


Donor, patient, interdepot transfer, interfaces


Note: This is an update of a previous post.
In the previous posts I outlined how Medinfo handled antibody screening and identification. This post reviews how antigen matching is used based on these results.
There are two modes, regular and emergency. If the patient has not had at least two ABO/D determinations and/or does not have a recent antibody screen within the past 3 days, then emergency mode must be selected with its own rules. Otherwise, the regular mode applies.
Regular Mode:
In general, if there is a clinically significant antibody, an RBC unit which has not been matched for the corresponding antigen or has the corresponding antigen cannot be routinely selected. However there is a hierarchy here also:
Examples of Regular Mode rules follow (these are not the complete lists but just provided to show the complexity of the process).
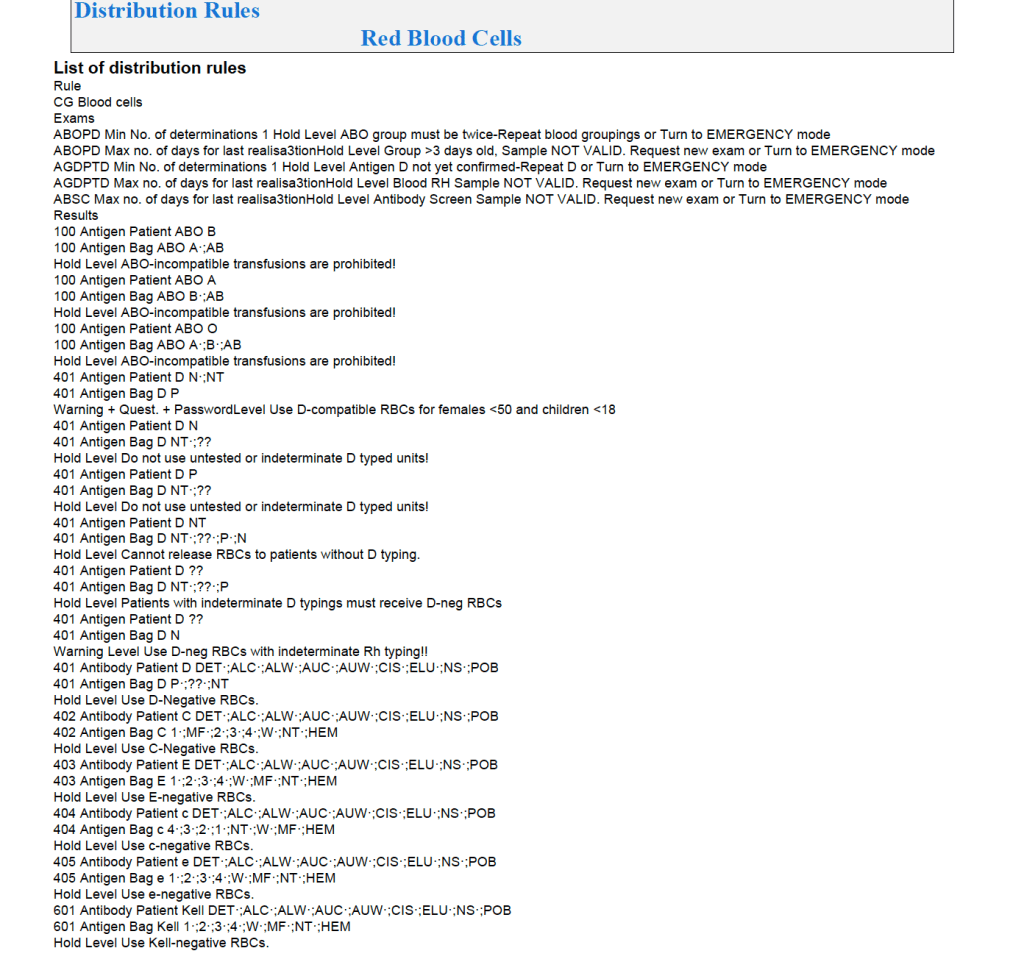

Emergency Mode:
This is much more restricted for selection of ABO/D and other antigen typings. An example follows:
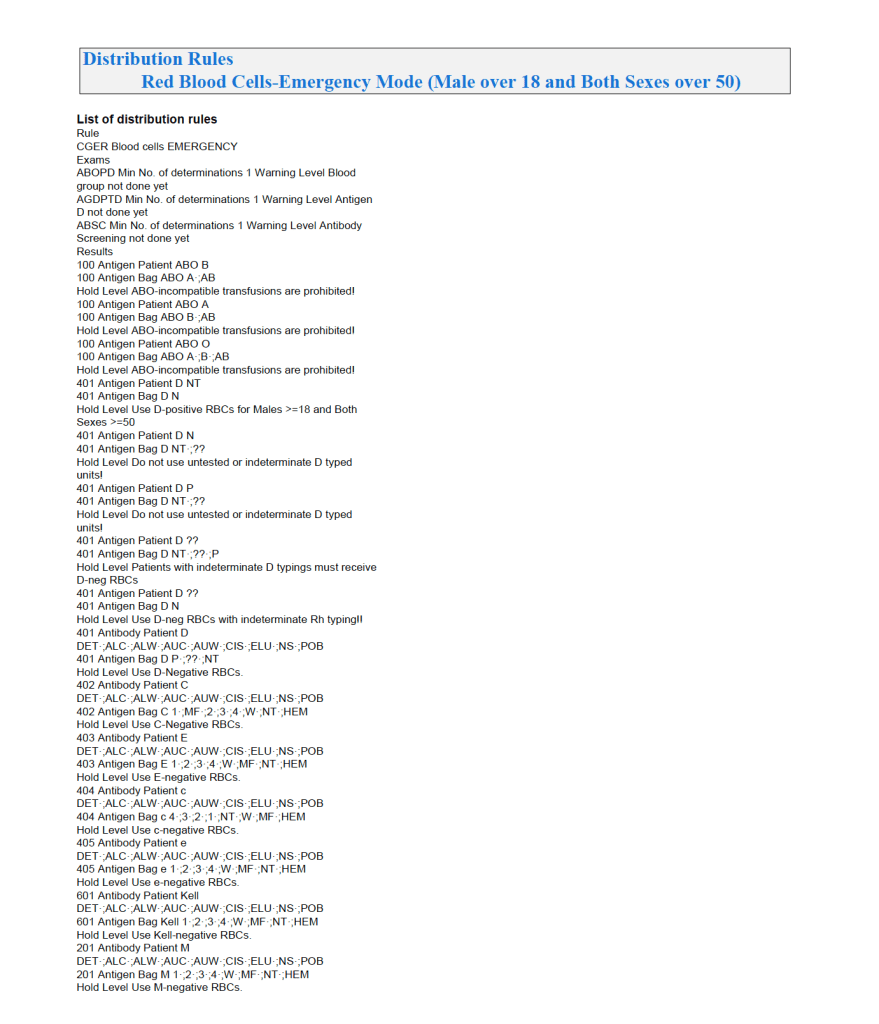
If low-titer group O whole blood is available, then a specific rule must be added to both regular and emergency mode to allow this to be given to any ABO type except Bombay.
This is an update of a previous post.
The final components from the component preparation center may be sent to various depots (freestanding location and/or hospital blood banks. There should be complete traceability for every step (from donor reception, collection, testing, and processing) transport between locations, and finally the exact storage site, which might include which refrigerator/freezer/incubator and even shelf/position number for each component is stored. The end of that document showed rules for type/antigen matching.
For disaster planning, rapid inventory enumeration by type is very important. This can be very time-consuming manually. With our Medinfo Hematos blood bank system, we could quickly get total inventory across the Qatar or by hospital in less than one minute. We could also quickly find antigen-matched units across the system and reserve it at any one site for another if necessary.
Smart blood bank dispensing refrigerators, as offered by Haemonetics and Angelatoni, may also serve as depots and take the place of a hospital blood bank for some dispensing. These solutions can also capture vital information about the storage conditions of the components and prevent release if the storage criteria are not met. They can also interface with blood bank computer systems and use the main system’s logic for the dispensation rules.
Upon receipt at the hospitals from the blood processing center, the forward ABO and D typing must be confirmed. We used D reagents which detected partial D so we would call such donor units as D-positive. However, if a patient type reagent insensitive to partial D types is used, it is possible for a unit to be typed as D-negative whereas in the donor center it might be D-positive. Sometimes, nothing types consistently as D-positive: all you can say is that with a particular reagent and lot number, there is or isn’t reactivity.
