


Detailed discussion of Medinfo processes for all blood bank donor and patient services


Enumeration: 5.8.2
Principle:
AABB requires that all donors be notified and counseled of abnormal test results in a timely manner. Notifications should be done based on required AABB and/or CE protocols including the requirements for follow-up testing at specified intervals.
Process:
References:
Enumeration: 5.8
Principle:
AABB requires that all donors be notified and counseled of abnormal test results in a timely manner. Notifications should be done based on required AABB and/or CE protocols including the requirements for follow-up testing at specified intervals.
Policy:
References:
5.7.1 PROCESS: Donor Marker Testing
Process:
References:
Enumeration: 5.7
Policy:
References:
Donor physical examination, along with the donor questionnaire, are important both for donor and patient safety. In general:
Is it safe for the donor to donate?
Is it safe for the patient to receive the blood even if it is safe for the donor to donate.
Any donor who does not feel well must not donate. This may be the single most important step in ensuring a safe blood supply.
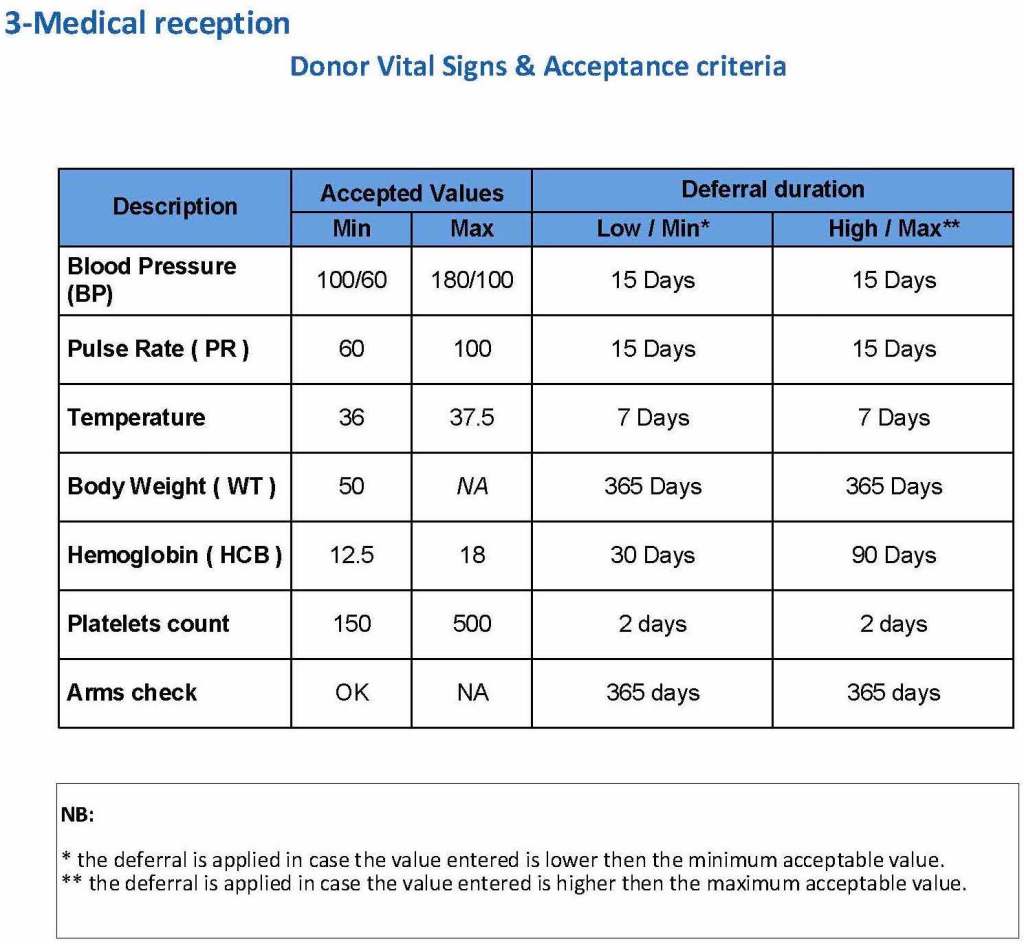
The donor physical examination includes the vital signs (blood pressure, pulse, temperature, heart rate, and temperature). I have attached a sample set of criteria for review. All are user-definable. Note how the arm examination is also included (looking for scarring, skin lesions, etc.)
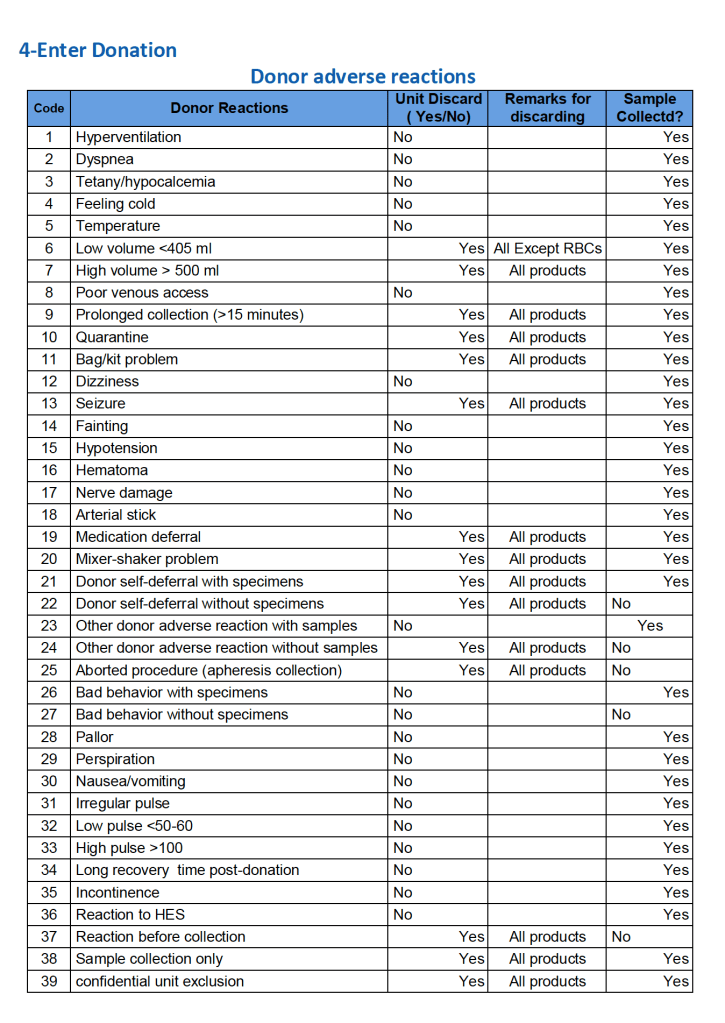
For all types of donations, there may be adverse reactions. These must be documented in the record along with the disposition of the donation. Will the donor need an extended deferral if the RBCs in the apheresis run are not returned? This can be built from the reaction documentation. Note the following sample table of reactions.
