Objectives:
- Donor criteria based on AABB standards
- Is it safe for the donor to donate?
- Medical history
- Current medical conditions
- Past medical conditions
- Medications
- Vaccinations
- Travel history
- High-risk behaviors
- SARS/MERS
- COVID-19 convalescent plasma CCP
- Prion diseases
- Donor medical examination
- Is it safe for the recipient to receive the donor’s blood?
- Donor registration issues
- Positive identification
- Donor deferral database
- Donor phlebotomy
- Safe volume to donate
- Anticoagulant-preservative solutions
- Time limit for phlebotomy
- Post-donation care
- Donor reactions—Dx and Rx of the following:
- Vasovagal
- Seizures
- Air embolism
- Arterial stick
- Hematoma
- Donor Apheresis
- Plateletpheresis
- Plasmapheresis
- Plateletpheresis with concurrent plasma collection
- RBC collection
- Combined platelet, plasma, and RBC collection
- Autologous donation
- Predeposit
- Perioperative
- Intraoperative
- Postoperative
- Donor self-deferral
- Therapeutic Phlebotomy
- Therapeutic Apheresis
- Therapeutic plasma exchange/plasmapheresis
- Leukapheresis
- Thrombapheresis
- Red cell exchange
- Stem cell collection
- Column absorption technologies
- Clinical indications
- Writing orders for above procedures
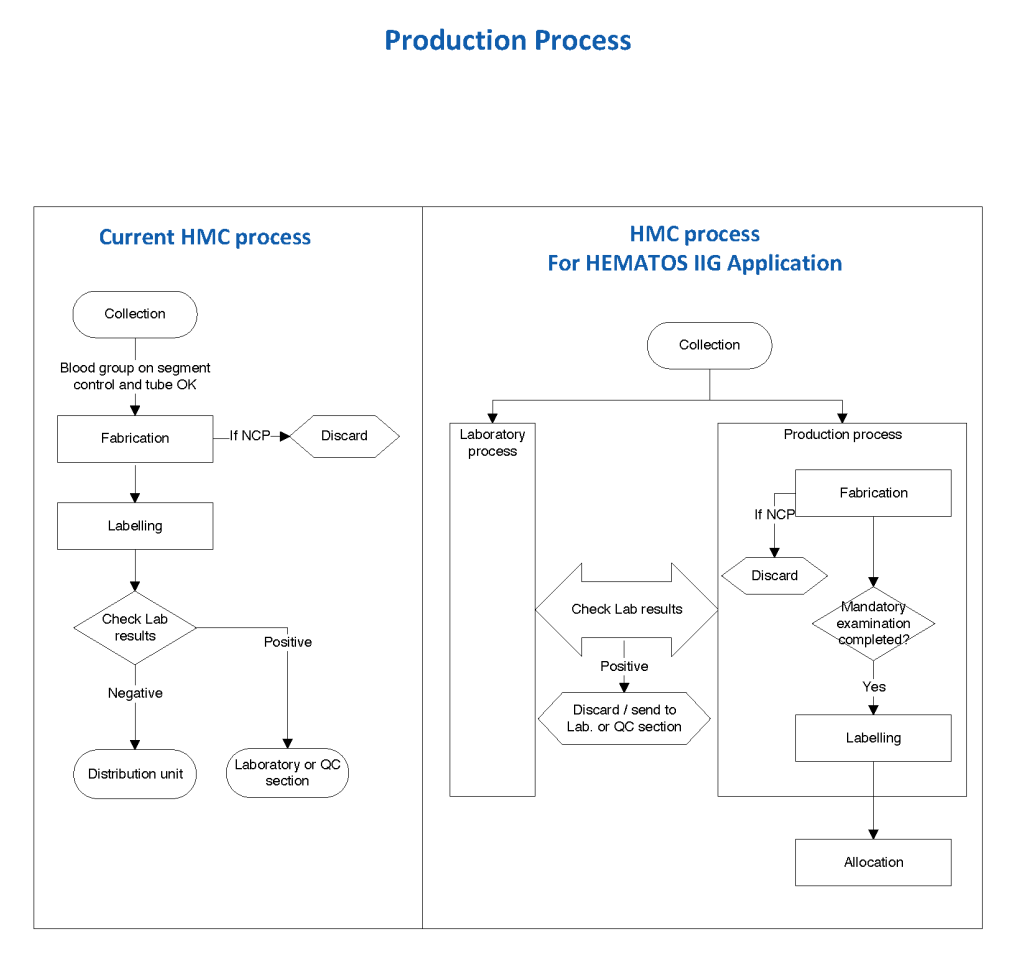
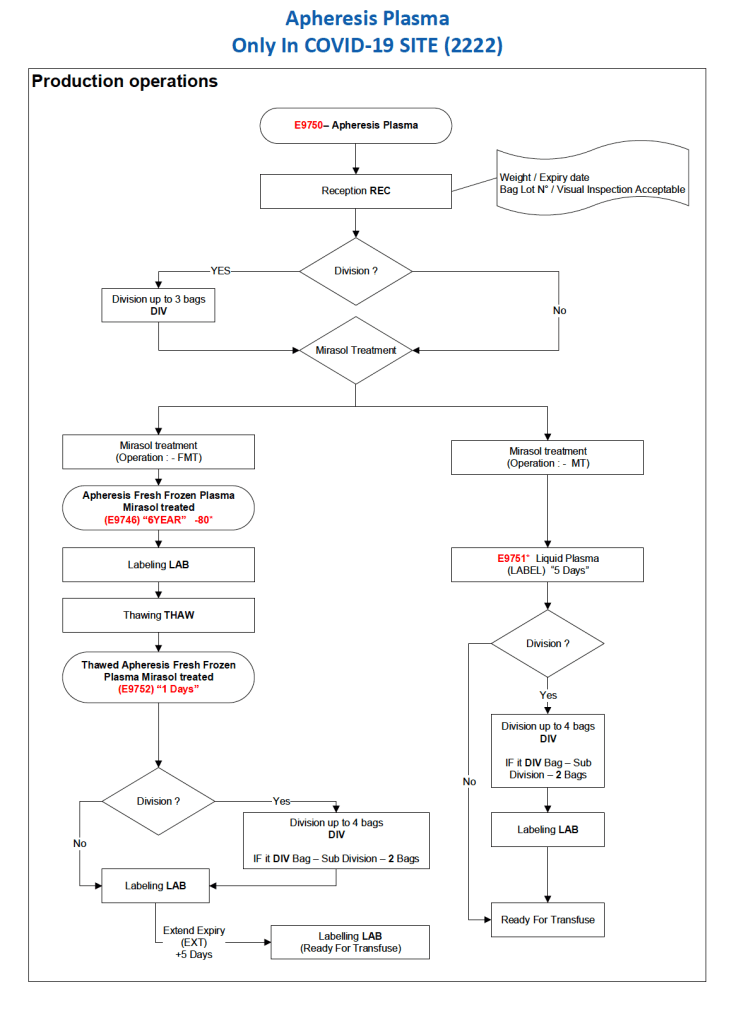
- Component Processing:
- Manual
- Automated—Reveos
- Pathogen Inactivation Mirasol
- Buffy coat vs classic platelet-rich plasma platelets and pools
- Platelet Additive Solution PAS
- FFP, FP24, thawed plasma
- Cryoprecipitate
- Cryo-poor plasma (plasma, cryoprecipitate-removed)
- COVID-19 convalescent plasma CCP
Clinical Responsibilities (after proven competence):
- Triage of donor requests
- Handling of donor reactions
- Approval of therapeutic phlebotomies
- Assistance with therapeutic apheresis
Assessments:
- Pre-training/baseline
- Competency documentation for clinical responsibilities (#11 above)
- Post-training
Working Hours:
- 0900-1700, Saturday through Wednesday
- Must carry pager for clinical responsibilities
Reviewed 17/8/20