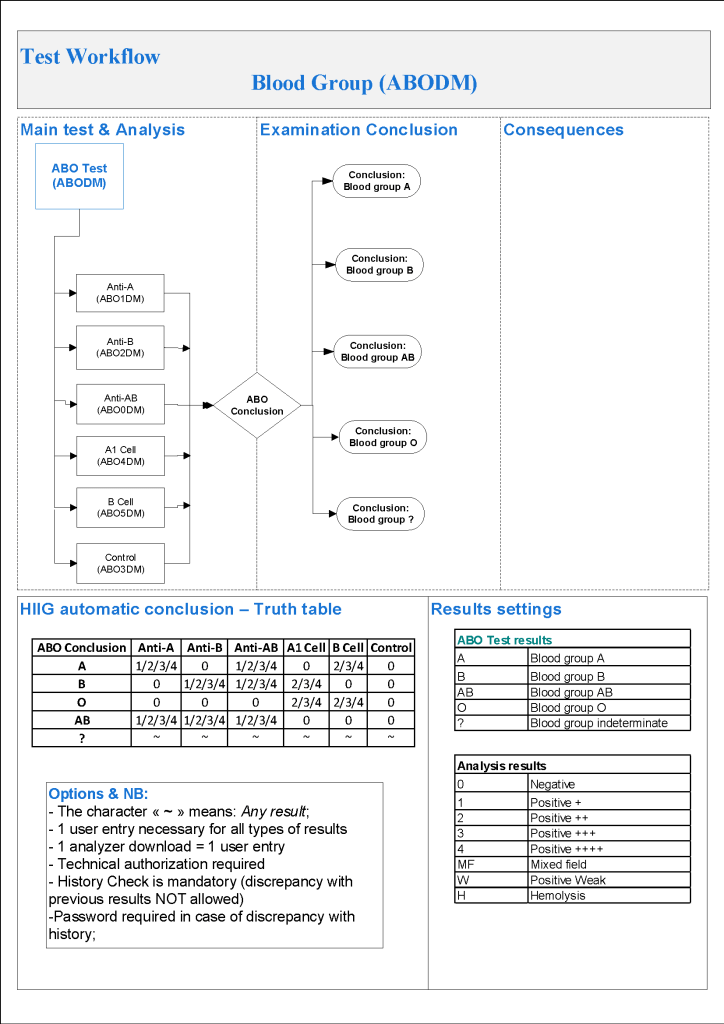
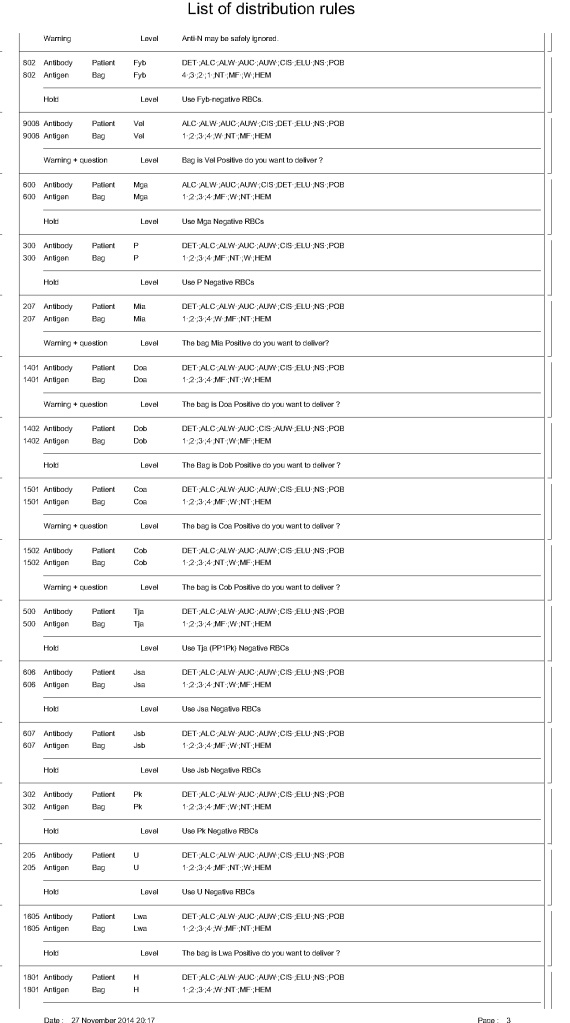
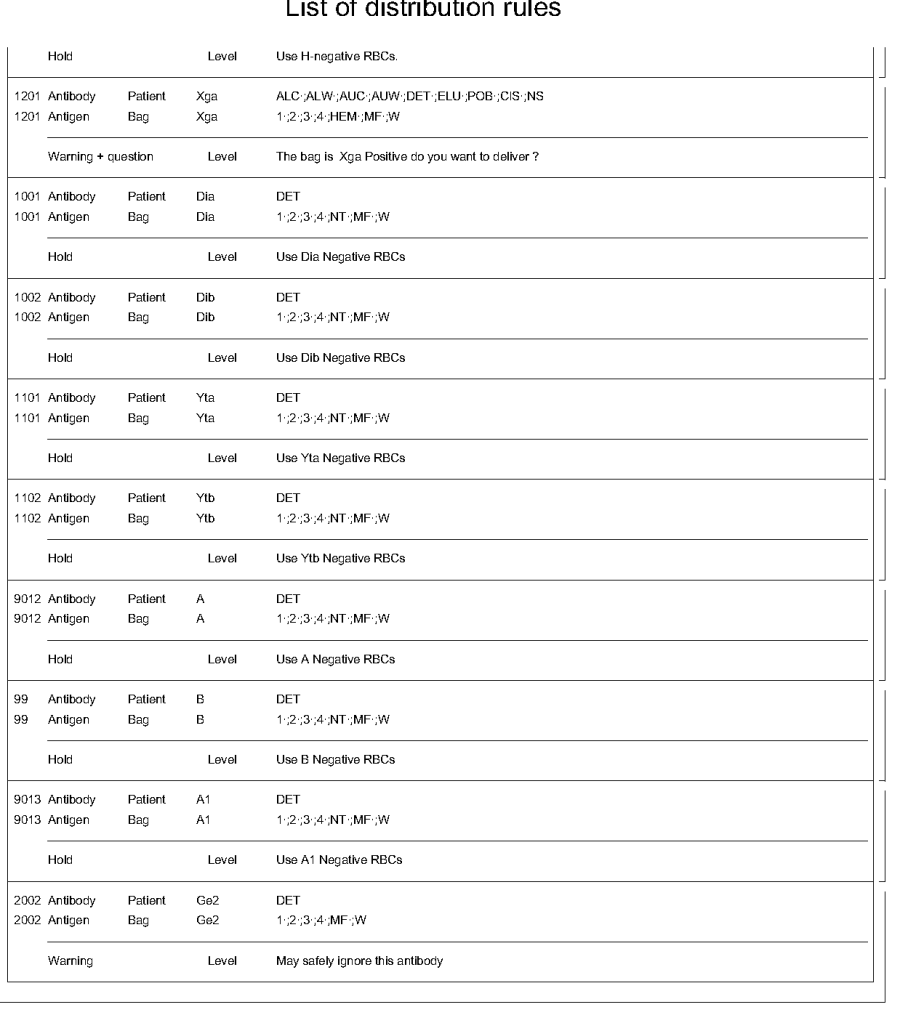
For donor ABO typing, I always had both automated and manual methods set up on the blood bank computer system Medinfo Hematos IIG. The automated method had a bidirectional interface between Medinfo and the instrument. Medinfo did not need a separate middleware. A truth table was prepared for acceptable results for automatic interpretation. Other results had to be manually interpreted by someone with the appropriate security level.