To Be Continued
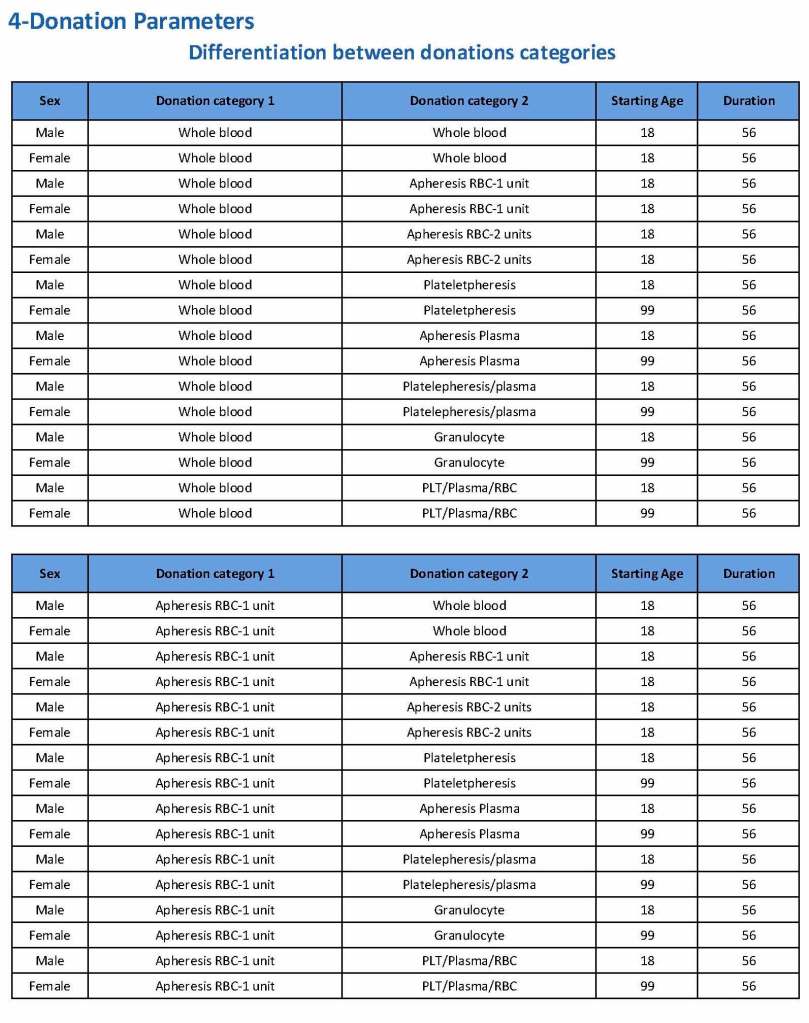
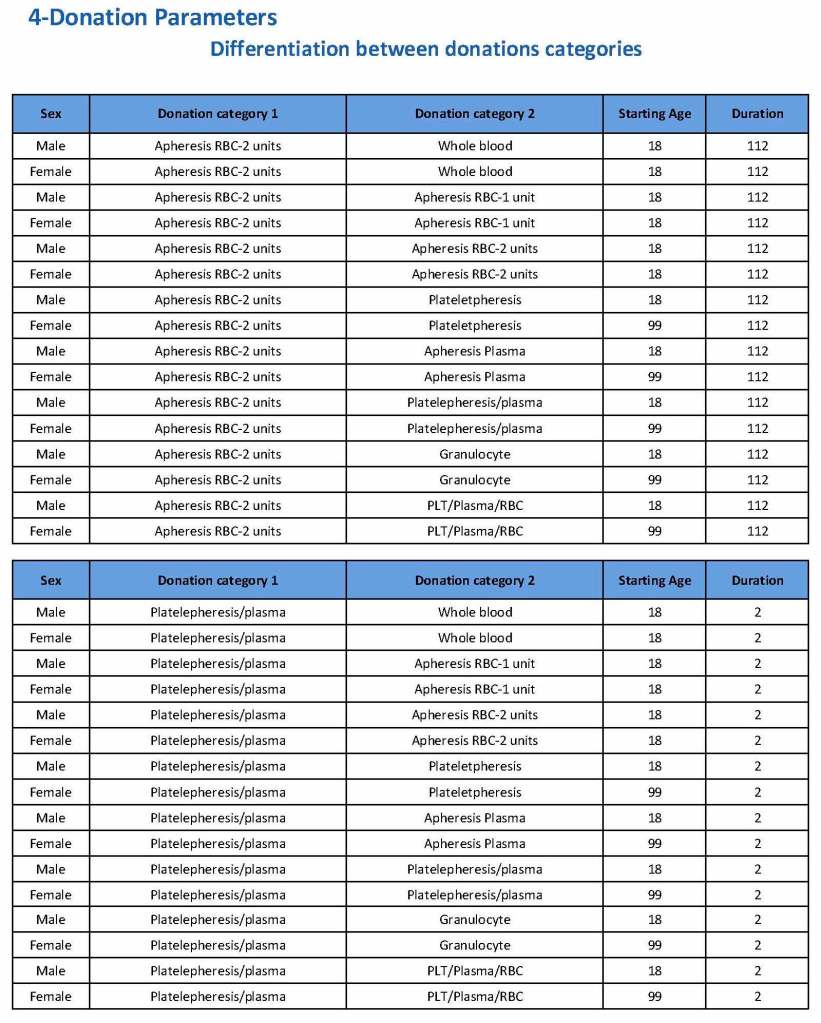
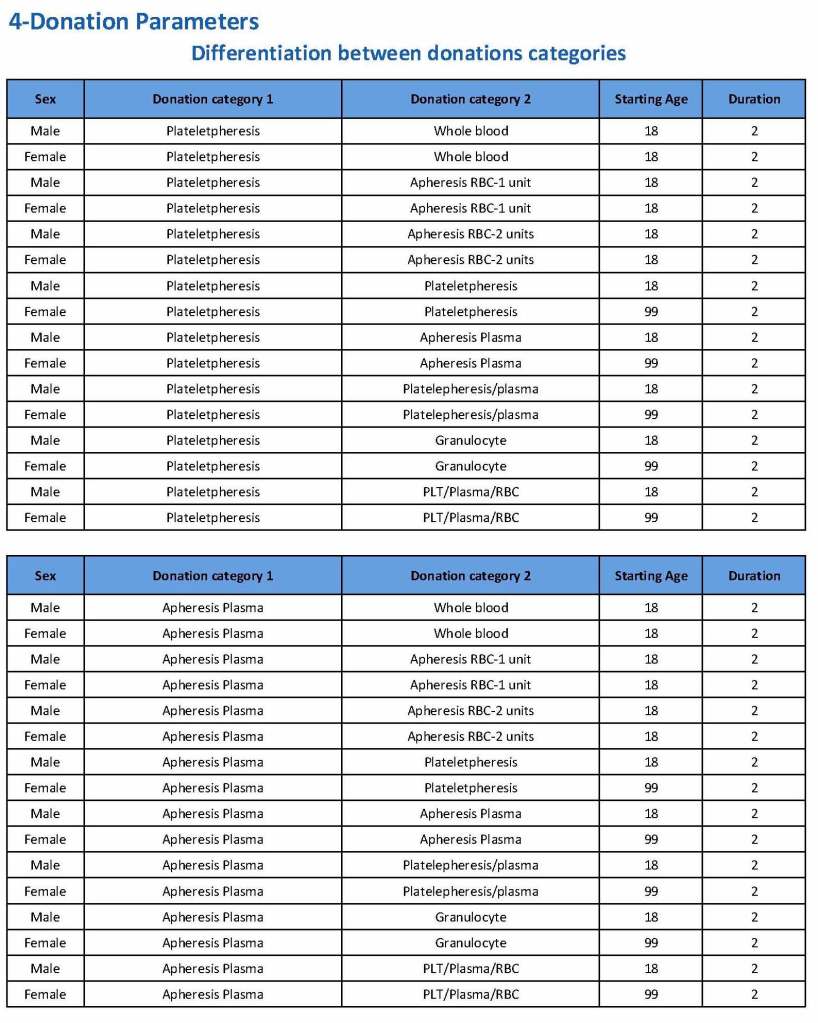
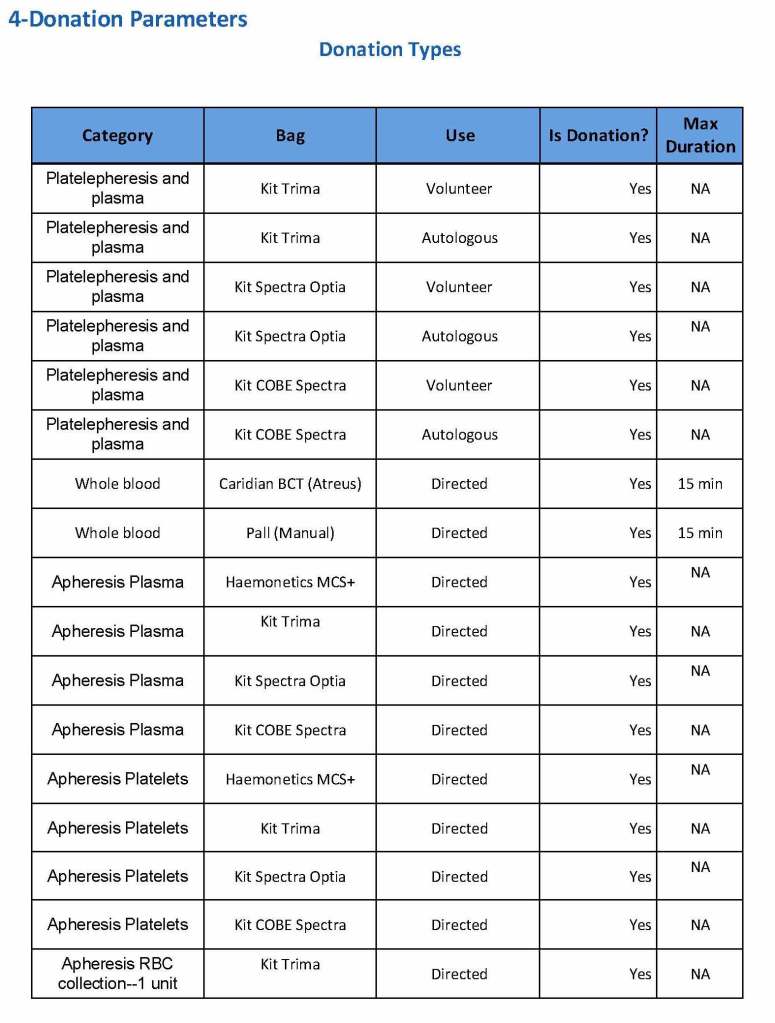
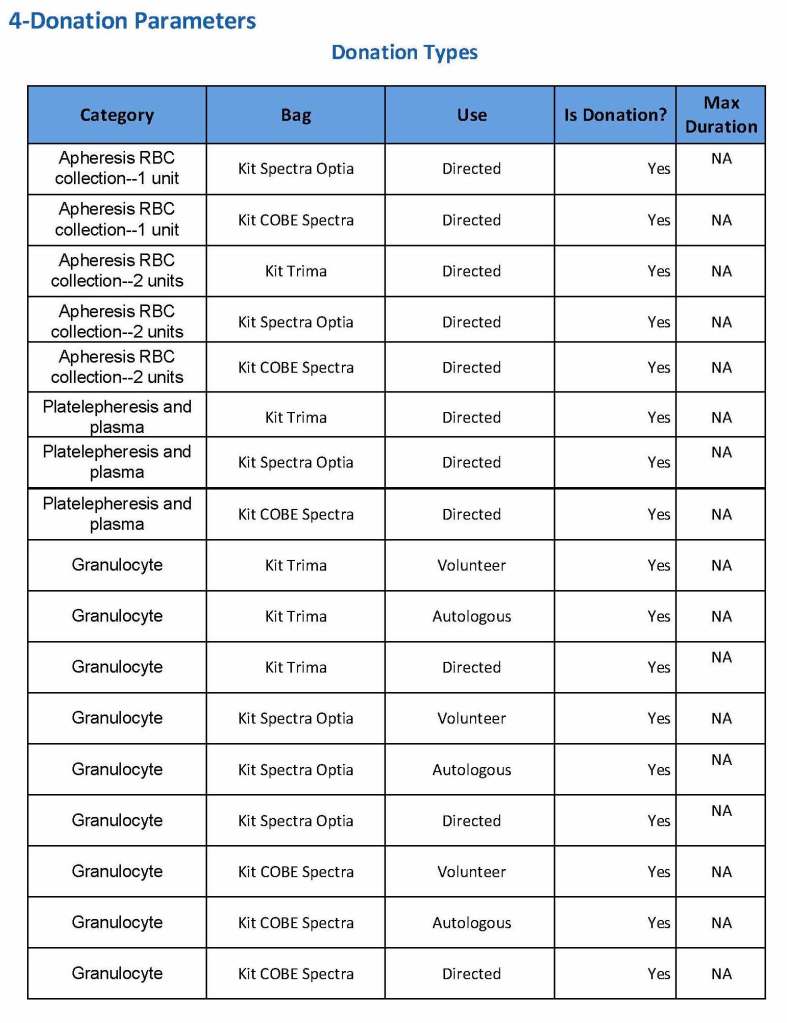
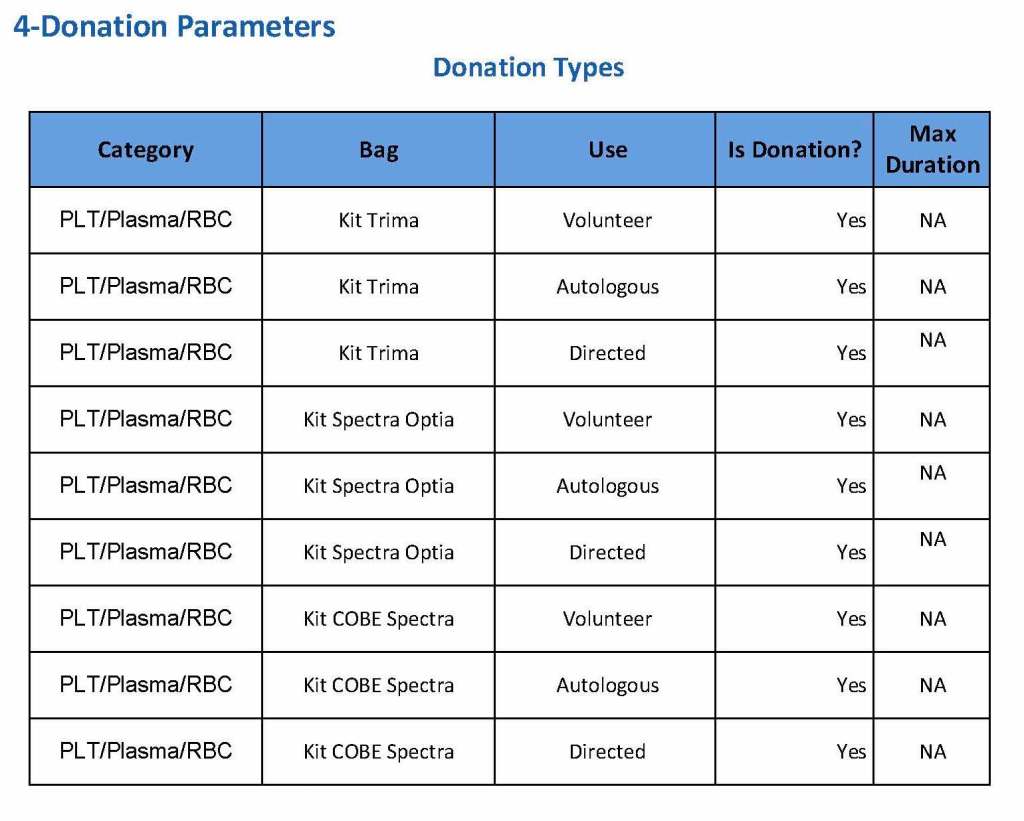
Donation can be whole blood or apheresis-based. The sex and age for each donation type is specified. At HMC, we did not accept females for