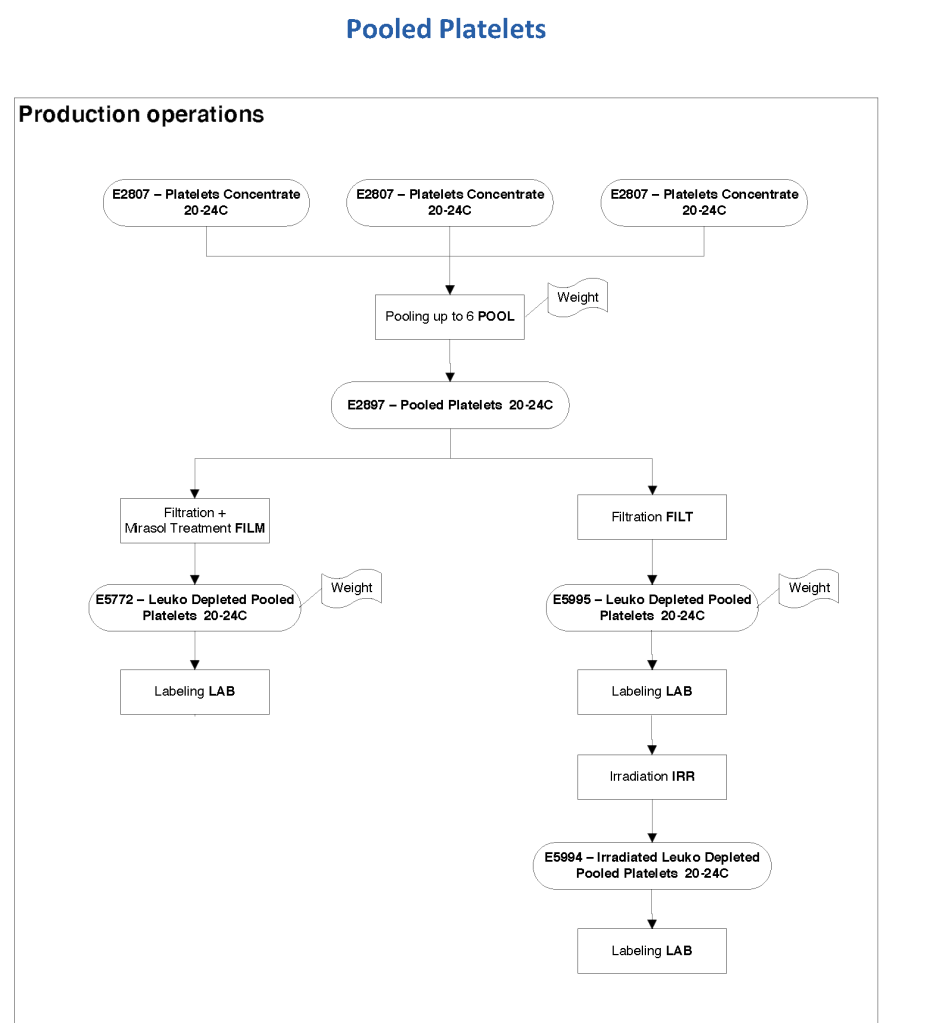
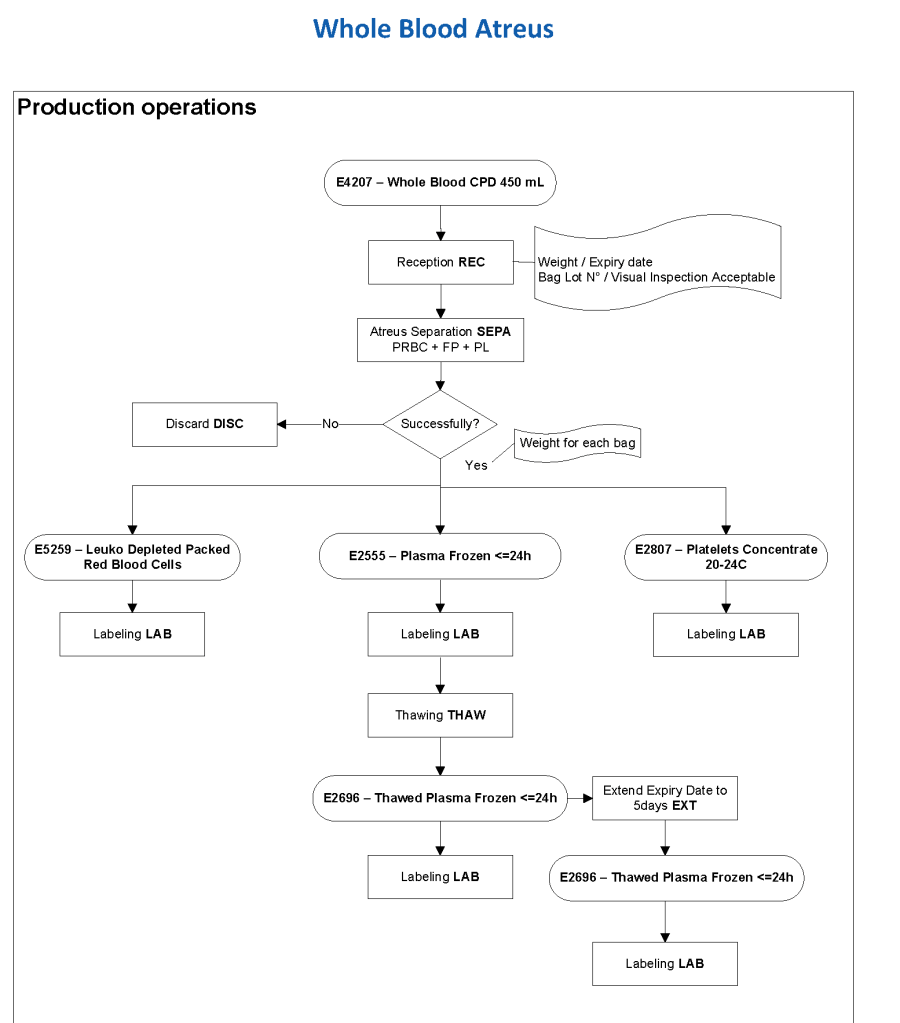
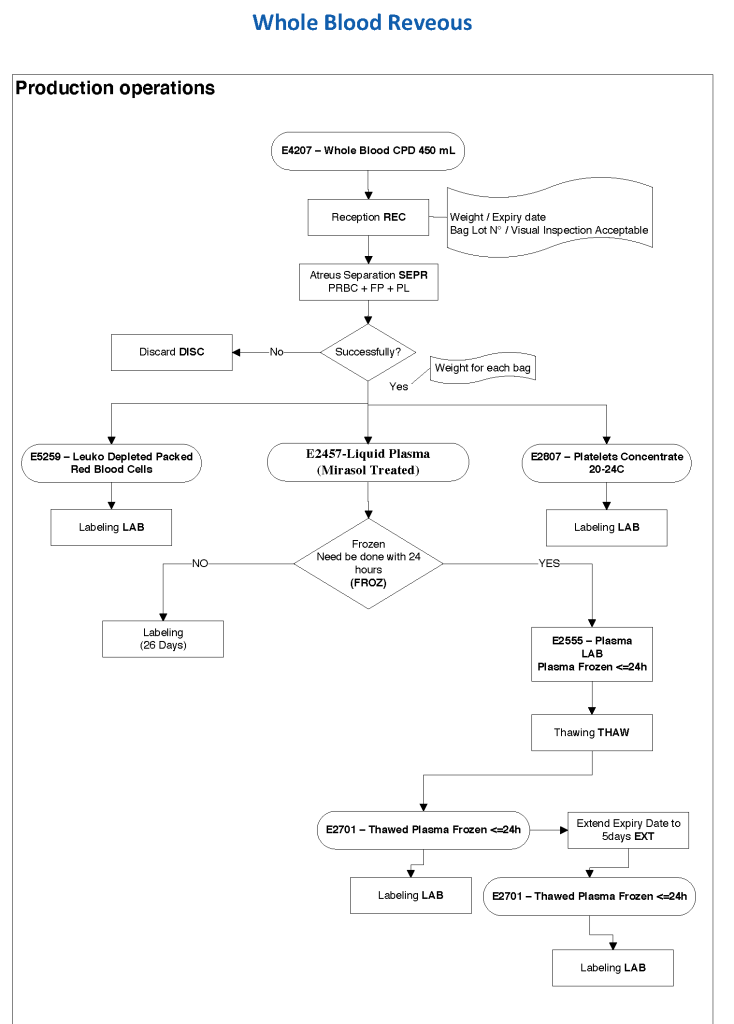