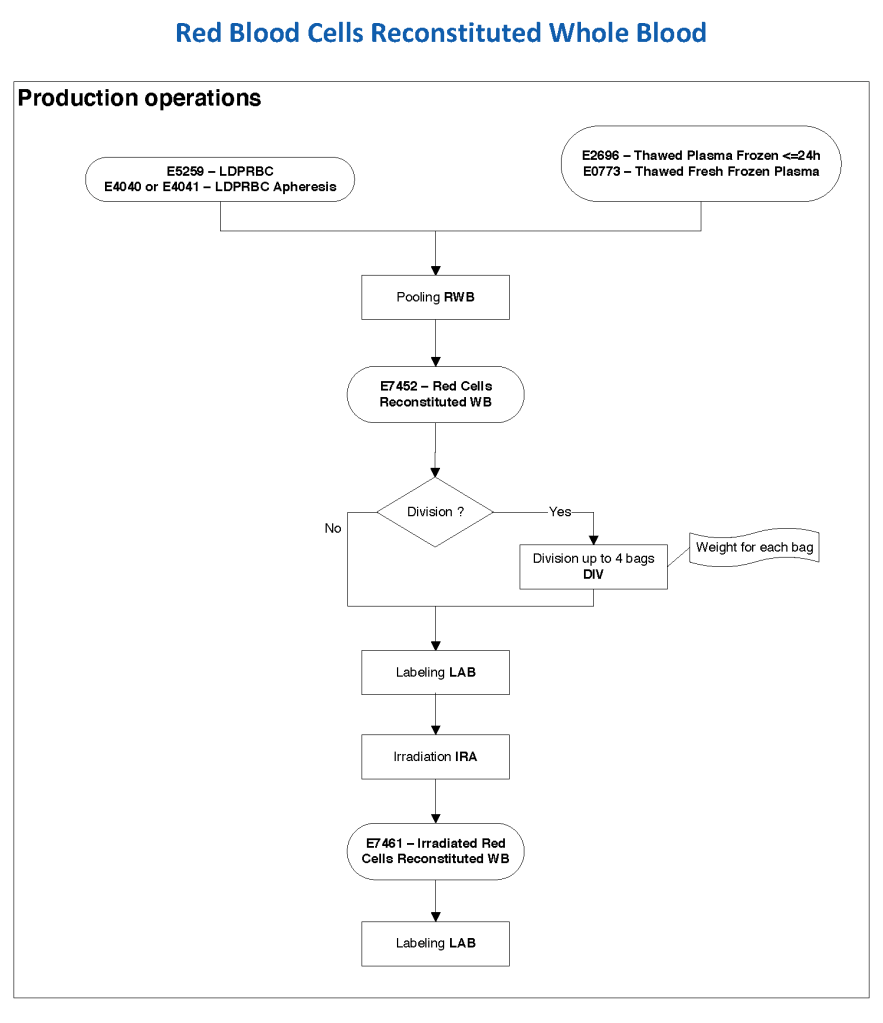
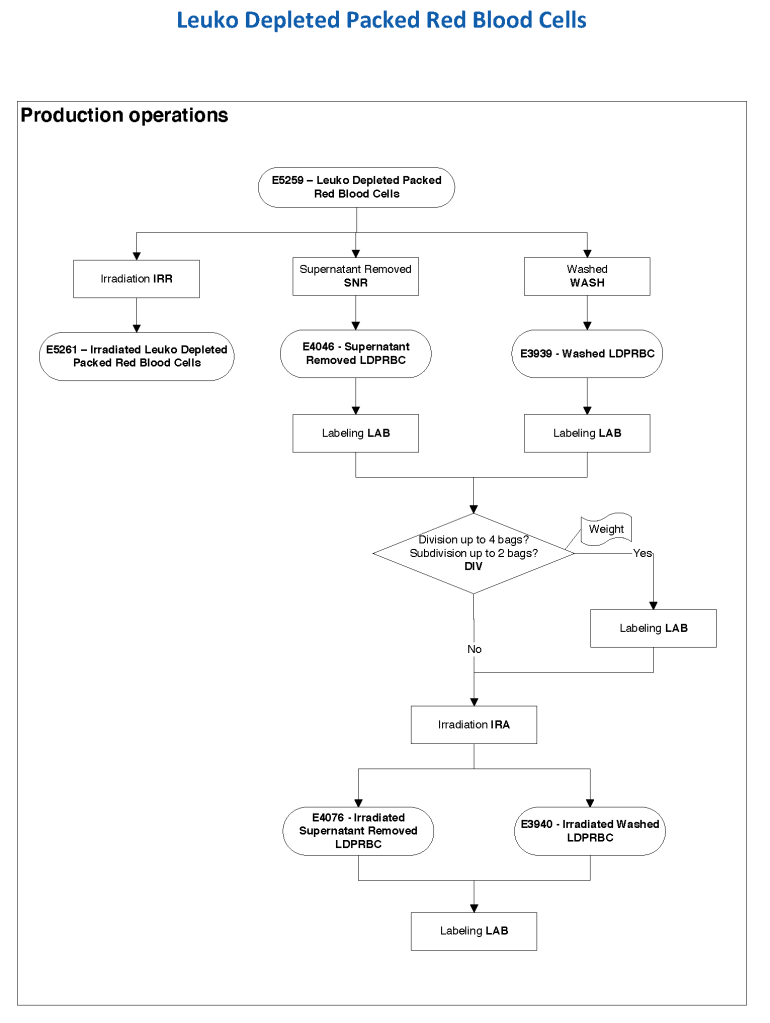
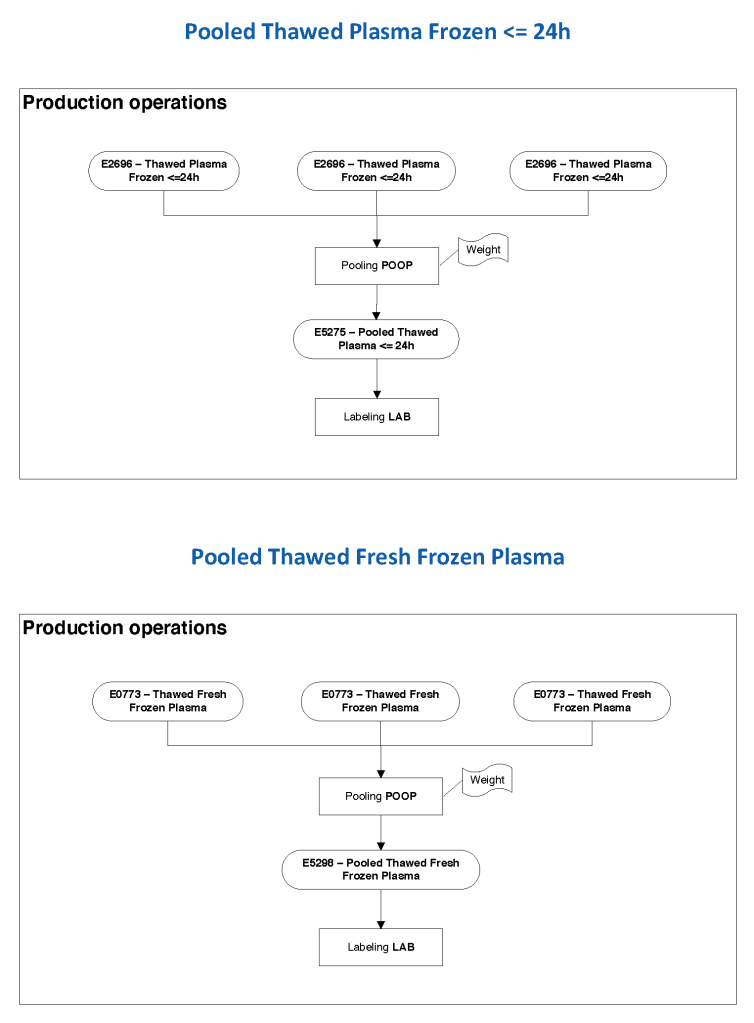