platelet or plasma donations, so the starting age is listed as 99 years. Otherwise, in accordance with Qatari law, the starting age for donation is 18. All these parameters are user-definable, and a transfusion medicine physician can override the rules if necessary.
For each and every combination of donations, the deferral interval must be specified. Examples follow. The temporary deferral period is in days:
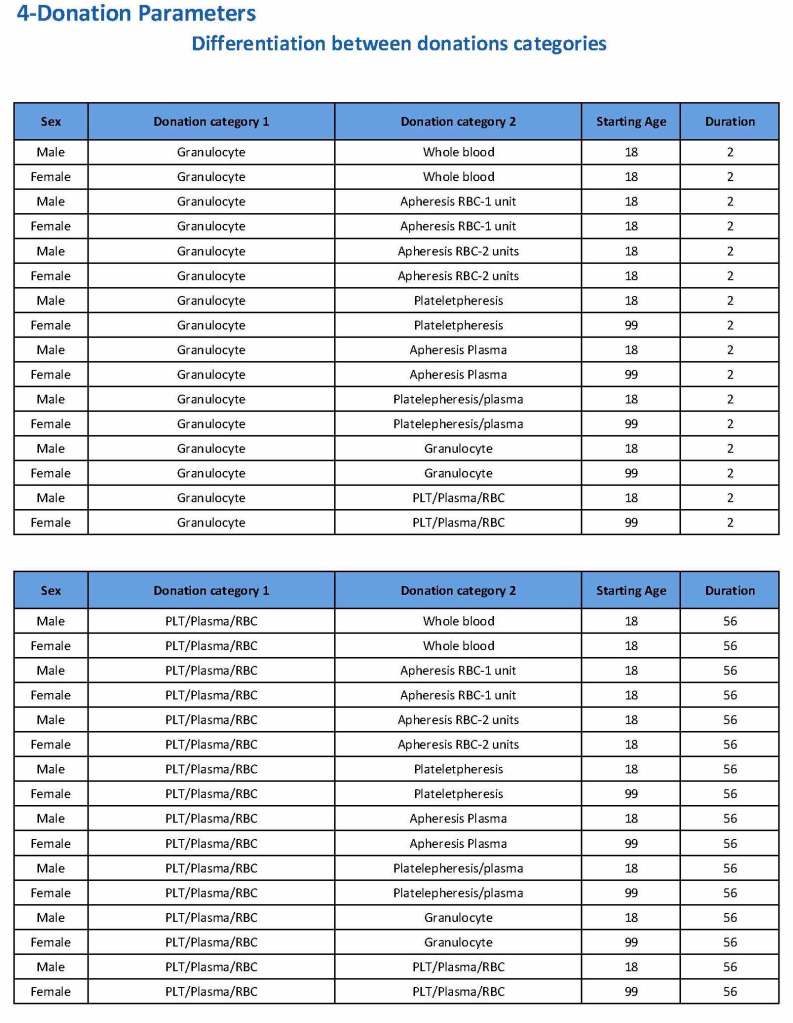
Previous donation whole blood, current donation whole blood: 56
Previous donor platelets, current donation whole blood: 2
Previous donation whole blood, current donation platelets: 56
Also note how for each possible combination there is an entry for male AND female. Females are restricted to whole blood donation and only RBCs will be made from the collection.
