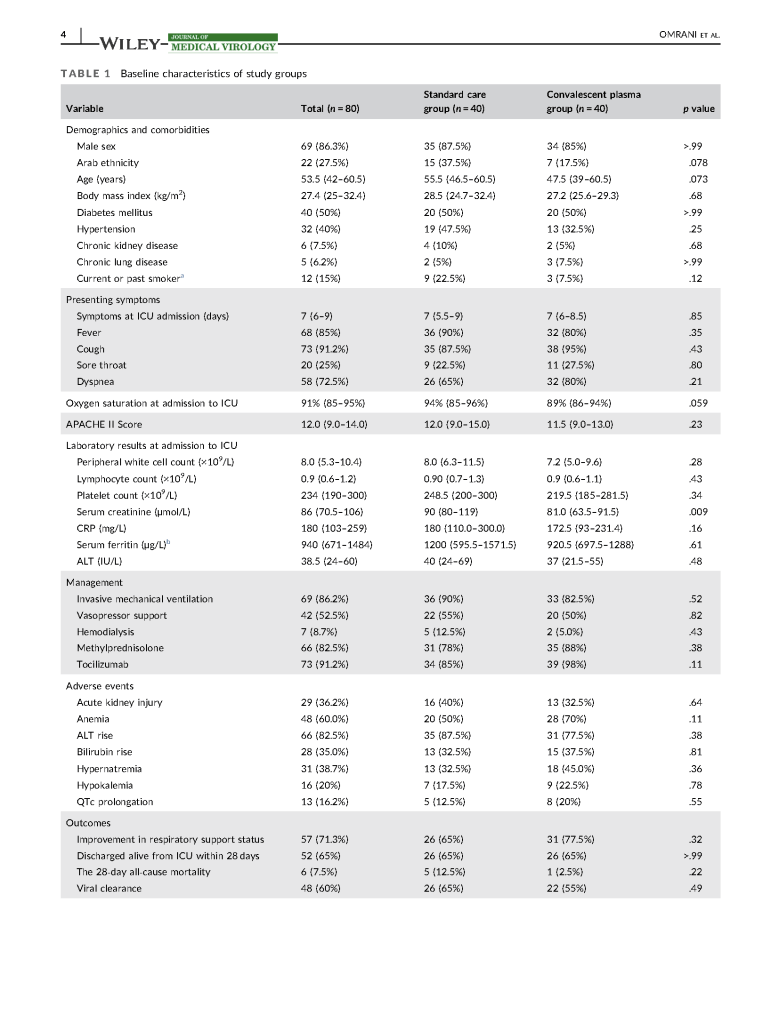
While I was Division Head, Laboratory Information Systems LIS at my previous position, I was asked to use the hospital information system HIS to collect information during the procedure analogous to what was done for dialysis.
I thought of the logistics: one apheresis nurse, one Spectra Optia machine, and one metal cage containing a theft-proof computer on a stand. There was no room for the patient’s bed with all this equipment—the nurse could not move around comfortably.
Second, what I was presented was a hodge-podge of screens on the HIS that the apheresis had to maneuver back and forth between for each measurement—none of the data entry was on one screen! Honestly, there wasn’t enough time to enter all the data between the screens AND look at the patient.
I remind everyone that therapeutic apheresis is not a benign procedure. The patient may be critically ill. The apheresis nurse must concentrate on the patient. The HIS team was more interested in the data collection, even at the expense of the patient.
LIS had not been engaged in building the pathway and the HIS wanted us to follow the dialysis template. They did not know that there are many types of therapeutic procedures, often with different data collection. There is no one-size-fits-all screen!
I refused. The nurse must concentrate on the patient, not the LCD screen. To use the HIS would have been harmful to patient care in this situation. We retained the manual, cellulose interface. We scanned the manual data form and uploaded it into HIS.
Lessons to be learned:
- HIS must engage LIS, and in particular Transfusion Medicine, when building anything for the blood bank. This is in accordance with international accreditation standards.
- We must never lose sight that we are treating the patient, not the computer screen. Especially in therapeutic apheresis, we must use the apheresis specialist nurse to monitor the clinical status of the patient, first and foremost!
- If the proposed computer process is worse than the manual process, keep the latter.
8/10/20