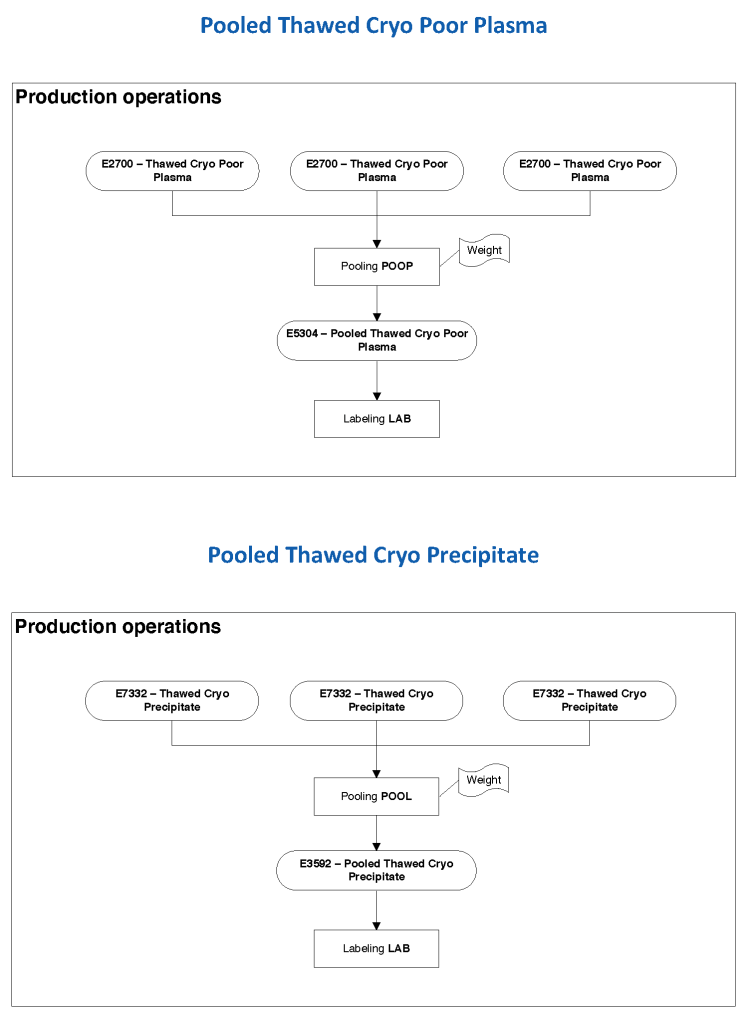
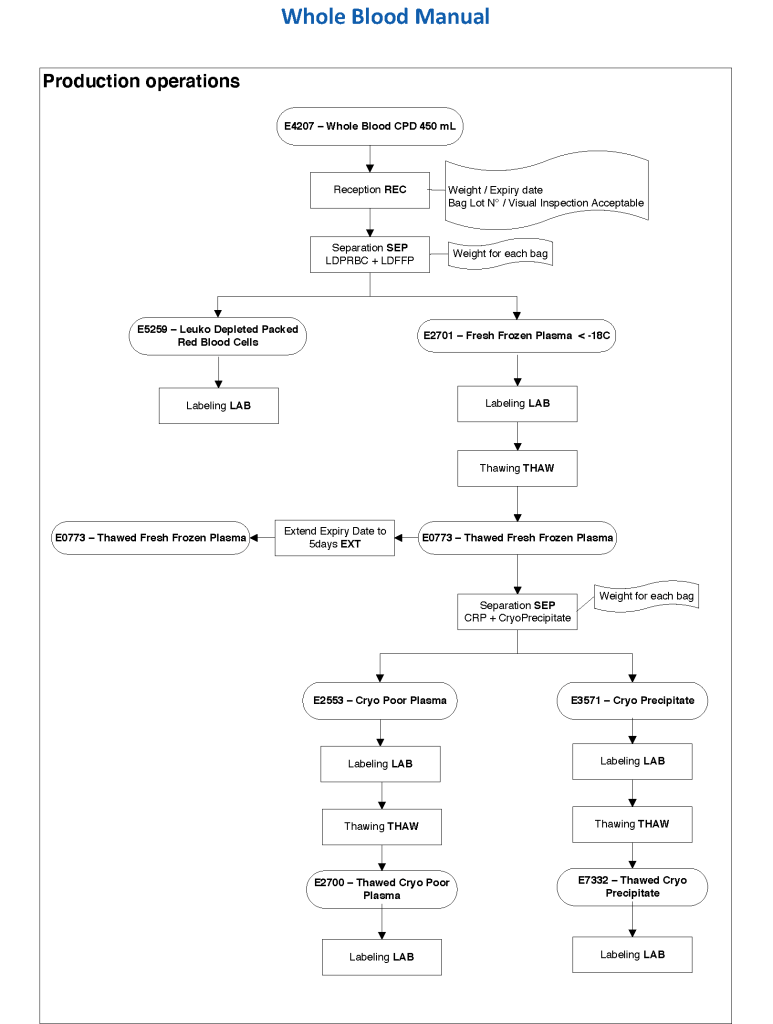
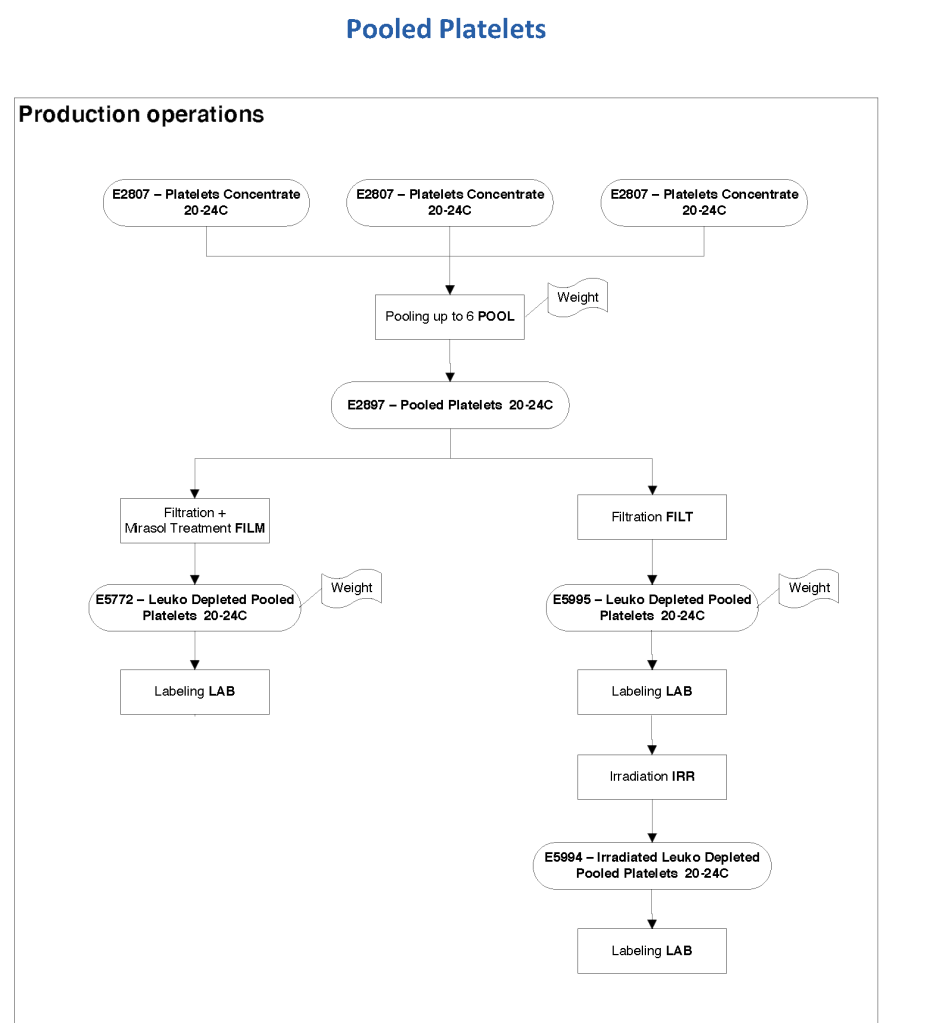
The manufacturer’s recommendations for the particular reagents in use were strictly followed. One used the range {0, 1, 2, 3, 4} as acceptable. Another used {0, 2, 3, 4}. Controls were included. Refer to the following flow diagram.
Most importantly, Medinfo could be configured for any set of reagent values.
2/10/20