If there is a collection incident and the apheresis procedure is not completed, the interval will be set to 56 days. This will be covered in the post on donor adverse effect reporting.
To Be Continued:
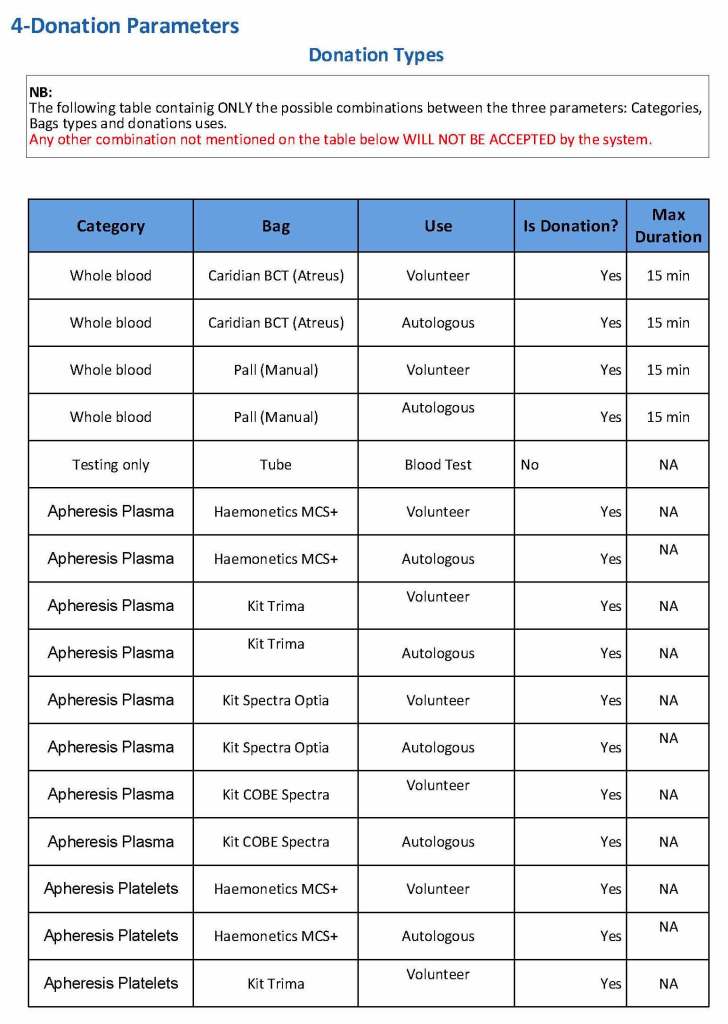
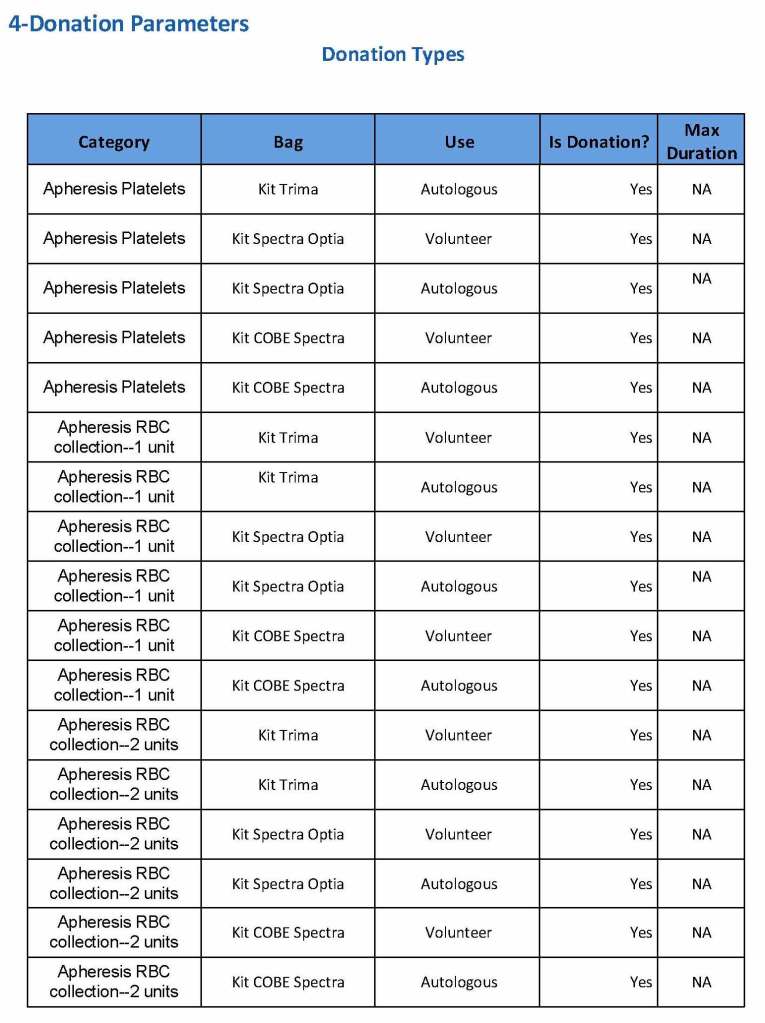
At the time of registration, the type of donation must be specified. In my last position, this could include whole blood for automated Reveos, whole blood for cryoprecipitate, plasmapheresis, COVID 19 convalescent plasmapheresis, plateletpheresis, concurrent platelet and plasmapheresis, concurrent platelet, plasma, and RBC apheresis, RBC apheresis-one unit, and RBC apheresis-2 units.
