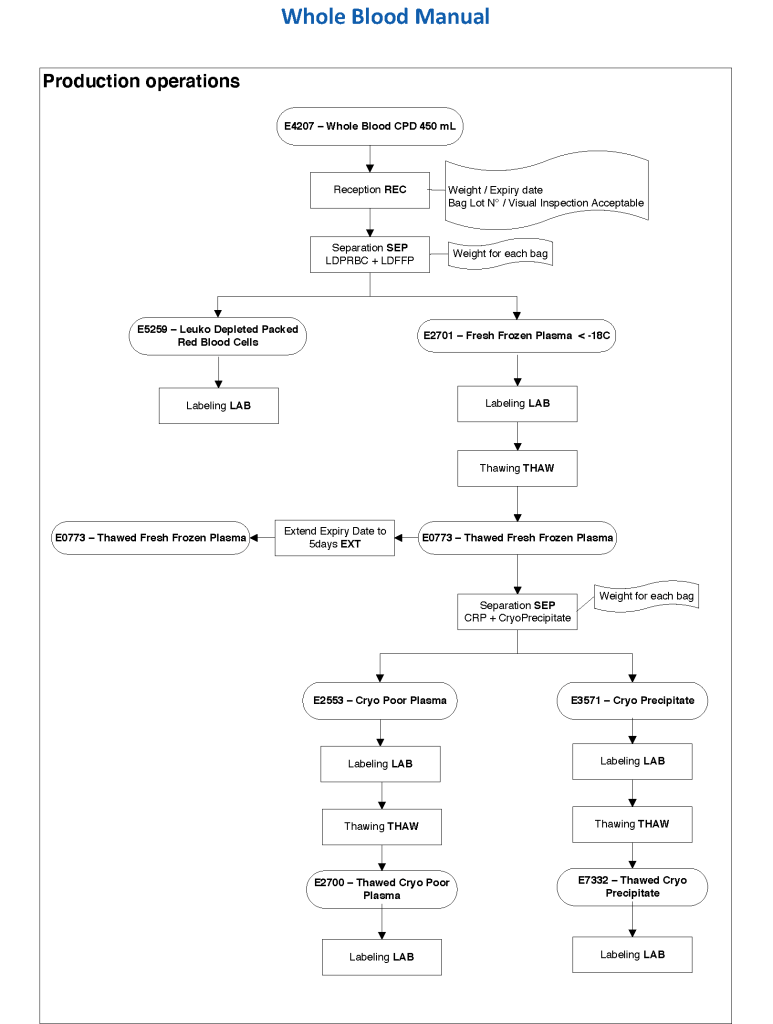
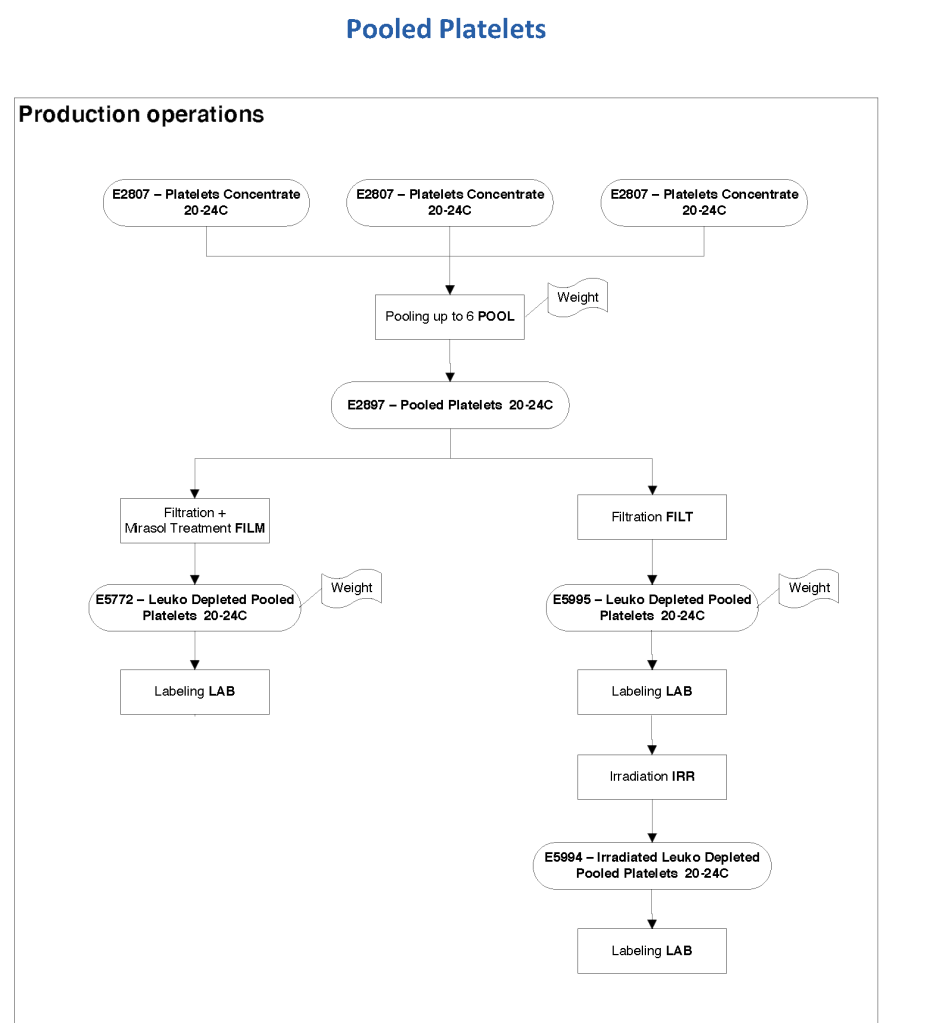
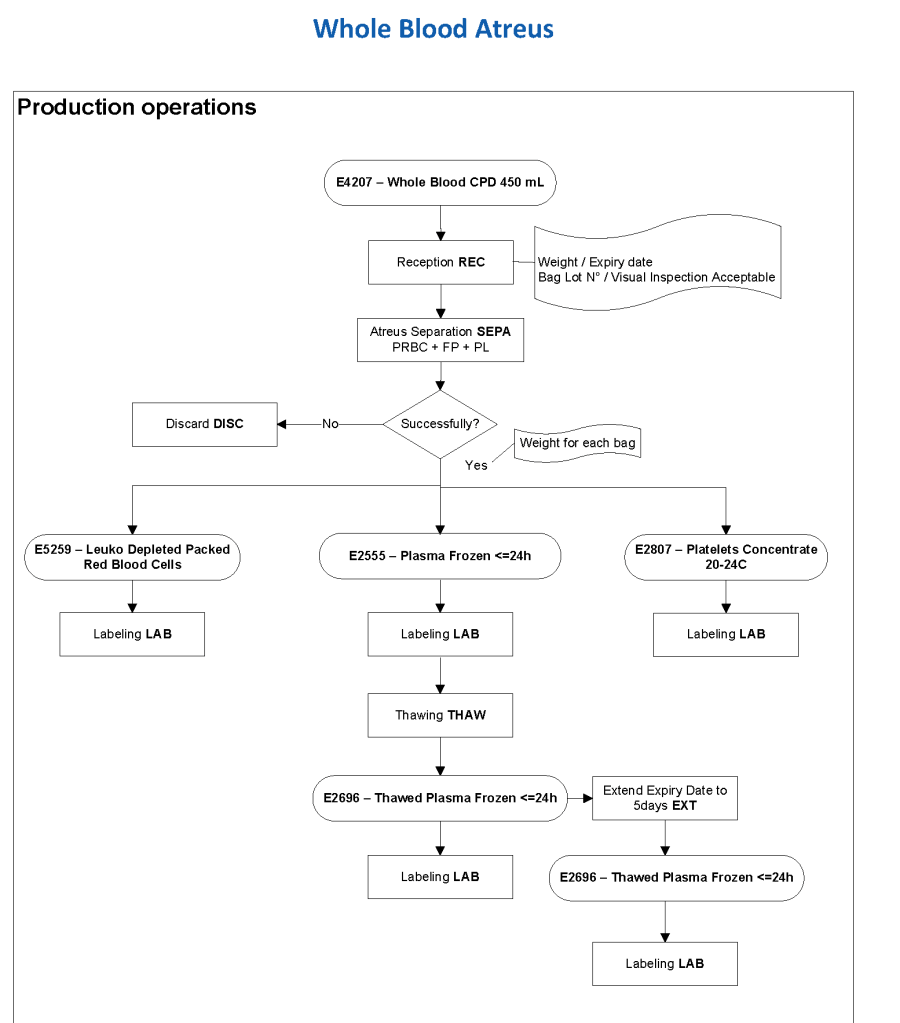
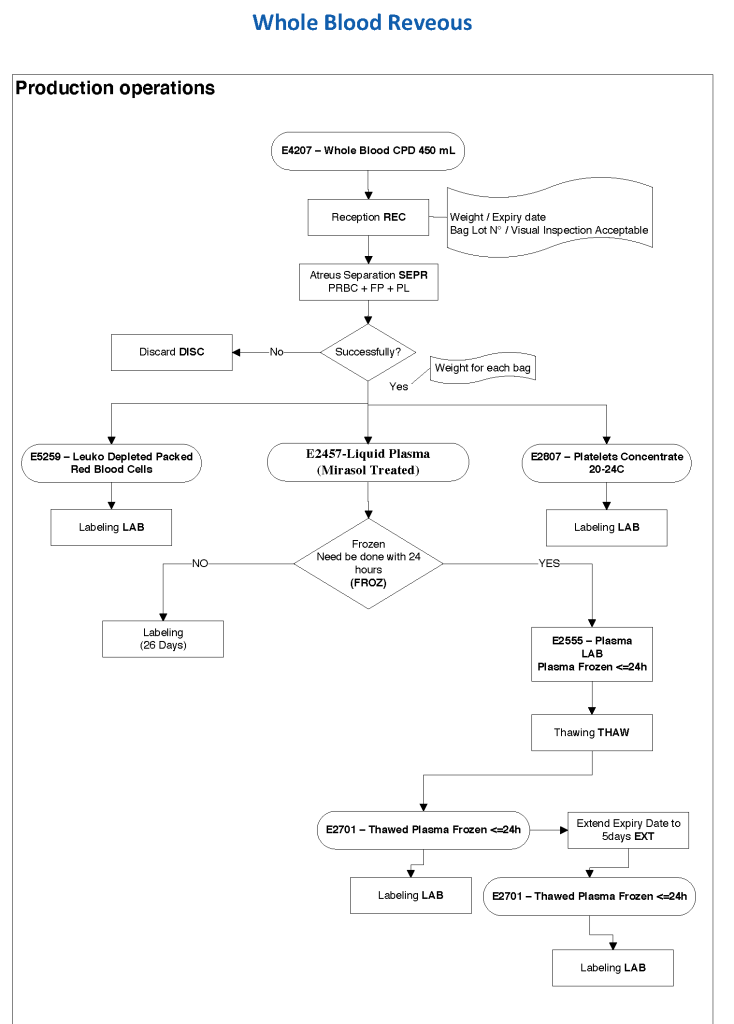
This series of the processes for donor marker testing has demonstrated the complexity and flexibility of designing processes. In Medinfo, you can custom-design the criteria based on your local, national, and international requirements. The end-user client must specify what he wants, and I reiterate: Be careful what you ask for, you may get it. Seek assistance if you are uncertain of what to use.
My criteria were based on several international standards, e.g. AABB, US FDA, CE, and Australian. I strongly recommend you start with a set of standards and localize it for your needs.
For example, US FDA/AABB do not have a screening criteria for brucellosis since it is rare in their jurisdiction. However, it is relatively common in the Middle East so I added a donor screening question for it. International AABB does allow for variance with US FDA criteria so if you are outside the USA, embrace this. The advantage of customizable software allows you do localize it to your needs. A turnkey system, e.g. from the USA, may not allow such changes.
Finally, there are emerging pathogens that are constantly changing the donor criteria (e.g. SARS-CoV-2, MERS, SARS, Zika). The software must be robust and allow rapid alteration to meet new donor screening criteria. This is a constant uphill battle and requires a lot of time to keep up and validate any changes.