The greatest complexity is for RBCs since potentially so many antigens exist. Criteria for matching/ignoring certain antigens must be made. Critically significant antibodies such as the Kell, Duffy, Kidd, and certain Rh (D and c) must be antigen matched. A robust blood bank computer system can enforce these rules.
For other components, antigen/typing may be less important. In fact, in most situations, any type of platelets can be given to anyone (except neonates). Despite the potentially incompatible plasma, there is rarely significant hemolysis. In fact, if pooling platelets without regard to blood types is done, a platelet transfusion is a common cause of a positive direct antiglobulin test DAT—something that is not clinically significant. No one died of a positive DAT by itself for this reason.
Specific rules for compatible plasma types are important, but nowadays, low-anti-B-titer group A plasma may be used like universal AB plasma. The challenge is to be able to perform the ABO titration (specifically anti-B) quickly—titration can be a slow process, even with automated equipment. A similar situation for low-titer, universal group O whole blood requires both anti-A and anti-B titration (I will return to this topic in a future post).
Process: Donor Collection
Zeyd Merenkov, MD, FCAP, FASCP
Independent Consultant in Transfusion Medicine
5.4.1 PROCESS DONOR COLLECTION:
Process:
References:
5.4 POLICY: DONOR COLLECTION
Policy:
References:
This is the start of a series of posts on how Medinfo blood bank software was designed for product delivery in the HMC system in Doha.
The overall process was:
It was also possible to release blood components directly from the Blood Donor Center to HMC hospital blood banks as a contingency. Client hospitals outside the HMC system still had to obtain their components from HGH General Delivery Deposit.
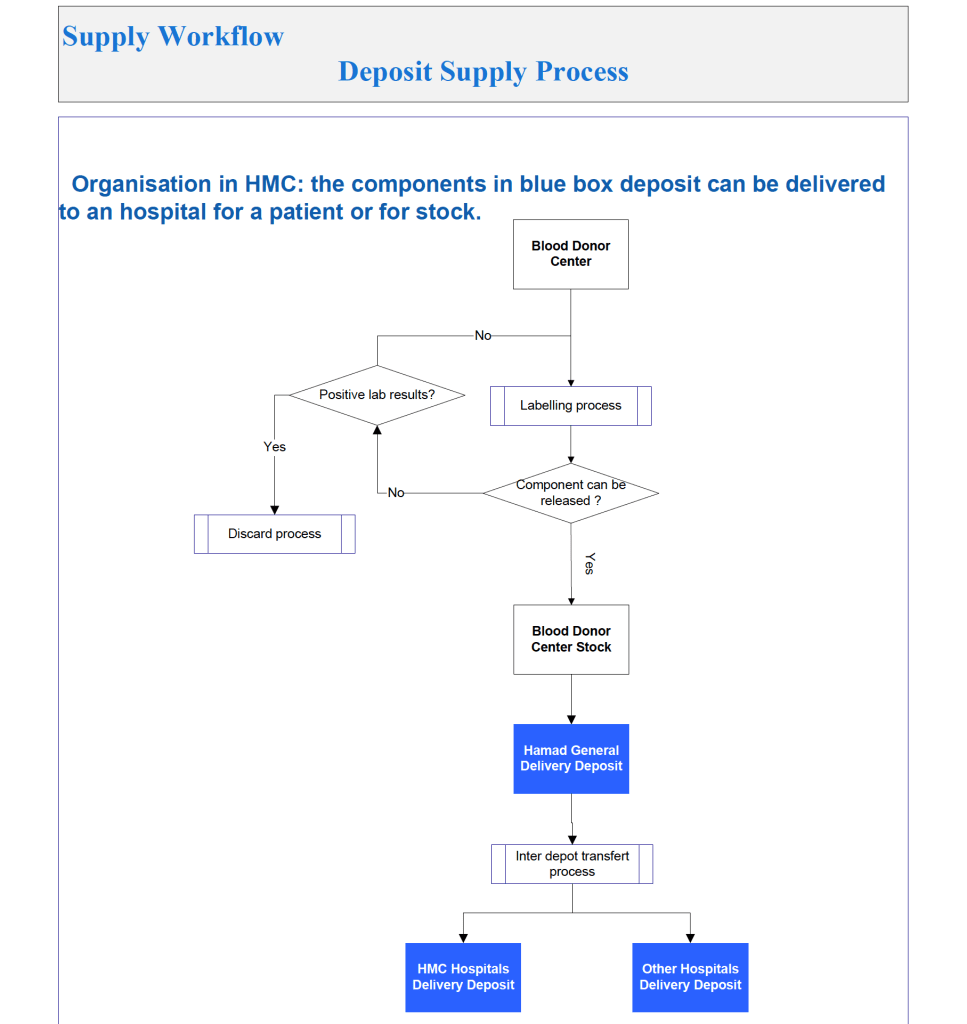
Processes and Software Building Part 7
This is an update of a previous post.