There is also a specimen-only donation without actual collection that includes database check, assignment of an ISBT specimen number, donor questionnaire, physical examination, and specimen collection only..
We specified which bag or kit could be used for each type of donation so when it was selected, only that bag type would be accepted by Medinfo
For each of these types we must specify what type of donation is permitted: volunteer, autologous, or directed.
Finally, we must indicate the maximum length of the procedure permitted. This applied to whole blood only and we set this at 15 minutes—this is user definable.
The following are a sample set of parameter settings for the above:
Note how we included contingencies for old bag sets and equipment (that we later discontinued) and for granulocyte collection (which we did not actually perform).
To Be Continued:
The potential donor enters the collection area. He takes a number and waits to be called. When called, he shows a picture identification card with a unique alphanumeric sequence. This is entered into the donor module software and the system checks the donor deferral database for temporary and/or permanent contraindications. If none are found, a consent form with an ISBT specimen number is generated.
In this post, we will consider:
Registration:
In the Middle Eastern region, multiple languages are used. Although Arabic may be the main language, not all the registration staff may speak it. English is commonly used as the main work language. The date may be entered as Common Era (Gregorian) and/or Hijri.
An issue is that for native Arabs, the only precise, unambiguous name spelling is in Arabic. English transliterations vary. Example, Muhammad in Arabic is very simple to write, in English it may be rendered as Mohamed, Mohammed, Muhammad, etc. The donor’s name should be recorded exactly. How do you register when the staff do not speak or type Arabic?
Fortunately, I have worked with software that is in UNICODE, meaning that the data does not have to be restricted to English (I wonder why the hospital information system we had at one institution could be sold in the Middle East and not have this capability!). That means we could perform registration and donor questionnaire tasks in multiple languages, and preferable the native language of the donor. We could even prepare database reports in Arabic.