Complete Interim Policy on Marker Testing
For your reference, the following is the complete set of marker testing algorithms I used just before I left HMC Doha:
Definitions:
Positive result for EIA means S/CO ratio >= 1.0
Positive result for LIA means particular pattern of bands as defined by the manufacturer
Indeterminate result for LIA means presence of bands not meeting positive criteria
- Hepatitis B:
- HBsAg non-negative, then:
- HBsAg positive with HBsAg confirmatory positive, regardless of other results: permanent deferral, refer to Infectious Disease clinic
- HBsAg positive with HBsAg confirmatory borderline or negative, repeat all HBV testing after 8 weeks
- HBsAg borderline: repeat all HBV testing after 8 weeks
- HBV-DNA positive confirmed, regardless of other results: permanent deferral, refer to Infectious Disease clinic
- If HBcAb positive, repeat after 8 weeks
- Repeat Hepatitis B Testing After 8 weeks:
- HBsAg positive with HBsAg confirmatory positive: permanent deferral, refer to Infectious Disease clinic
- HBsAg positive with HBsAg confirmatory borderline or negative: permanent deferral, refer to Infectious Disease clinic
- HBsAg borderline, permanent deferral, refer to Infectious Disease clinic
- HBV-DNA positive confirmed: permanent deferral, refer to Infectious Disease clinic
- HBcAb positive or borderline with negative HBsAg and negative HBV-DNA: review HBsAb level:
- If HBsAb level >= 100 mIU/mL (100 IU/L), donor may be reentered
- If HBsAb level < 100, then recommend to donor to receive booster HBV vaccine
- After HBV vaccine administration, retest after 30 days:
- If HBsAb level >= 100, donor may be reentered
- If HBsAb level < 100, donor is indefinitely deferred
- HBsAg, HBcAb, HBsAb all negative: reenter into donor pool
- Hepatitis C:
- HCV-RNA positive confirmed, regardless of other HCV results: permanent deferral, refer to Infectious Disease clinic
- HCV-RNA borderline: repeat all HCV testing after 6 months
- HCV-InnoLIA positive, regardless of other HCV results: permanent deferral, refer to Infectious Disease clinic
- HCV-InnoLIA indeterminate: repeat all HCV testing after 6 months
