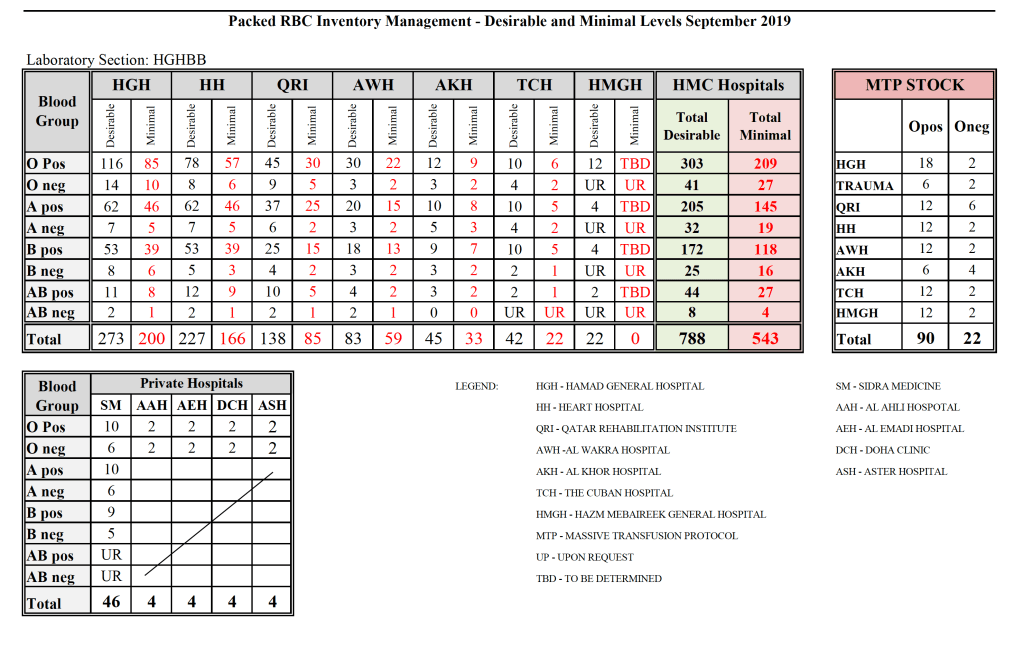
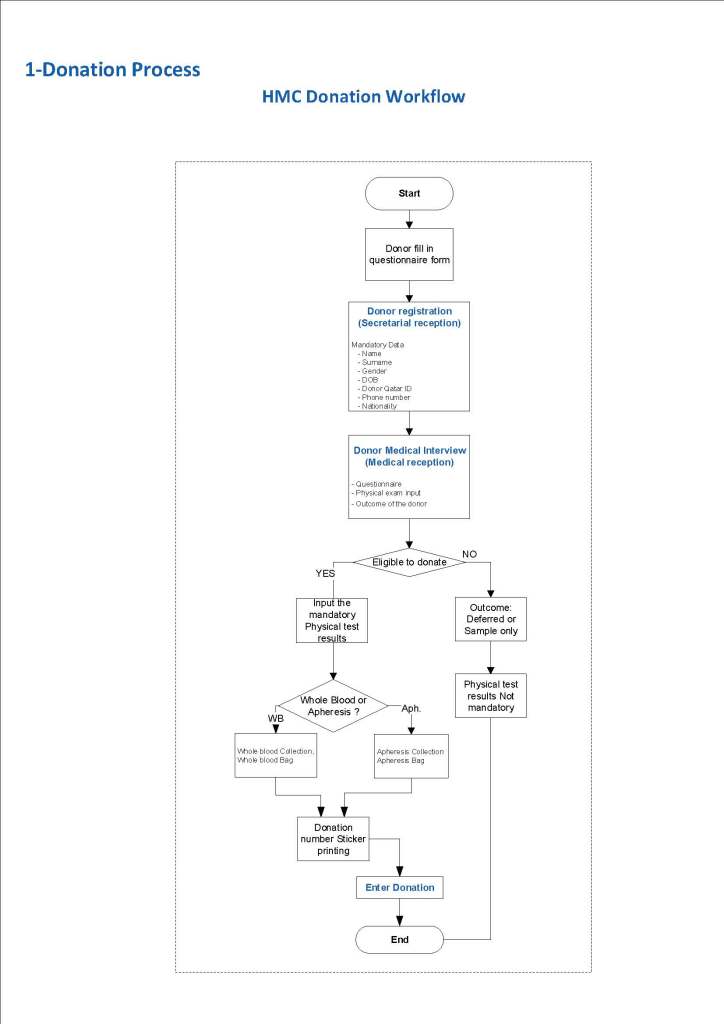
