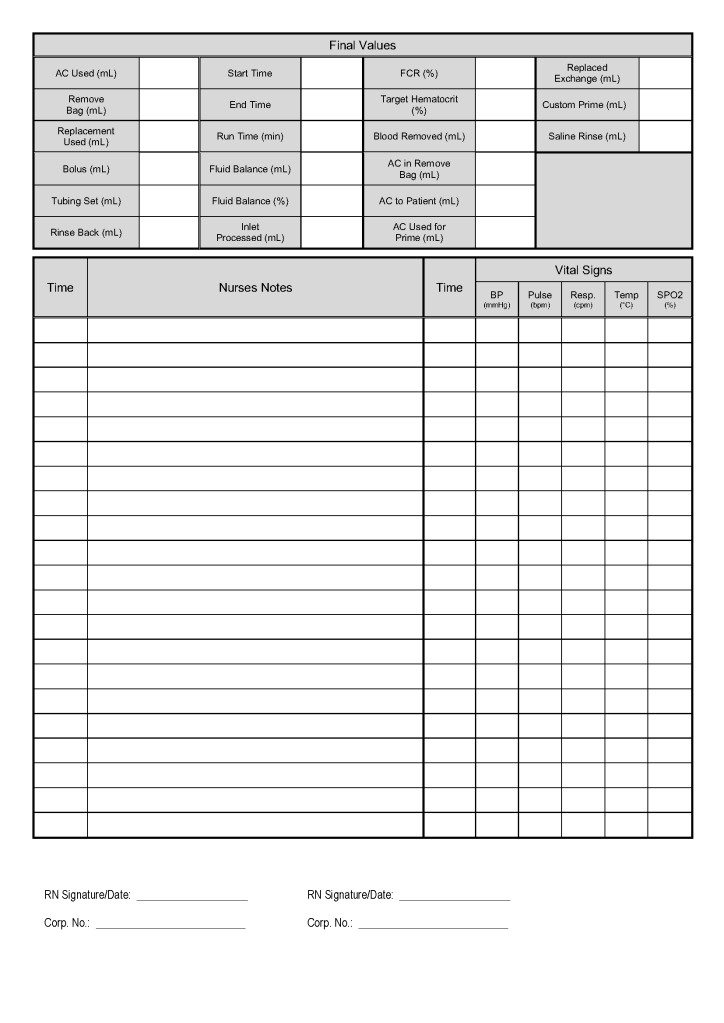
Principle:
Antigen typing of cells with large amounts of coating antibody (i.e. strongly positive DAT 3-4+) may not always be possible because the bound antibody may block available antigen sites. This policy is to clarify how to recognize and handle such situations.
Policy:
- Always follow the manufacturer’s instructions for the use of the typing reagent.
- In particular, note whether a control must be run with the test (e.g. D-control, D-diluent, etc.) or if it is included in the gel or glass bead card.
- If a control is required, use exactly what the manufacturer recommends.
- DO NOT SUBSTITUTE ANYTHING ELSE AS THE CONTROL!!
- In particular, note whether a control must be run with the test (e.g. D-control, D-diluent, etc.) or if it is included in the gel or glass bead card.
- Interpret the reactions exactly as the manufacturer indicates.
- If the test is invalid because of the control or any other reason, report the antigen typing as indeterminate and send for Transfusion Medicine Physician review.
- If the DAT is 3-4+ and the antigen typing shows no reaction (apparent negative), send the case to the Transfusion Medicine Physician for review and final interpretation. DO NOT ENTER THE RESULT AS NEGATIVE UNLESS THE TMP INSTRUCTS YOU TO DO THIS!!
- To rule out a blocking antibody, a special elution to gently remove the coating antibody may be needed so that the RBCs can then be typed (not acid glycine technique—rather, gentle heat elution.) The Transfusion Medicine Physician will decide whether to do this additional testing.
References:
- Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
- Technical Manual, Current Edition, AABB, Bethesda, MD, USA
- Guidelines to the Preparation, Use, and Quality Assurance of Blood Components, European Committee (Partial Agreement) on Blood Transfusion (CD-P-TS), Current Edition