- HCV-Ab positive, HCV-RNA negative, do HCV-InnoLIA:
- If HCV-InnoLIA positive, permanent deferral, refer to Infectious Disease clinic
- If HCV-InnoLIA indeterminate or negative, repeat all HCV testing after 6 months
- Repeat Hepatitis C Testing After 6 months:
- HCV-RNA or HCV-InnoLIA positive: permanent deferral, refer to Infectious Disease clinic
- HCV-RNA or HCV-InnoLIA borderline: permanent deferral, HCV infection not confirmed
- HCV-Ab positive or borderline without positive HCV-RNA or positive HCV-InnoLIA: permanent deferral, HCV infection not confirmed
- HCV-Ab negative, HCV-RNA negative, HCV-InnoLIA negative: reenter donor into donor pool
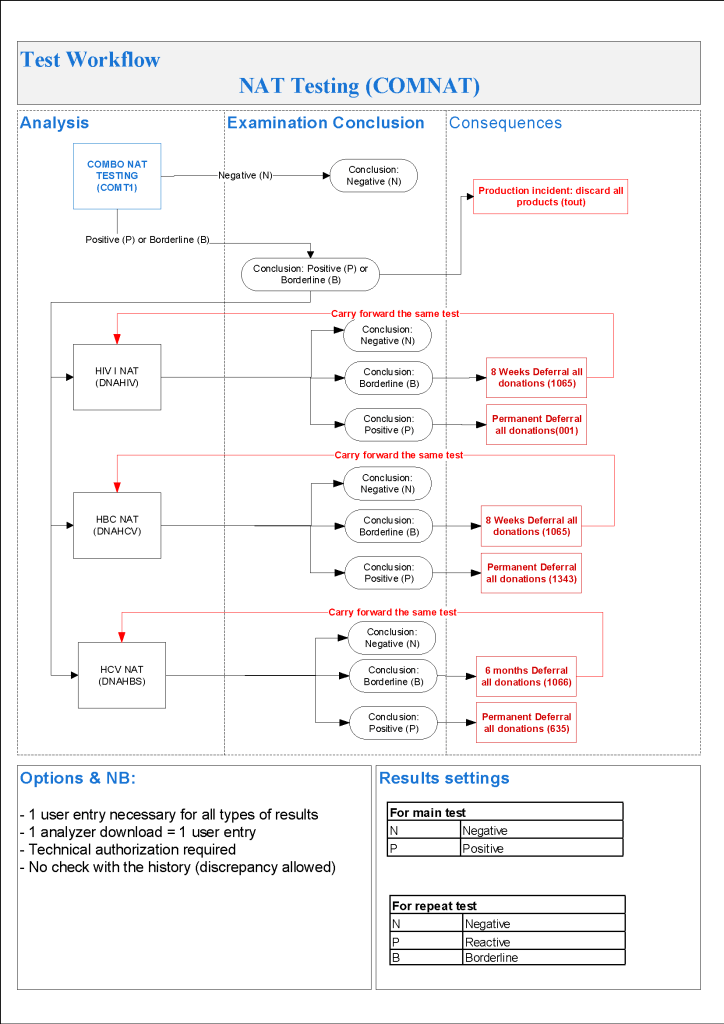
- HIV Testing:
- HIV-RNA positive confirmed, regardless of other HIV results: permanent deferral and do HIV-InnoLIA, refer to Infectious Disease clinic
- HIV-RNA borderline: do HIV-InnoLIA
- HIV-InnoLIA positive, regardless of other HIV results: refer to Infectious Disease clinic
- HIV-InnoLIA indeterminate: repeat all HIV testing after 8 weeks
- HIV Ab positive with negative HIV-RNA and/or borderline/negative HIV-InnoLIA: repeat testing after 8 weeks
- Repeat HIV Testing After 8 Weeks:
- HIV RNA positive and/or HIV-InnoLIA positive, regardless of other HIV results: refer to Infectious Disease clinic
- HIV-InnoLIA and/or HIV antibodies indeterminate: permanent deferral, HIV infection not confirmed
- HIV Ab negative and HIV-RNA negative and HIV-InnoLIA negative: reenter into donor pool
- HTLV 1/2 Testing:
- HTLV Antibodies positive, then do HTLV-InnoLIA:
- HTLV InnoLIA positive for HTLV-1 and/or HTLV-2: refer to Infectious Disease clinic
- HTLV InnoLIA indeterminate or negative, repeat HTLV Ab and HTLV InnoLIA testing after 6 months
- Repeat HTLV Testing After 6 Months:
- HTLV 1/2 antibodies positive, permanent deferral and do HTLV InnoLIA
- HTLV 1/2 antibodies indeterminate, permanent deferral and do HTLV InnoLIA
- HTLV InnoLIA positive for HTLV-1 or HTLV-2: refer to Infectious Disease clinic
- HTLV InnoLIA indeterminate, donor permanently deferred.
- Issue letter HTLV-Not Confirmed
- HTLV 1/2 Ab negative and HTLV InnoLIA negative, reenter donor.
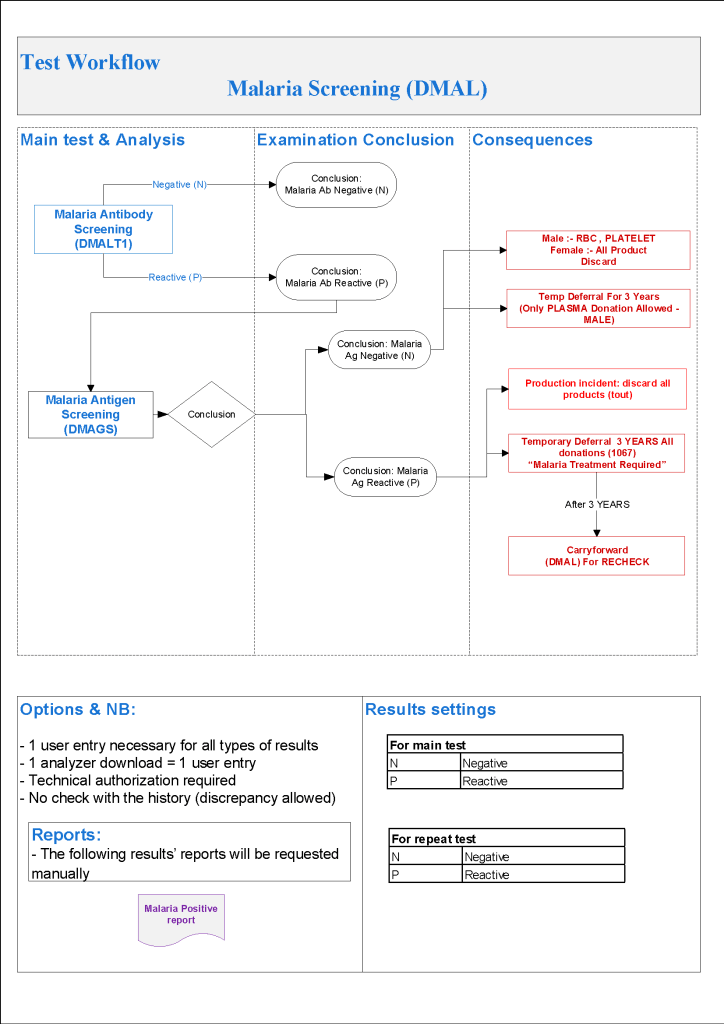
- Malaria Testing:
- Defer donor if he has been in malarial endemic zone within the past 3 months
- If travel to malarial zone > 3 months, do malarial antibody testing:
- Malaria antibody negative: no deferral
- Malaria antibody positive, perform malarial antigen test:
- Malaria antigen test positive, refer to Infectious Disease clinic—defer until 3 years after cessation of treatment
- Malaria antigen test negative:
- RBCs and platelets must be destroyed.
- Repeat malarial antibodies after 3 years:
- If malarial antibody test positive, donor must not be used for RBC components but may be used for plasma production
- If malarial antibody test negative, reenter donor for all components
- Defer donor if he has received malarial treatment (not prophylaxis) for 3 years
- Perform both malarial antibody and antigen testing:
- Defer based on section 5.2
- Syphilis Testing:
- Syphilis Ab test positive or indeterminate: do InnoLIA-Syphilis test
- InnoLIA-Syphilis test positive: permanent deferral, refer to Infectious Disease clinic
- InnoLIA-Syphilis test borderline or negative: defer for 1 year, then repeat all syphilis testing.
- Repeat Syphilis Testing after 1 Year:
- Syphilis antibody testing negative, reenter into donor pool
- Syphilis antibody positive or borderline: do InnoLIA-Syphilis test
- InnoLIA-Syphilis test positive: permanent deferral, refer to Infectious Disease clinic
- If InnoLIA-Syphilis borderline or negative: permanent deferral, syphilis not confirmed
References:
- Revised Recommendations to Reduce the Risk of Transfusion-Transmitted Malaria, Guidance for the Industry, US Department of Health and Human Services, FDA, Center for Biologics Evaluation and Research CBER, April, 2020
- Use of Serologic Tests to Reduce the Risk of Transfusion-Transmitted Human T-Cell Lymphotropic Viruses Types I and II, Final Guidance for Industry, February 2020
- Draft Guidance for Industry: Recommendations for Requalification of Blood Donors Deferred Because of Reactive Test Results for Antibodies to Human T-Lymphotropic Virus Types I and II (anti-HTLV-I/II), CBER, September 2018
- Guidance for Industry: Nucleic Acid Testing (NAT) for Human Immunodeficiency Virus Type 1 (HIV-1) and Hepatitis C Virus (HCV): Testing, Product Disposition, and Donor Deferral and Reentry, US Department of Health and Human Services, Center for Biologics Evaluation and Research CBER, May 2010
- Guidance for Industry: Requalification Method for Reentry of Blood Donors Deferred Because of Reactive Test Results for Antibody to Hepatitis B Core Antigen (Anti-HBc), US Department of Health and Human Services, Center for Biologics Evaluation and Research CBER, May 2010
- Product inserts, InnoLIA-Syphilis/HCV/HIV/HTLV, Immunogenetics, Singapore
- Malaria Section, Australian Red Cross brochure, 2007
25/8/20