Medinfo had an elegant solution to the registration process. It read the local identity card issued by the Ministry of Interior and accessed (read-only) the demographic data on that donor. Just by reading the bar code one received both English and Arabic name fields:
This would generate the demographic fields in the registration:

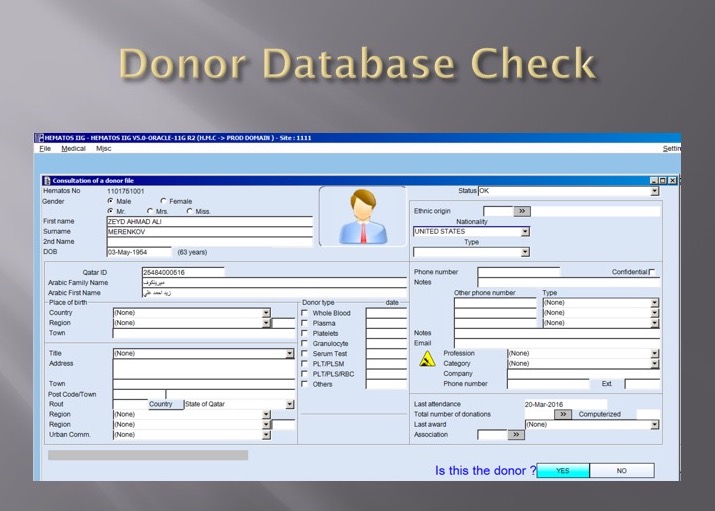
The blood bank software would check the national donor deferral database and list any deferrals/contraindications to donation and the next eligibility date. It would also list what type of donations were permitted (e.g. for females, only RBCs could be collected and processed: if a whole blood unit was collected, then the platelets and plasma would NOT be permitted to be processed and were discarded.)
Medinfo used a unique key field, the Medinfo Hematos Donor ID for the database. This was not the same as the national ID card. All records were indexed against this number.
Donor Deferral Database:
Medinfo imported donor data from a previous system and added this to its own database. Thus, there was only one database to check. The database listed all previous donations: dates, type, status (complete, aborted). Any contraindications would be prominently shown in RED.
Donor Consent and Assignment of Donor Unit (ISBT Specimen) Number:
If there were no contraindications, Medinfo generated a donor consent in English and Arabic and the unique donor unit number for the current encounter:

Registration Parameters:
Medinfo enforced registration according to the format of the identity card. The donor ID format was built into Medinfo. If the entry deviated from this, it was rejected and registration could not continue:
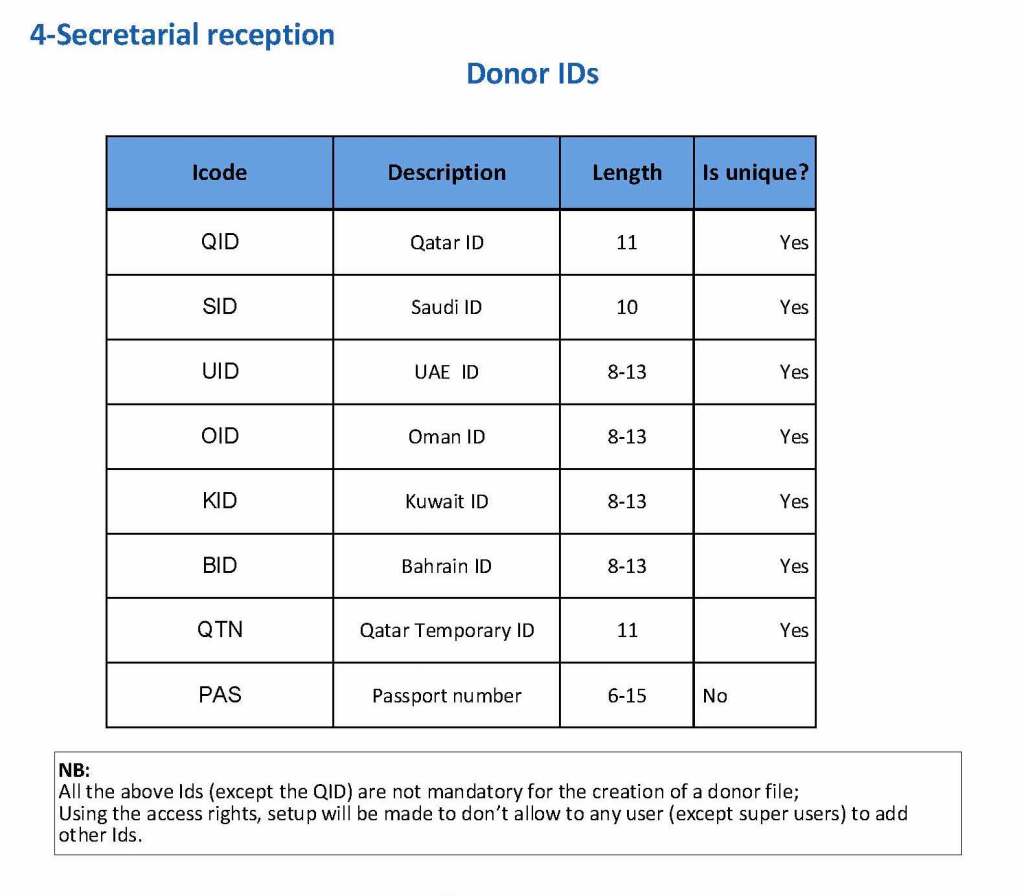
The registration type would be selected (volunteer, autologous, directed, or paid). In Qatar, paid donations were not permitted:
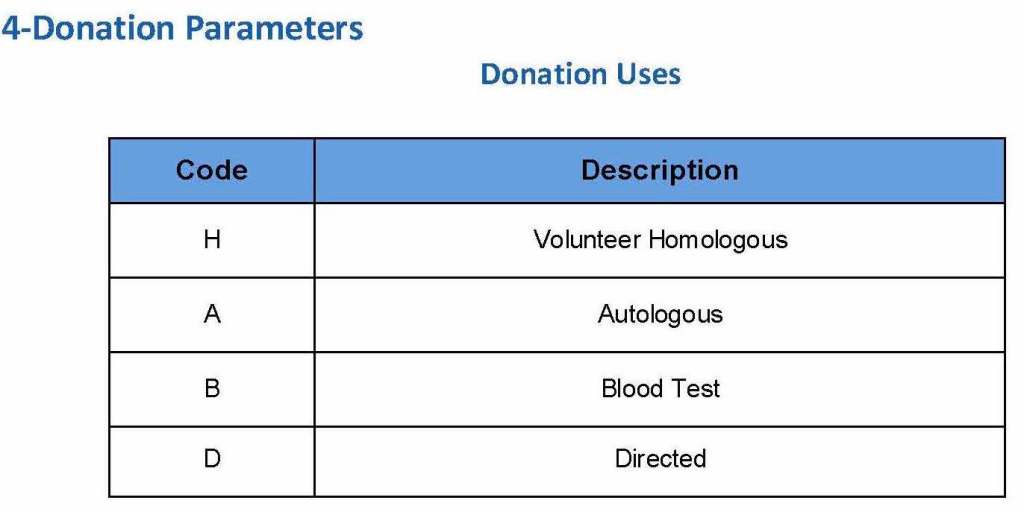
Next, the donation type had to be selected:
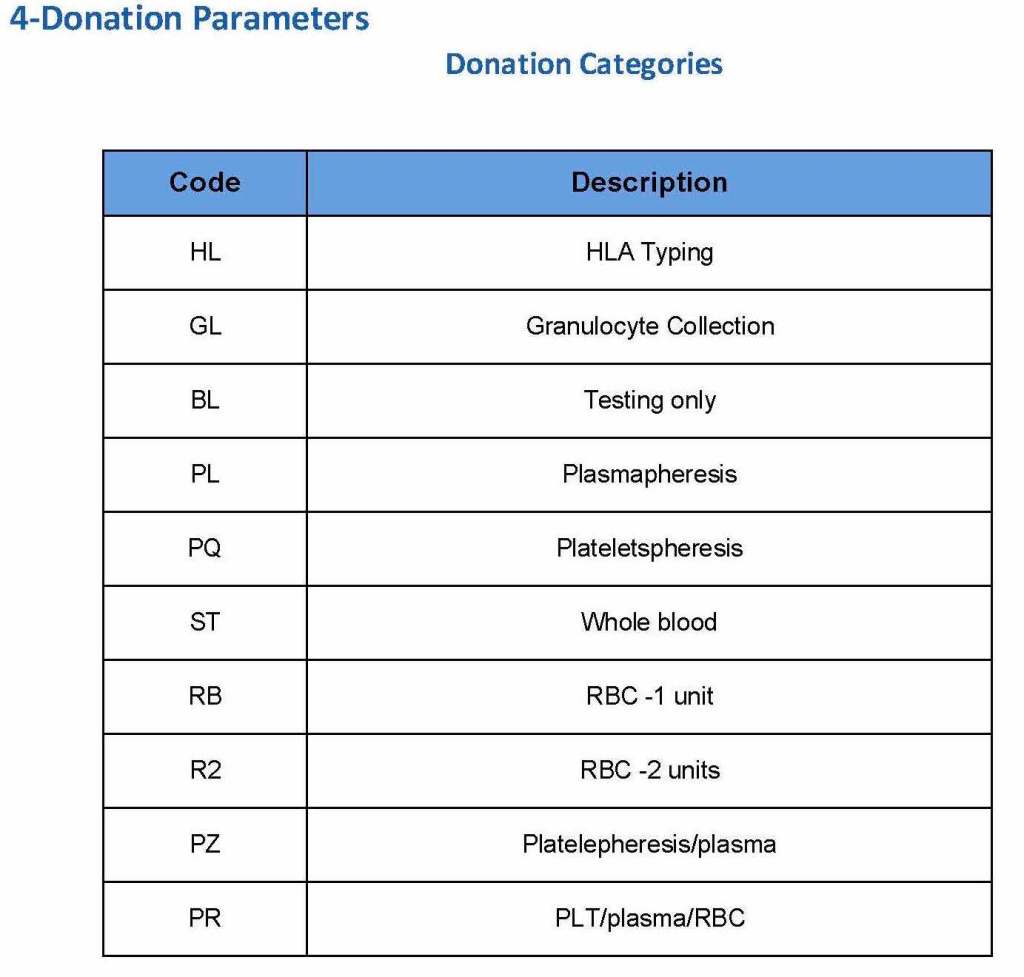
At the time of registration, the type of collection bag (or kit if using Reveos) was automatically set in Medinfo. I will consider this further in the next post of this series to determine eligibility based on the previous donation interval.
