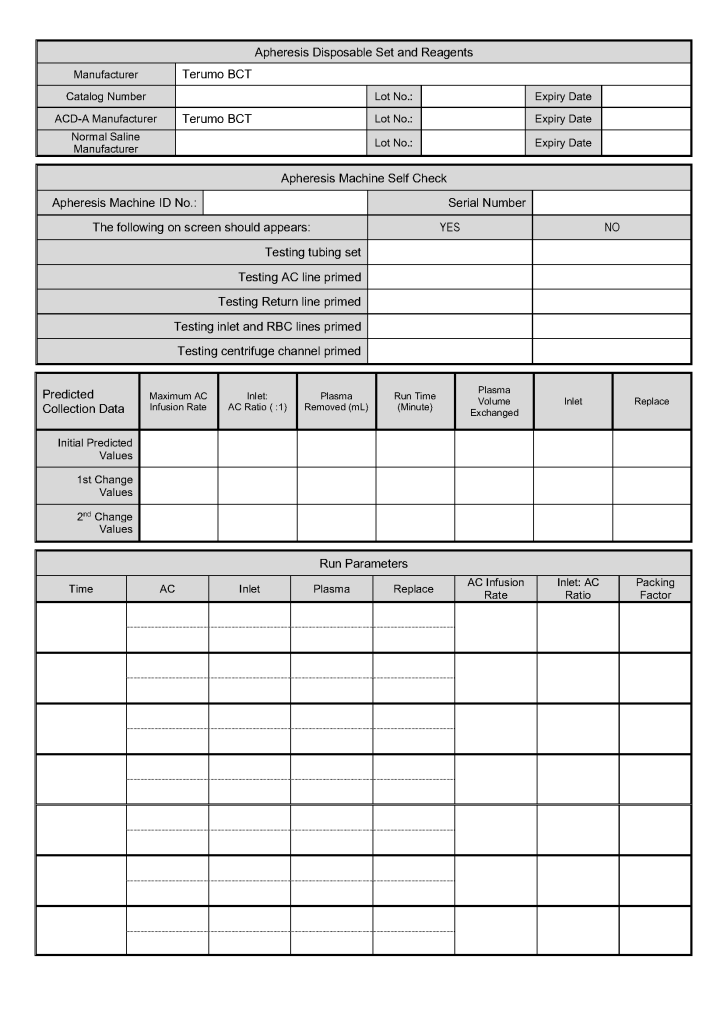
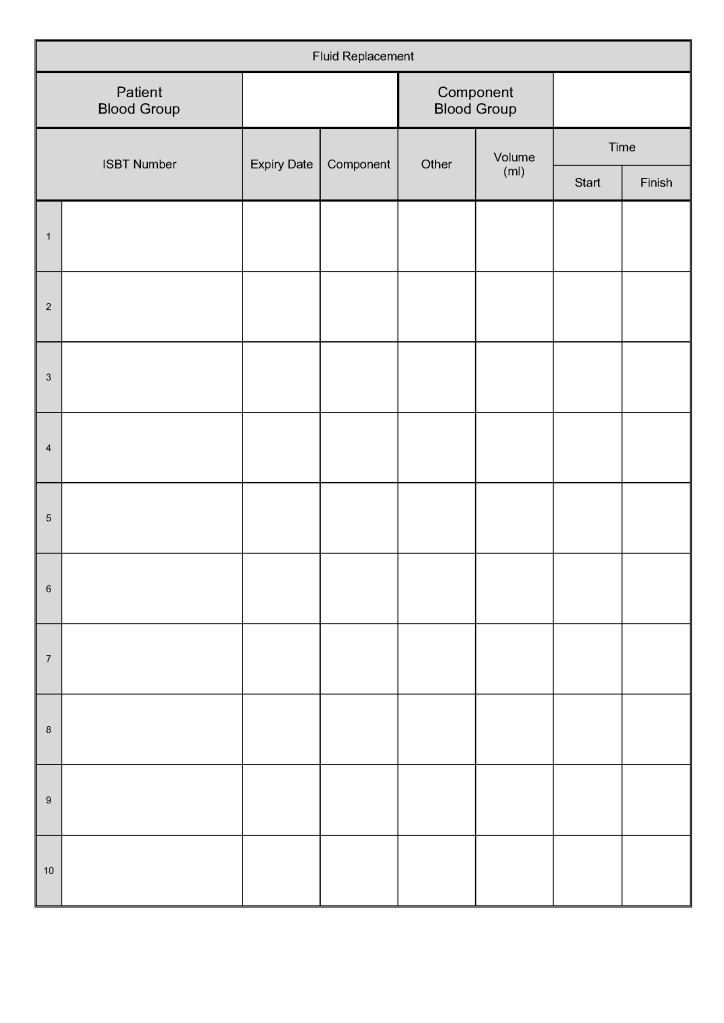
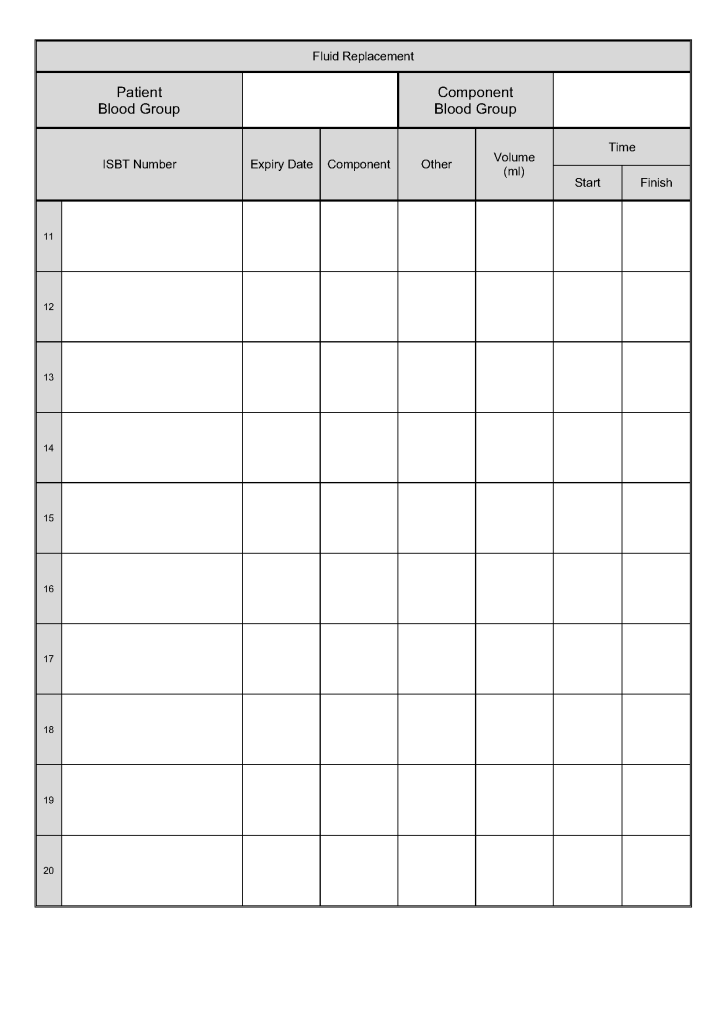
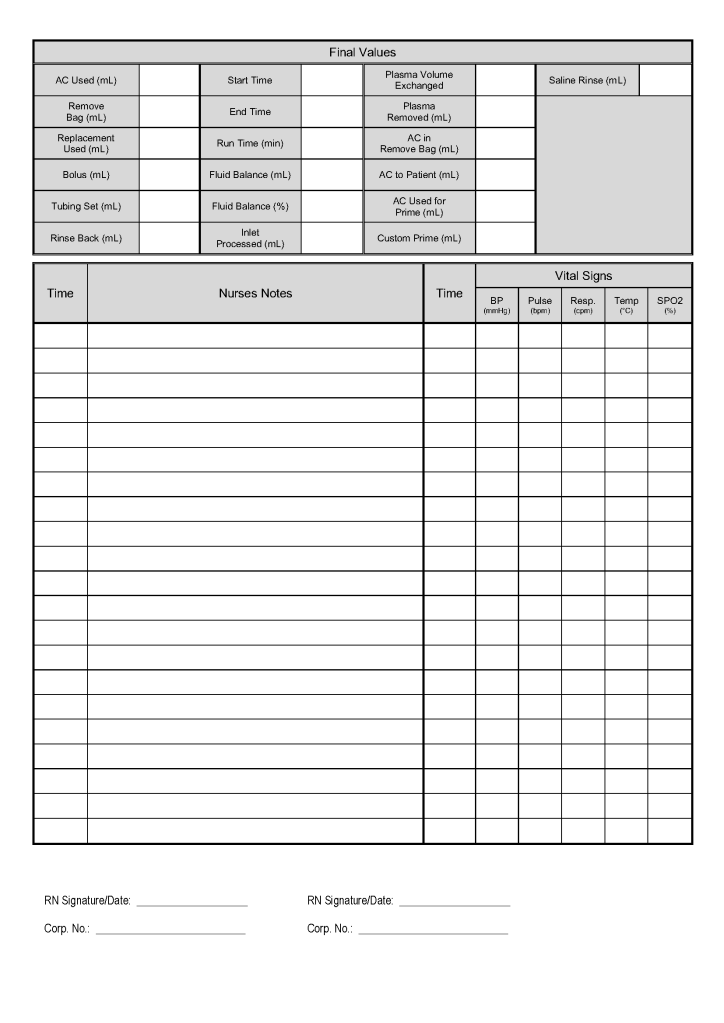
In all my years practicing medicine outside the United States, I have come to appreciate working with a diverse group of health care professionals from many countries and cultures and with many different primary languages.
I learned that there are many different international standards and not all agree with each other. Yet, despite the apparent contradictions, they all work to improve patient care and were generally successful. It made me reconsider my roots and think less dogmatically and be willing to learn from other perspectives.
This applies in many ways. First, which English should we use? Most people are at least somewhat aware of American English, but there are differences with British, Australian, and international English—even the term “blood bank” may have different meanings: is it a hospital transfusion service, is it a donor center, or some combination of the two?
I have worked at many sites where I was the only person native in English. I always tried to conceive how difficult it could be for someone non-native to understand and communicate in a highly technical and highly Germanic structured language. I considered the scenario where I had to work in another language exclusively and perform all my tasks—I highly respect my staff having to cope with this.
English technical writing includes a lot of passive voice, subjunctive mode, perfect tenses, and participles. How formidable a barrier are these to staff whose native languages may not use these structures?
I am not saying that English is the best language to perform the work in, but it is most prevalent one so everyone must cope with it. I told many of my staff to learn German to better understand English grammar.
What bothers me is that certain software vendors and visiting lectures send speakers and staff who ONLY think in American English and American culture. I can think of several anecdotes:
One speaker was talking about hyperlipidemia and used non-SI units. He kept stating cholesterol > 200 and LDL-cholesterol > 120 to an audience who only used SI units. Could the audience quickly convert to cholesterol > 5 and LDL > 3? Did they know what a temperature of 104 F was 40 C?
Another speaker for a software company used an analogy of collecting maple sap and making maple syrup—in a presentation in the Middle East. How many in the audience even knew what maple syrup is?
In building a series of software modules, some company staff used 24-hour clock and others used 12-hour clock. It was chaos trying to define a 24-hour interval between the different modules.
Finally, I think of the Aesop’s fable about the mother who gave birth to a very ugly child, but to her, he was the most beautiful child in the world—so much so that she entered him into a beauty contest. Well, each of us is the “mother” to our documents and memos. The writing looks good to us and is perfect, but do our staff interpret it the same way we do?
I had my staff read my documents and then explain back to me what I was trying to say. I was shocked at the differences in many cases. After this, I always included a validation step to have other people read and interpret what I was saying—and correct any misconceptions in the writing before I finally released the document.
In summary, it is a whole new world outside the United States. Don’t assume everyone thinks the same way or uses the same criteria to accomplish goals. Be open to this and you will have a rewarding international career.
30/10/20