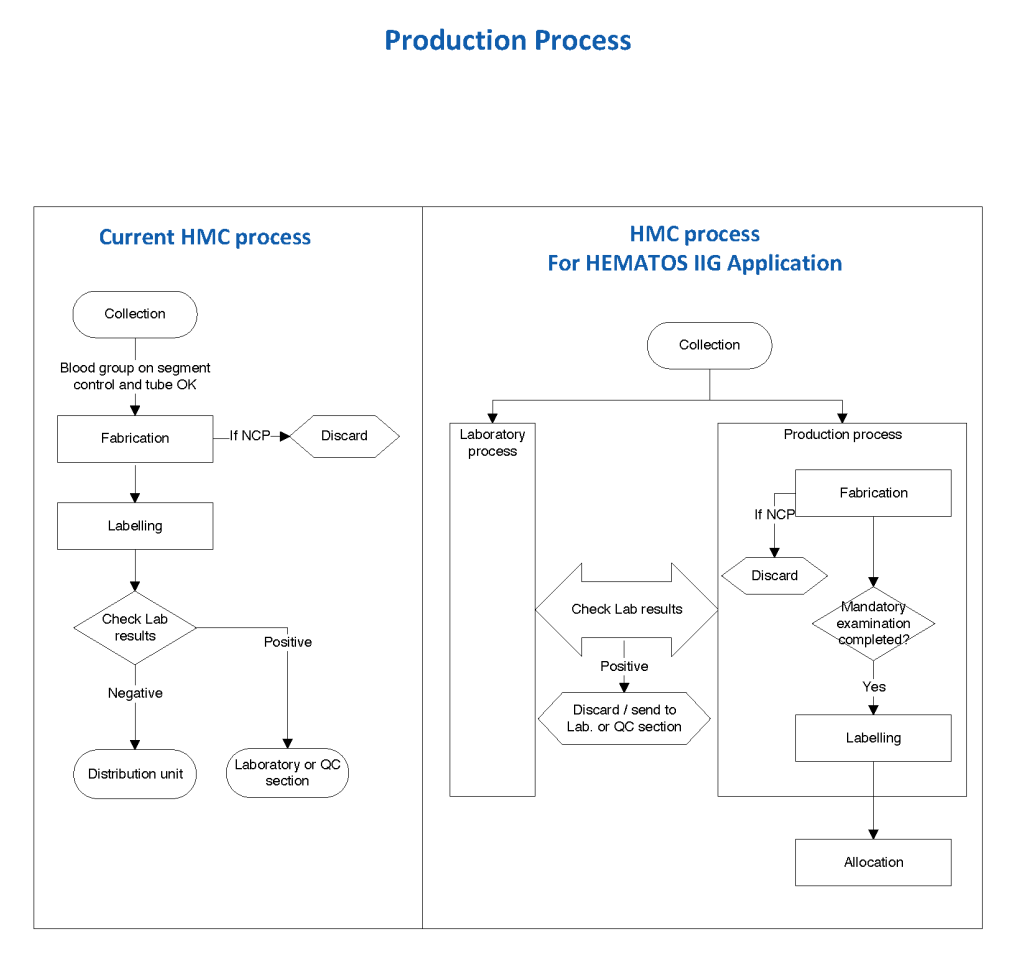
Exchange transfusions using reconstituted whole blood were much more common in the past. Much of the time IVIG now takes care of hemolytic disease of the fetus/newborn HDFN.