Blood Bank instruments may perform tests and release test results in a numerical or alphanumeric format or both. For example, nucleic acid and enzyme immunoassay may release a qualitative result (e.g. positive, reactive, borderline/gray-zone, negative, nonreactive). Alternatively, the machine may release the signal to cutoff ratio (S/CO) as a numeric result.
Blood bank software may use either kind of result on which to base interpretative rules for acceptability of the donor. The qualitative result criteria are based on the quantitative SC/O but the equipment automatically interprets this. The S/CO ratio of 1 is the cut-off point. Thus a value of 0.99 is negative and the value 1.01 is positive. But is it really so clear-cut since the difference between the two is so small? Thus, some people have added the term gray-zone for values close to but below the cutoff. Could a value of 0.95 be an early infection?
I personally prefer to see the actual cutoff but use the manufacturer’s criteria for interpretation. As a physician, it is good to review the S/CO on serial exams. If a borderline or gray-zone result becomes positive, then perhaps the original result indicated early infection. The question still remains, what is the gray-zone? 0.95 to 0.99, 0.90 to 0.99, etc. Some accrediting schema have not used gray-zone for interpretation.
With Medinfo’s blood bank software, I could choose either option or both—or at least store the S/CO as a nonreported result for subsequent review. I could even chose, test by test, in a series between reporting either S/CO or the qualitative result.
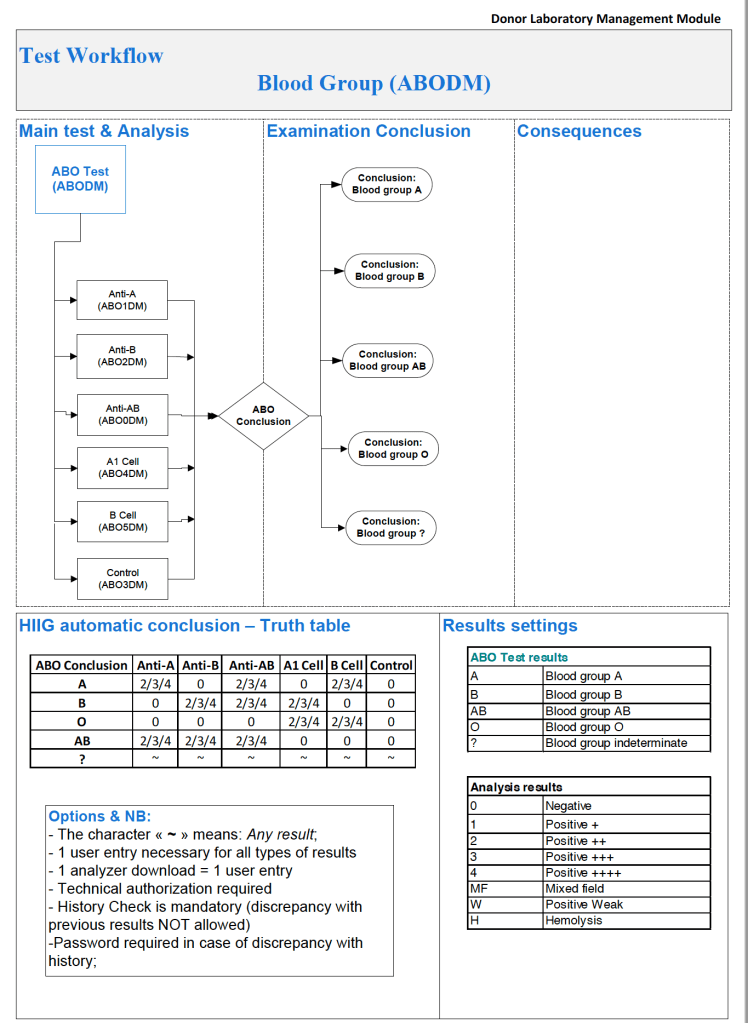
Semiquantitative results, e.g. in {0, 1+, 2+, 3+, 4+} are qualitative and could also include mixed field (mf) and hemolyzed (h). I showed examples of this with ABO/D antigen typing in a previous post—see attachment.