At each donation site, the allowable types of donations and kits could be set. Based on the donation parameters above, staff could not select the wrong type of bag/kit (e.g. an apheresis kit for a mobile donation).
To Be Continued:
This is a first is a series of detailed posts of how I collaborated with Medinfo (Nice, France) to build customized donor software for both Saudi National Guard Health Services and Hamad Medical Corporation Doha.
In particular, we were using a non-turnkey software which could be built to order. If we didn’t know what we were currently doing, how could we build something better?
At both sites, we had good manual systems in effect and prepared detailed mapping of the current state. We reviewed our variance reports to see where we needed to bolster the system and improve the critical control points (page 2).
We studied the software options and prepared a draft Medinfo future state (page 3), from which we started to build the system. We did this in small stages so we could test it and adjust our settings as needed—without being charged extra (unlike a general laboratory software I had been working with at the same time).
To do this, I engaged early a team of my most computer-literate staff to work as Super Users. In the Donor Center, this consisted of nurses and technologists.
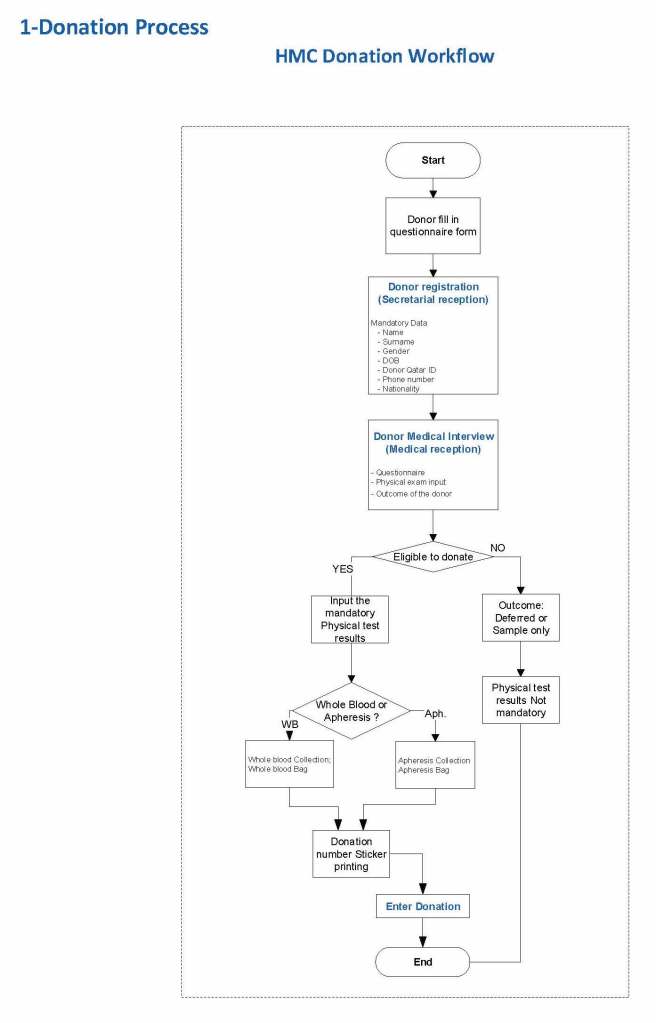
We then prepared a Medinfo flow chart overview from this:
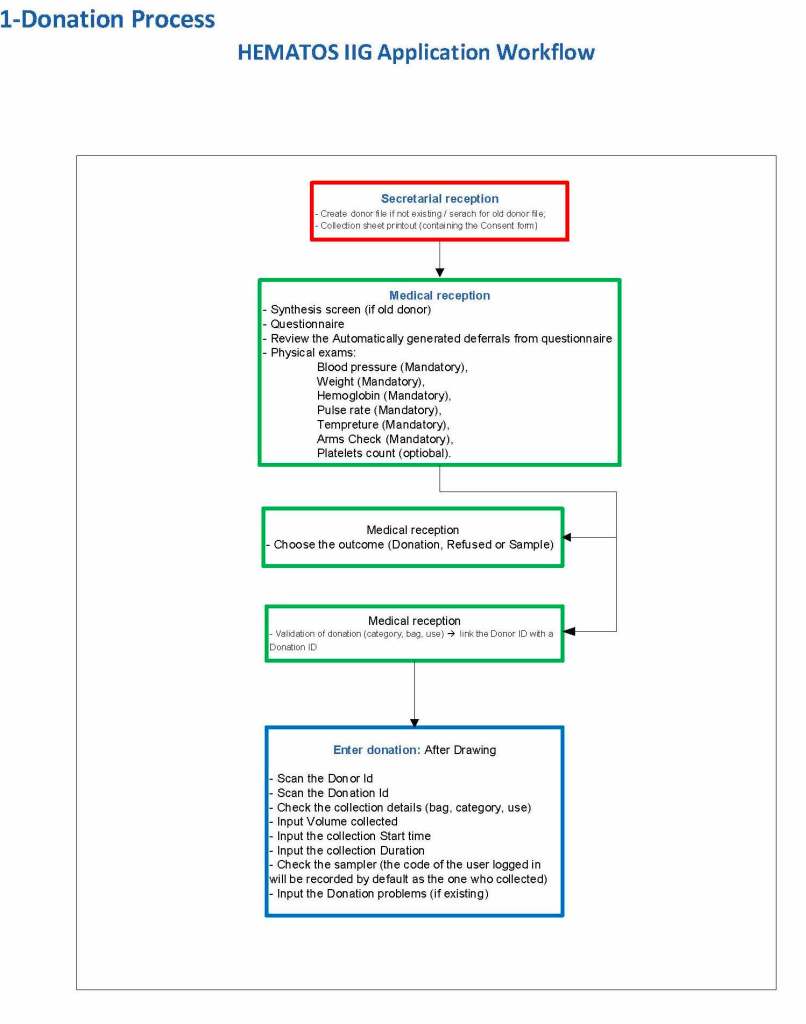
To Be Continued: