This is the second part of a lecture I gave to medical students at National Guard Health Affairs in Riyadh.








Includes registration, questionnaire, physical exam and arm check, collection, marker testing, component separation, donor immunohematology testing
This is the second part of a lecture I gave to medical students at National Guard Health Affairs in Riyadh.








This is an updated version of a medical student lecture I gave at National Guard Health Affairs in Riyadh.










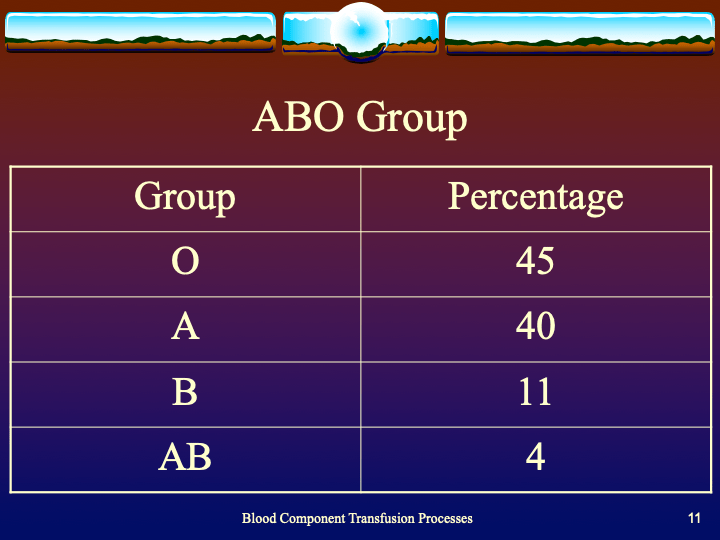

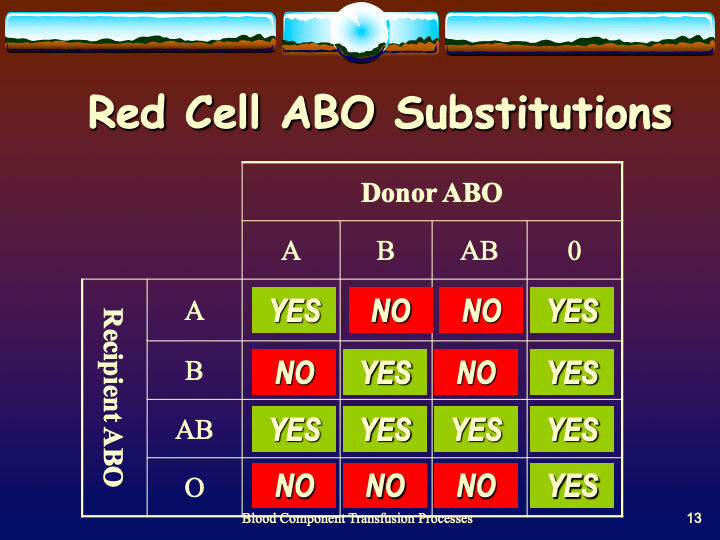
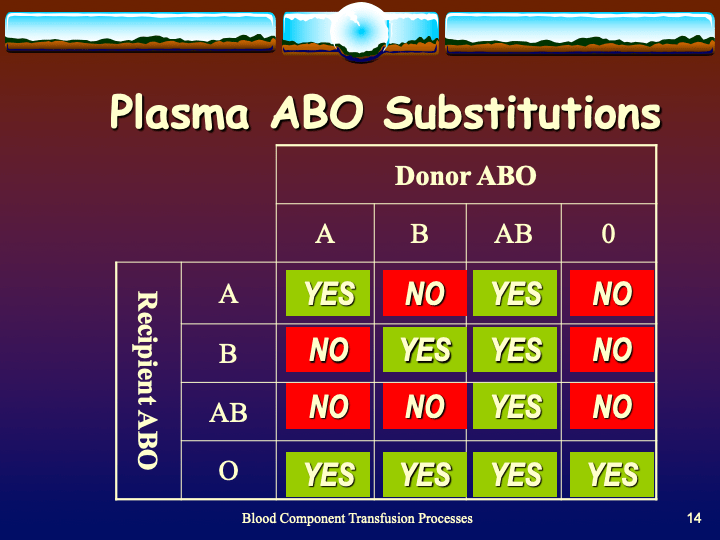
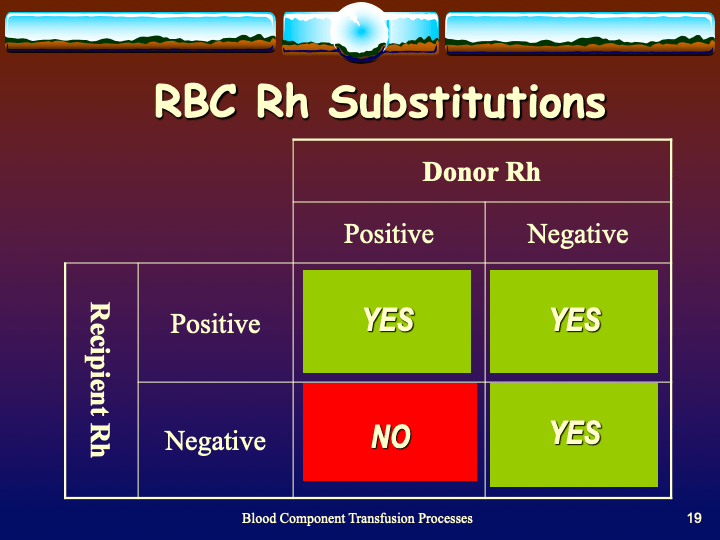
To be continued
5.2.1 PROCESS: DONOR QUESTIONNAIRE
Process:
References:
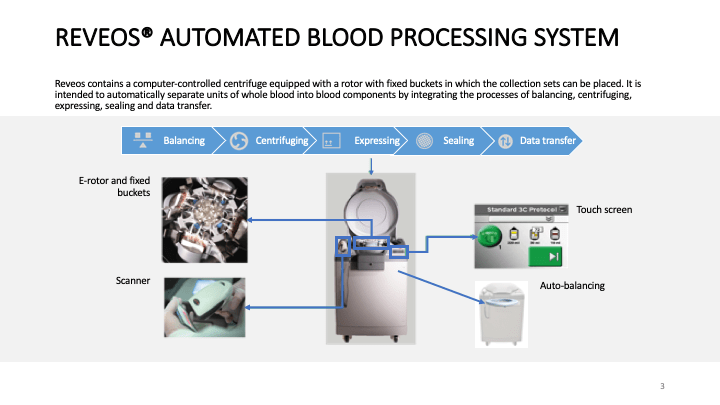
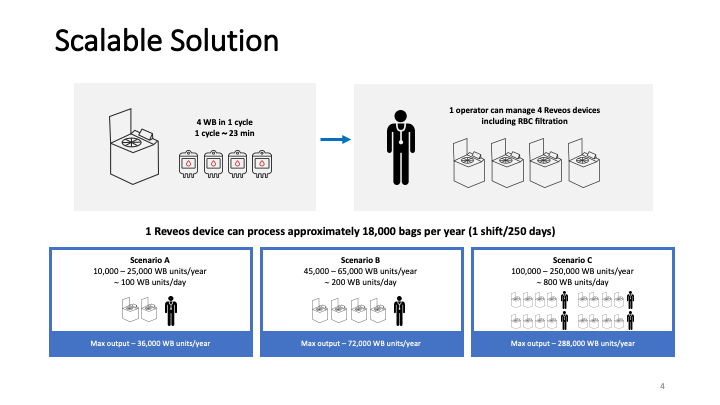
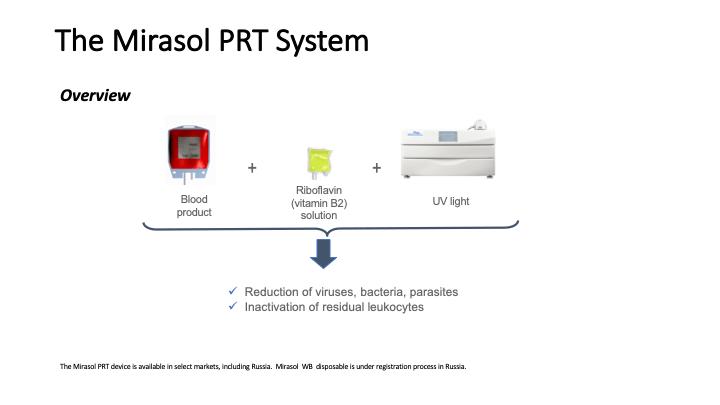
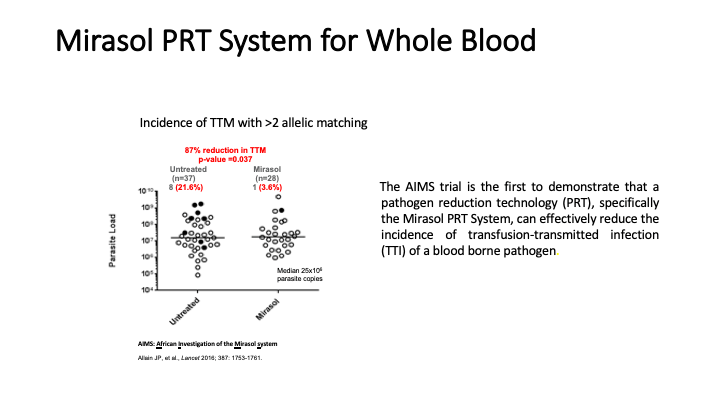
This is the virtual talk I gave at the Russian Transfusion Congress in Moscow on 13 May. It gives a brief description of automated component processing and riboflavin-based pathogen inactivation and then discusses the use of these technologies together to free up labor and enhance the quality of products, i.e. improved GMP, especially if a dedicated blood bank computer system is used to enforce production rules.


















Policy Donor Medical Questionnaire
5.2 POLICY: DONOR MEDICAL QUESTIONNAIRE
Policy:
References:
5.1.1 PROCESS: Donor Registration
Process:
References:
Number: 5.1
Policy:
References:
5.0.1 PROCESS: Donor Recruitment and Campaigns
Process:
References:
5.0 Donor Recruitment and Campaigns Policy
Policy:
References:
This is the final section of this base medical student lecture originally given at NGHA Riyadh. It has been updated to current technologies and current types of blood components.