On the contrary, the results from blood production equipment may include parameters such as time of preparation, original volume, final volumes for each component, platelet yield index as an indirect measure of platelet count. When there is pooling, the final total volume is critical to determine if pathogen-inactivation procedures and platelet additive solution can be used. This is a much more complicated interface.
This is a sample process document for donor physical examination. Compare it to the previous post Policy: Donor Physical Examination.
5.3.1 PROCESS DONOR PHYSICAL EXAMINATION:
Process:
References:
This is another example of a short concise policy statement. The process example will follow:
5.3 POLICY: DONOR PHYSICAL EXAM
Policy:
References:
This is an updated version of a previous post.
Interfaces—General Considerations:
When buying equipment while planning/implementing new laboratory software, I originally had a rule not to purchase anything that the vendor did not have a ready interface. Even that was not so clear since some vendors had interfaces listed as alpha, beta, and completed.
Could you use an alpha or beta interface? Was it safe for patient care? What was the development cycle for new interfaces with your vendor—months, years?
Even if the vendor had a completed interface? How “complete” was it? Did it accept all data from the machine? Did the data stream require reformatting? Who would write the transformational script?
Even if the vendor could support it, could your local IT organization and the local agent’s IT staff do it? I had plenty of headaches over this. The best equipment with the best interface that the local agent could not support was worthless to me.
Some finished interfaces took months to install because of connectivity issues. What version of the operating system OS was used? Was it secure? Did our IT department accept that OS version (e.g. Windows 7) and the provided malware protection? I have seen malware spread across a network from the interface software installed by the vendor, threatening the entire corporate system.
Did the solution require middleware? What were the implications of having middleware and its affect on the main software program, especially at the time of its upgrade or the main software?
I have seen vendors using Windows 2000 for their interface software as late as 2017. It was difficult for some of them to update to current, more secure versions. Anyway, our corporate IT department gave them all a deadline to update to the current operating system—they all complied or risked losing all connectivity to the network.
Almost every instrument vendor has told me that they can communicate with my laboratory system. I guess that is true: one talks in Russian and the other Sanskrit—they do communicate but is it effective? Talking is not necessarily useful communication!
I remember one open EIA machine that had a TCP/IP port but it was not functional by the standard protocols. One had to emulate a serial port to get some rudimentary communication. The port’s light blinked, however. I never imagined that someone would put a nonfunctional port as a mere decoration.
On the other hand, I have had excellent experience with another software vendor Medinfo. Even if the vendor did not have the interface developed, they could build it from scratch in a few weeks. Paradoxically, it was faster to build these new interfaces than some so-called already interfaces.
I must emphasize: This is a collaborative team effort between the blood bank information system, software vendor, instrument vendor, and your institution’s IT staff. There must be excellent cooperation between them for a successful result.
When installing the Medinfo Hematos IIG software, many of our most important interfaces (the Terumo mixed shaker, Trima, Reveos and its predecessor Atreus, Mirasol illuminator) had no developed interfaces when we started. This was a risk, but actually those interfaces were developed in a few weeks and fully functional. In fact, we were the first site in the world to have those interfaces working—and without any Middleware.
In general, the blood bank software vendor installed the completed interface and did some low-level testing. Then, my blood bank computer team did the testing. The final responsibility for testing and acceptance was with the end-user blood bank team and me as Head of the Laboratory Information Systems.
In general, I wrote the validation protocol and assigned the tasks to the Medinfo Super Users. To perform this testing, we still had to register donors and collect and then export the specimens to the donor marker testing laboratory before the actual interface testing could begin. This was all done in a special test domain separate from the production domain. The interface did not go live until the validation was completed and accepted and then was moved to the production environment.
I made the validation criteria and reviewed all data as Division Head of Laboratory Information Systems. Representative screen shots were made. All data was sent to me. My final acceptance was required before the interface could be activated.
Automated component processing (Reveos) and component modification are more complicated and will be covered in a future post.