Processes and Software Building—Part 19
In the previous post I outlined how Medinfo handled antibody screening and identification. This post reviews how antigen matching is used based on these results.
There are two modes, regular and emergency. If the patient has not had at least two ABO/D determinations and/or does not have a recent antibody screen, then emergency mode must be selected with its own rules. Otherwise, the regular mode applies.
Regular Mode:
In general, if there is a clinically significant antibody, an RBC unit which has not been matched for the corresponding antigen or has the corresponding antigen cannot be routinely selected. However there is a hierarchy here also:
- Absolutely prohibited release—no one can override the logic (e.g. giving group O to a patient with anti-H)—not even the transfusion medicine physician can override this
- Restricted release—only certain staff can release the incompatible or untested unit (e.g. giving C-positive unit to someone with anti-C)
- Least-incompatible for WAIHA: requires transfusion medicine physician approval
- Informational release: authorized staff may release antigen-incompatible or untested unit but a pop-up menu appears and asks them to accept (e.g. Lewis untested unit in a patient with anti-Lea).
- Antigen-specificity matched—the usual mode for patients with antibodies
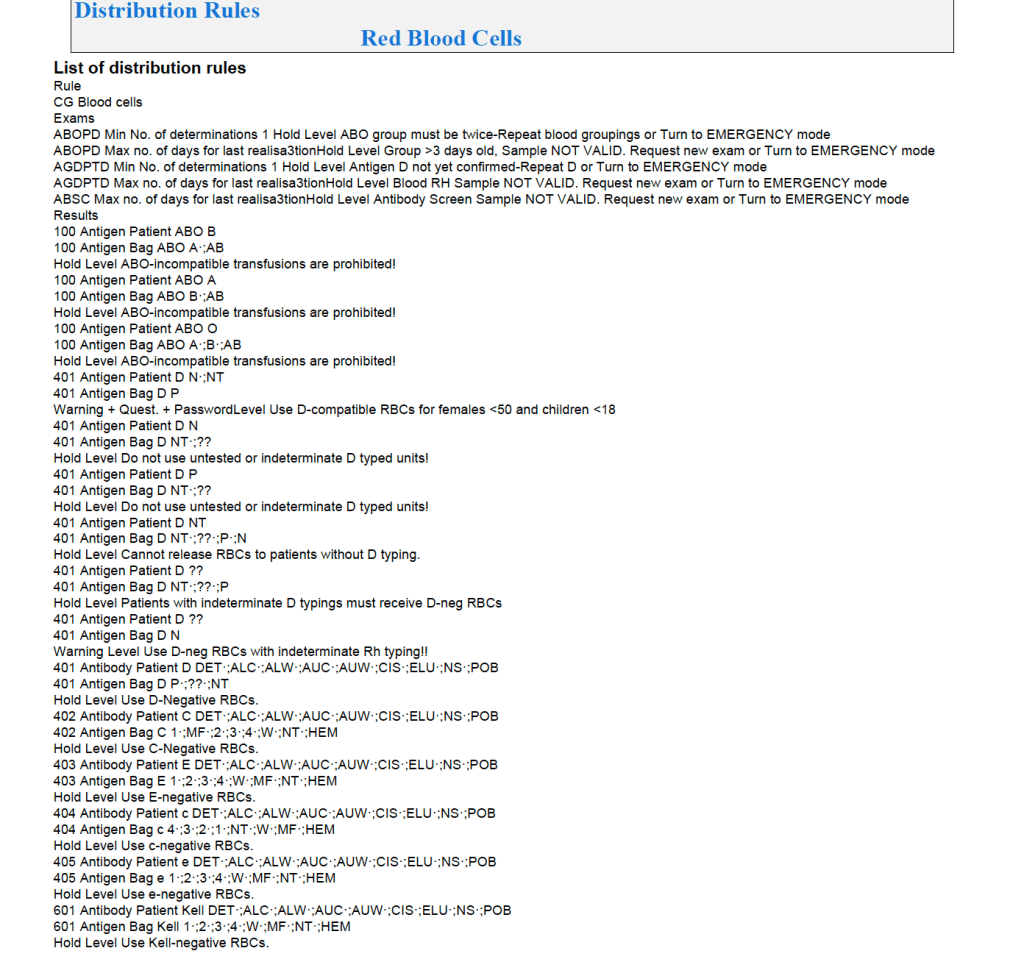
Examples of Regular Mode rules follow (these are not the complete lists but just provided to show the complexity of the process).

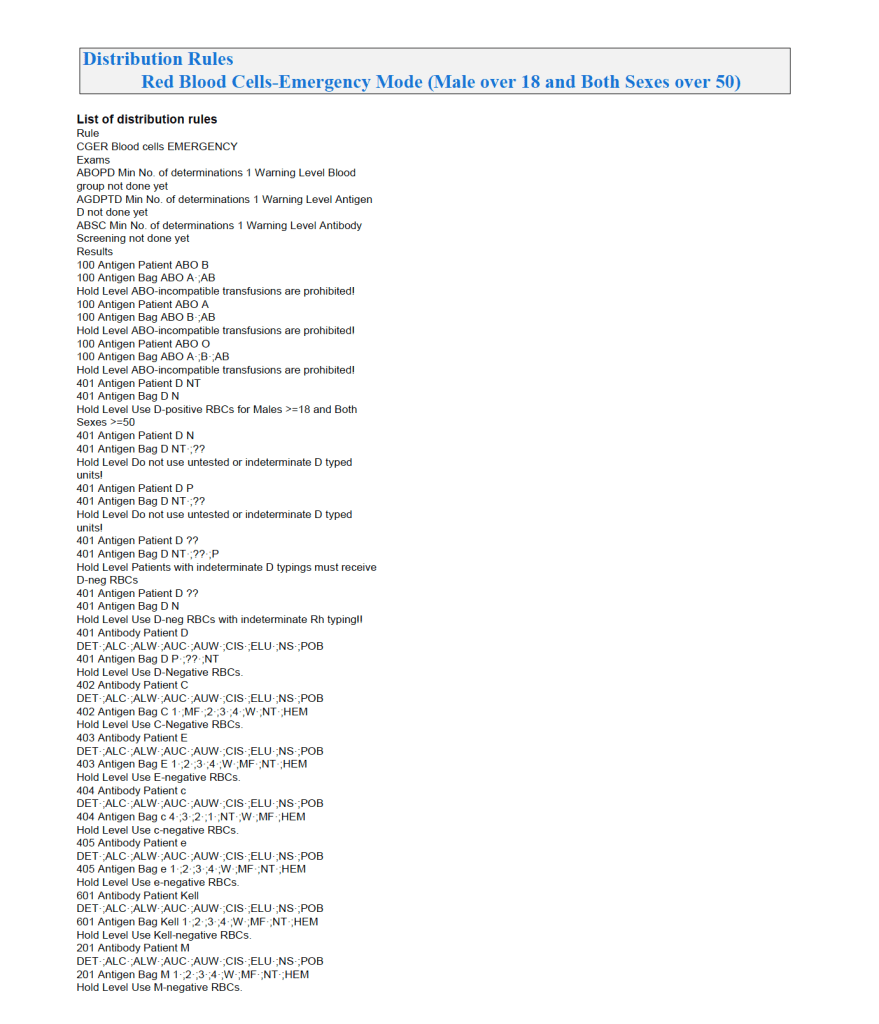
Emergency Mode:
This is much more restricted for selection of ABO/D and other antigen typings. An example follows:
A similar hierarchy exits for platelet and plasma allocation and will be considered in a future post.
To Be Continued:
18/7/20