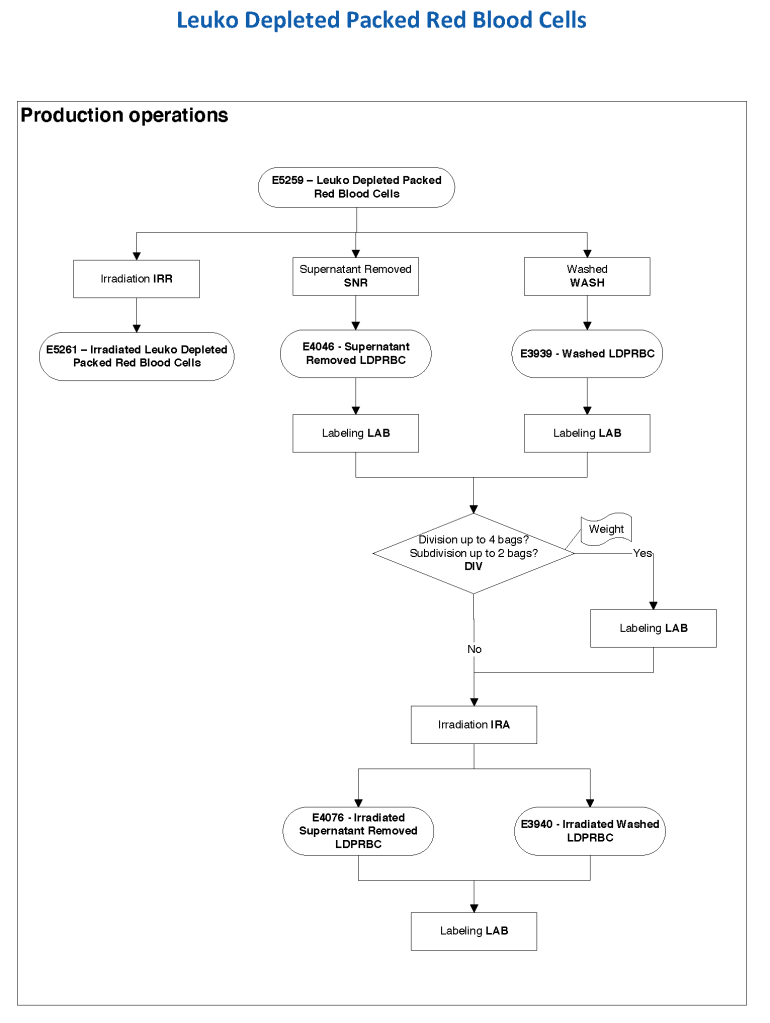
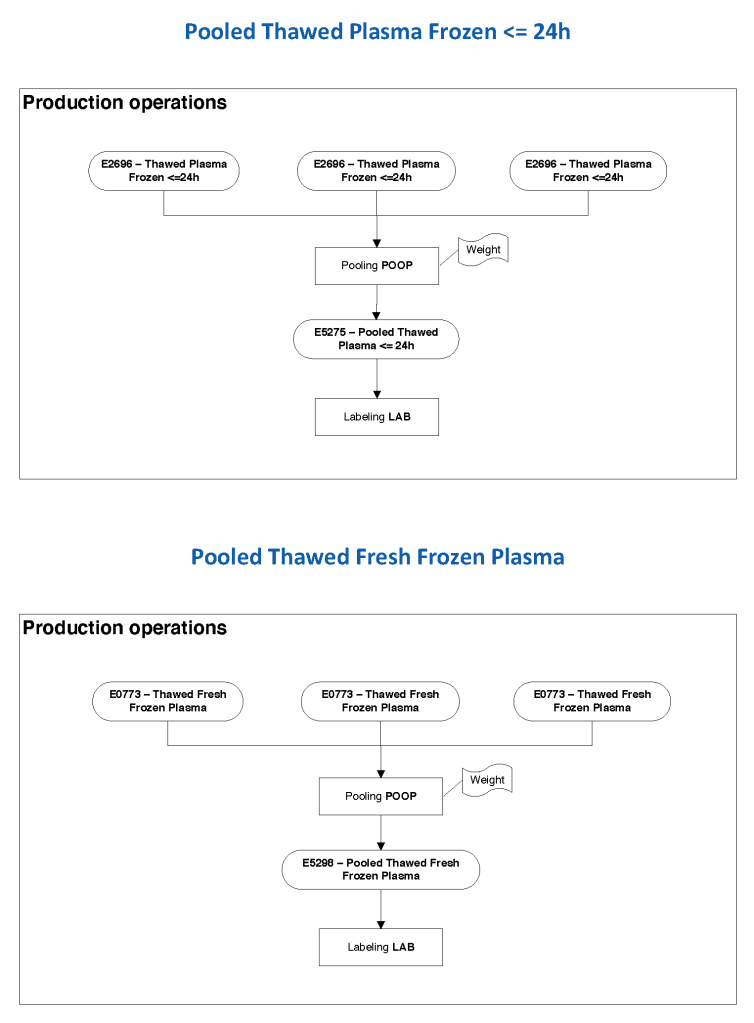
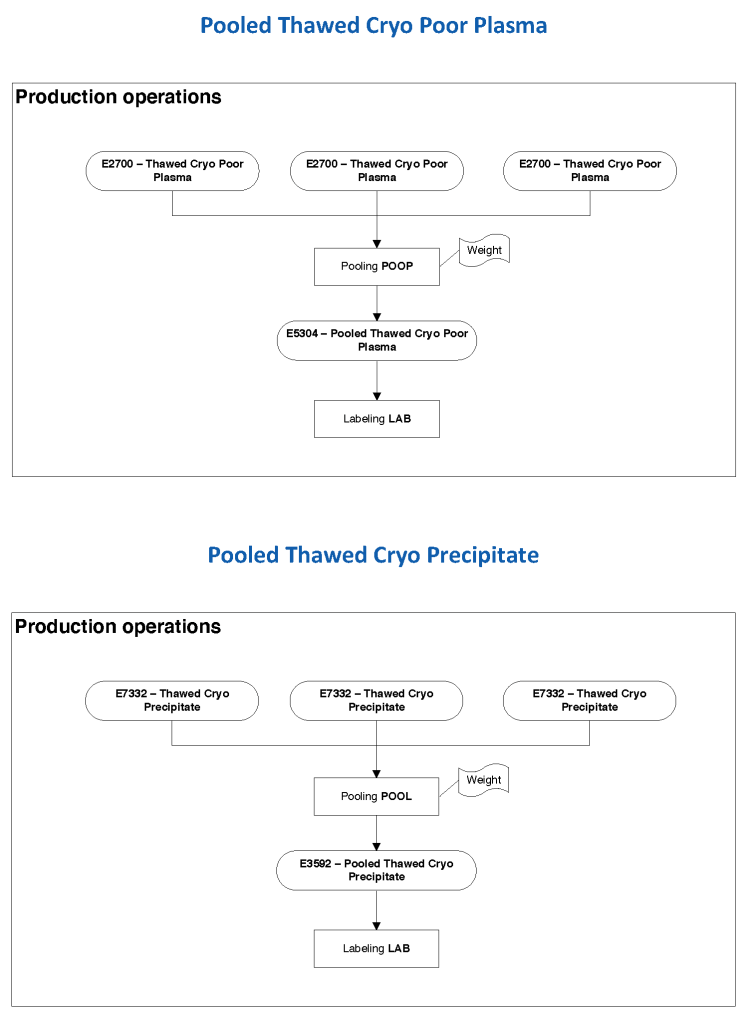
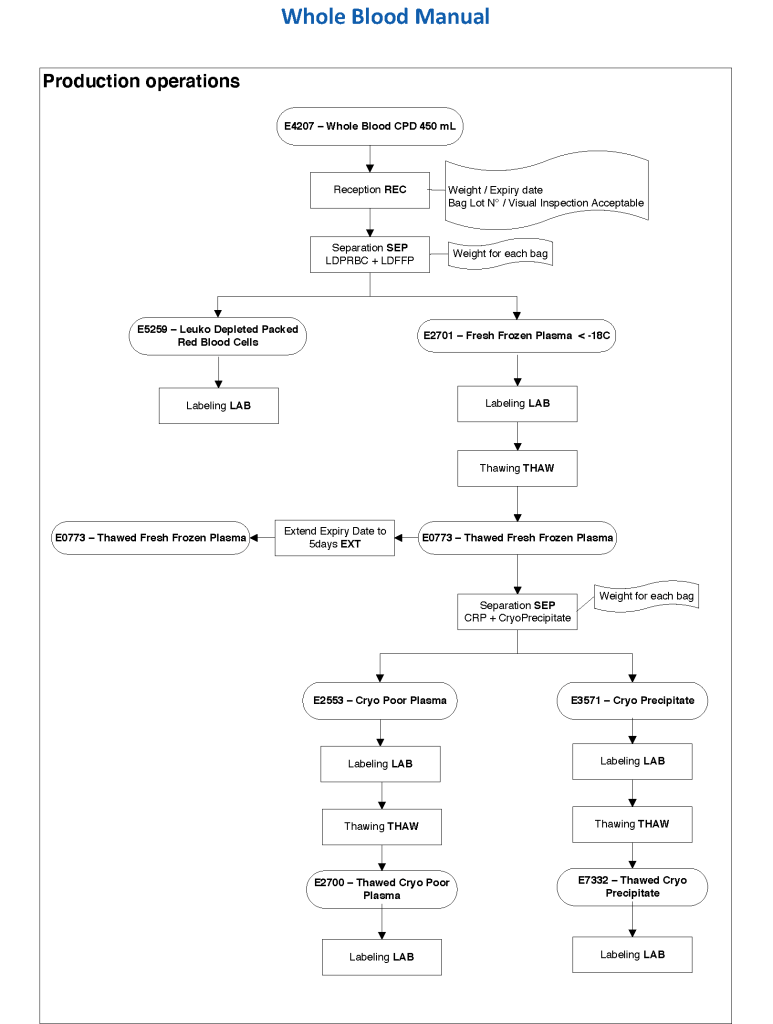
In Medinfo, we took a fresh (<= 14 day old) packed RBC in SAGM, group O, Rh-compatible and mixed it with a unit of group AB plasma—the desired hematocrit could be achieved by adjusting the amount thawed plasma that we added. The product could then be aliquoted and irradiated. Note that I medically chose to use either FP24 or FFP.
Here is the Medinfo process:
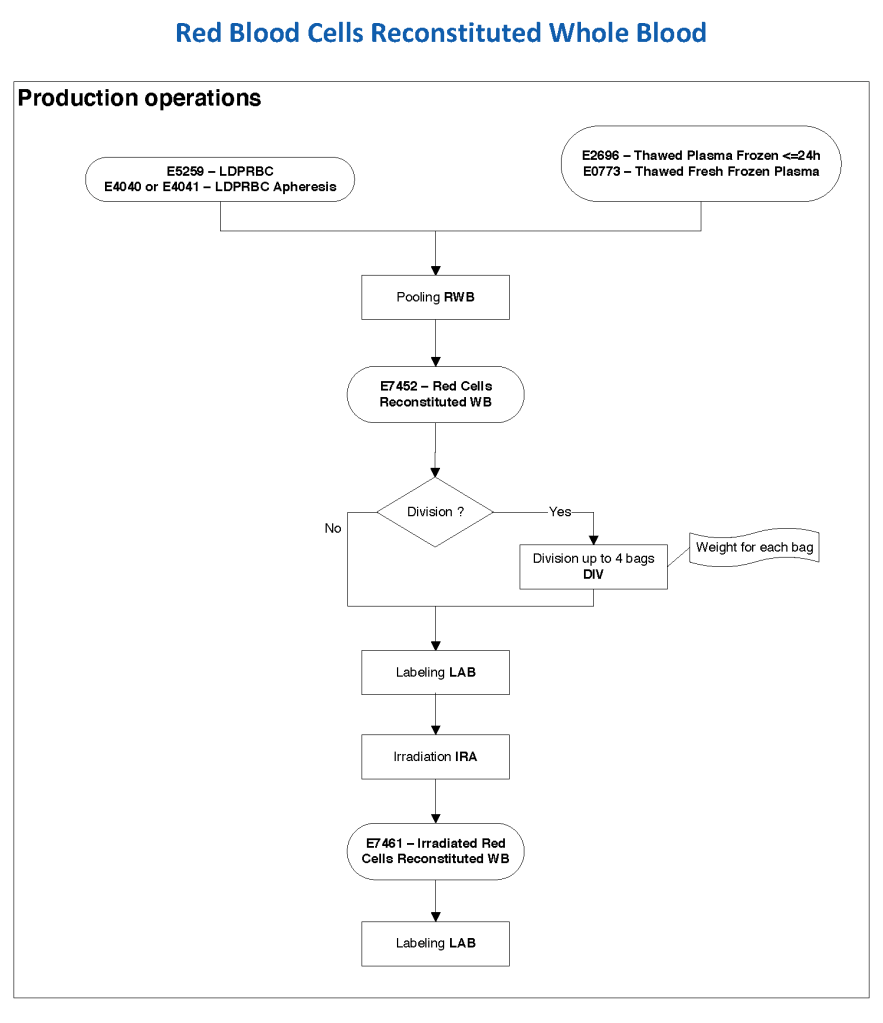
19/9/20