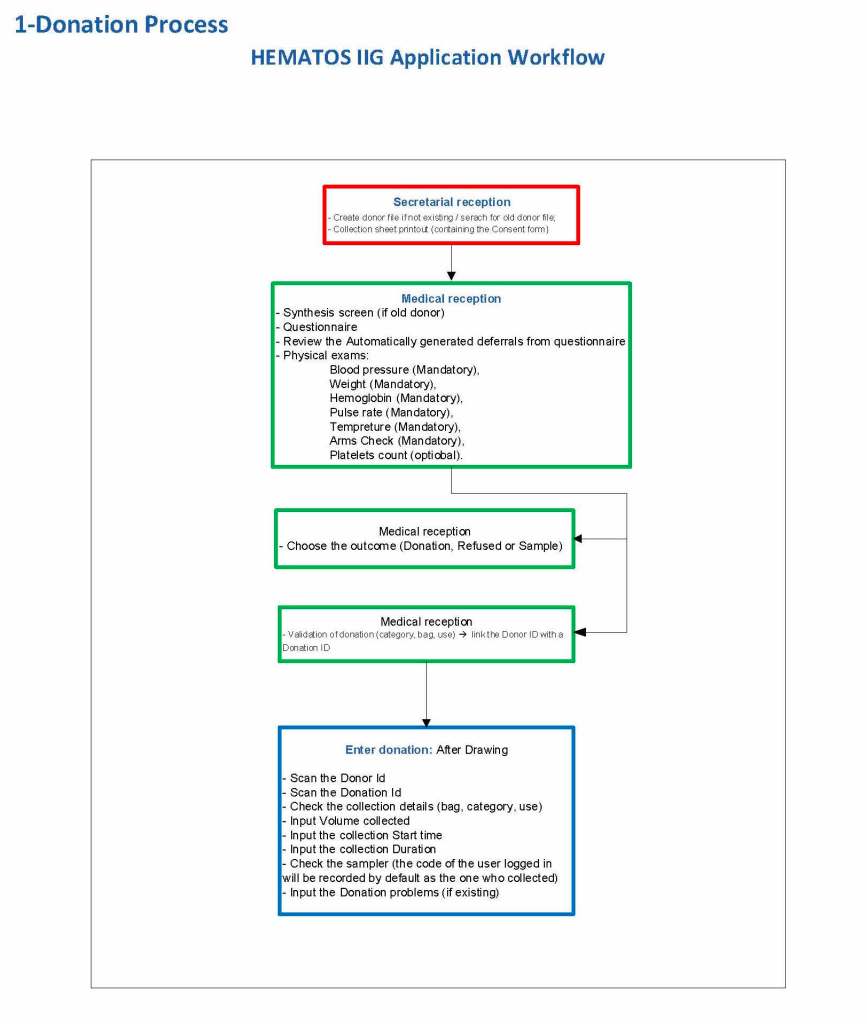
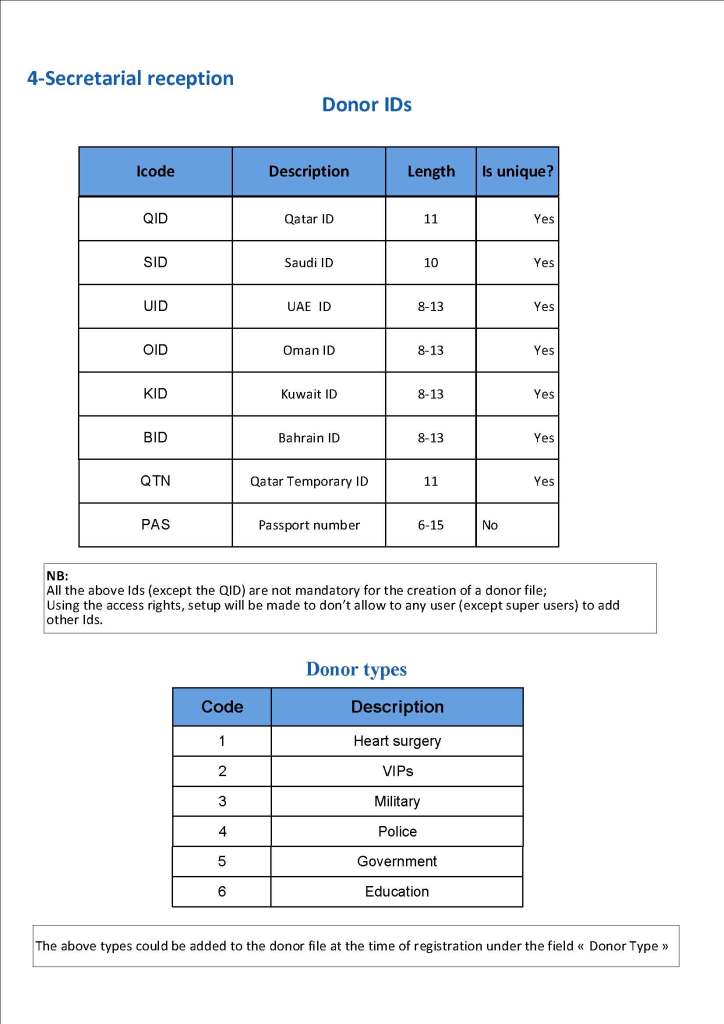
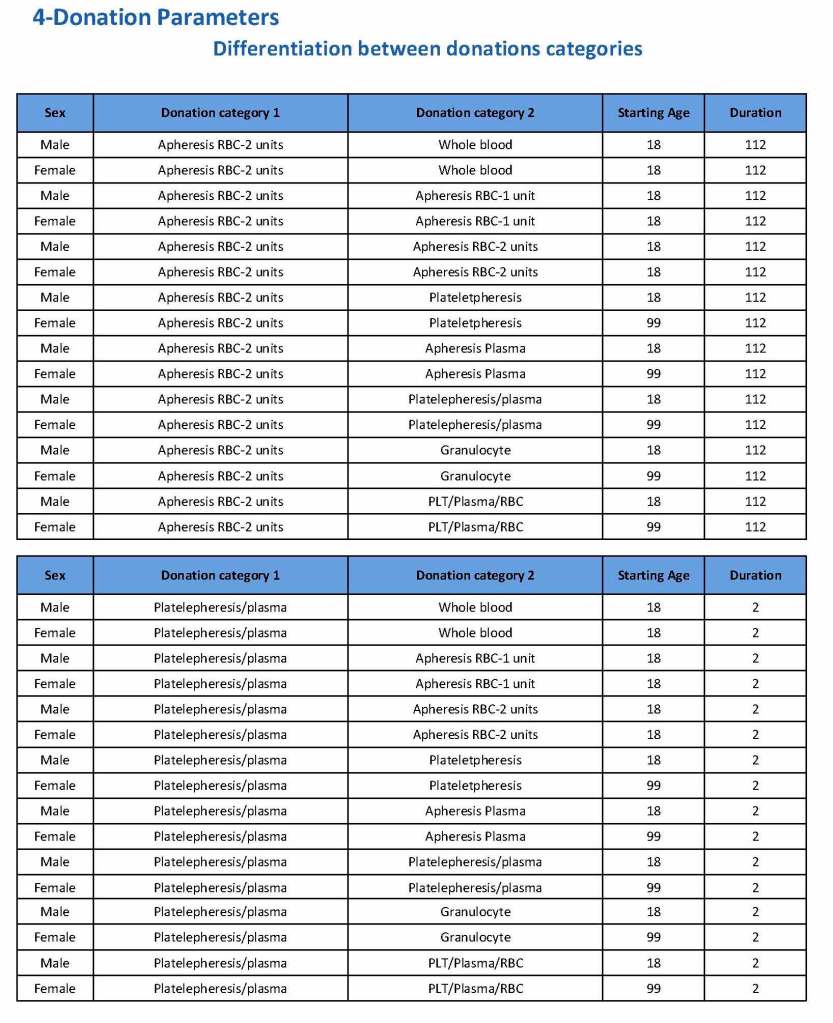
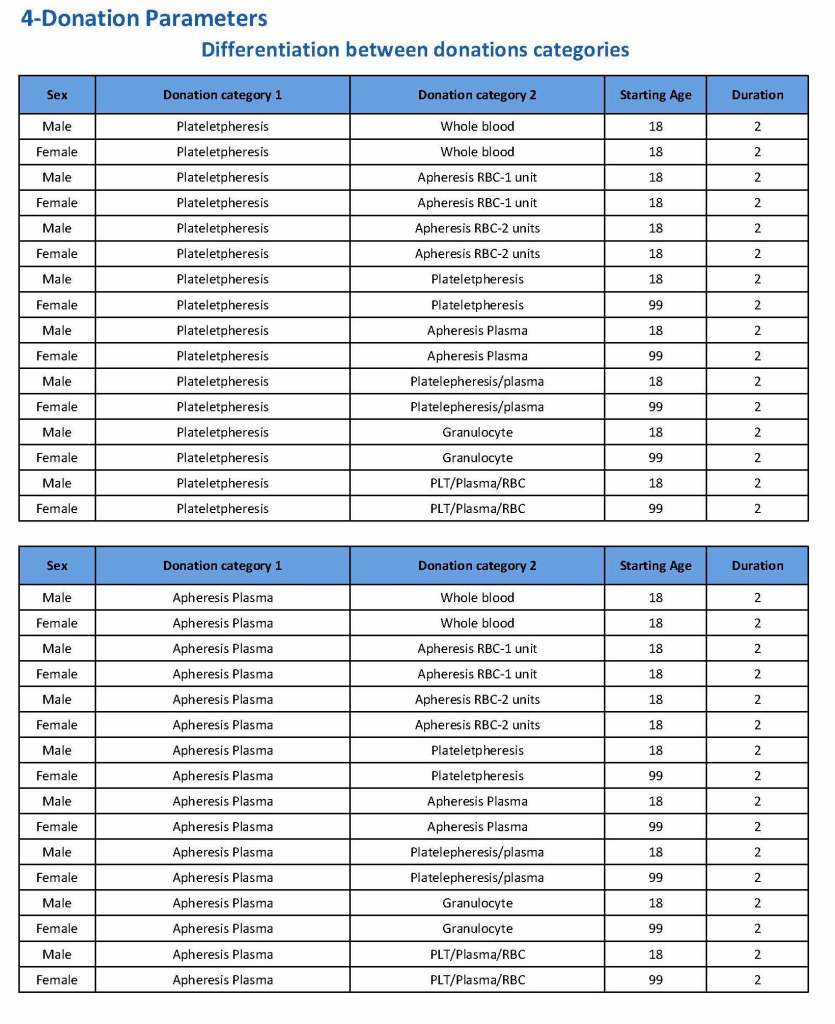
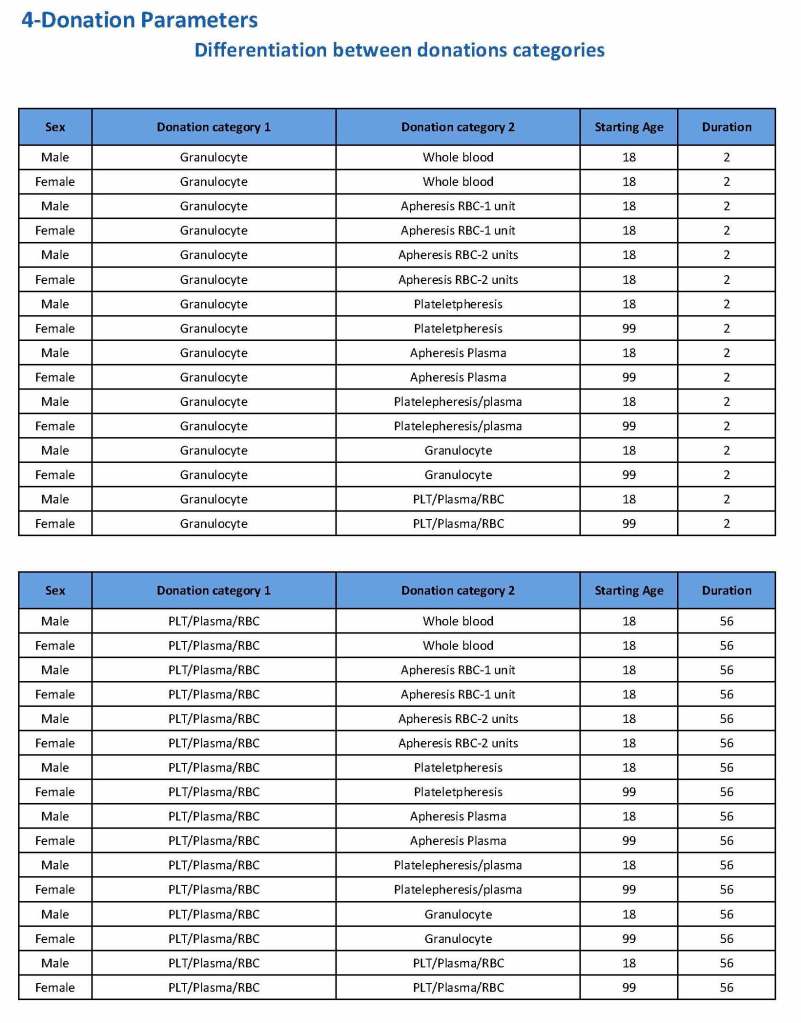
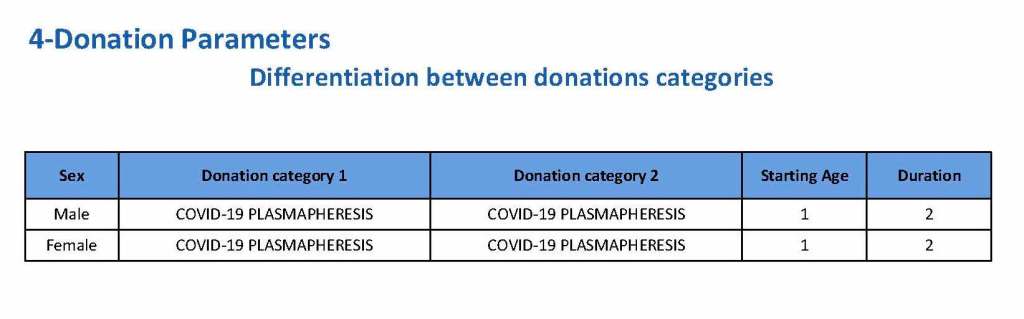
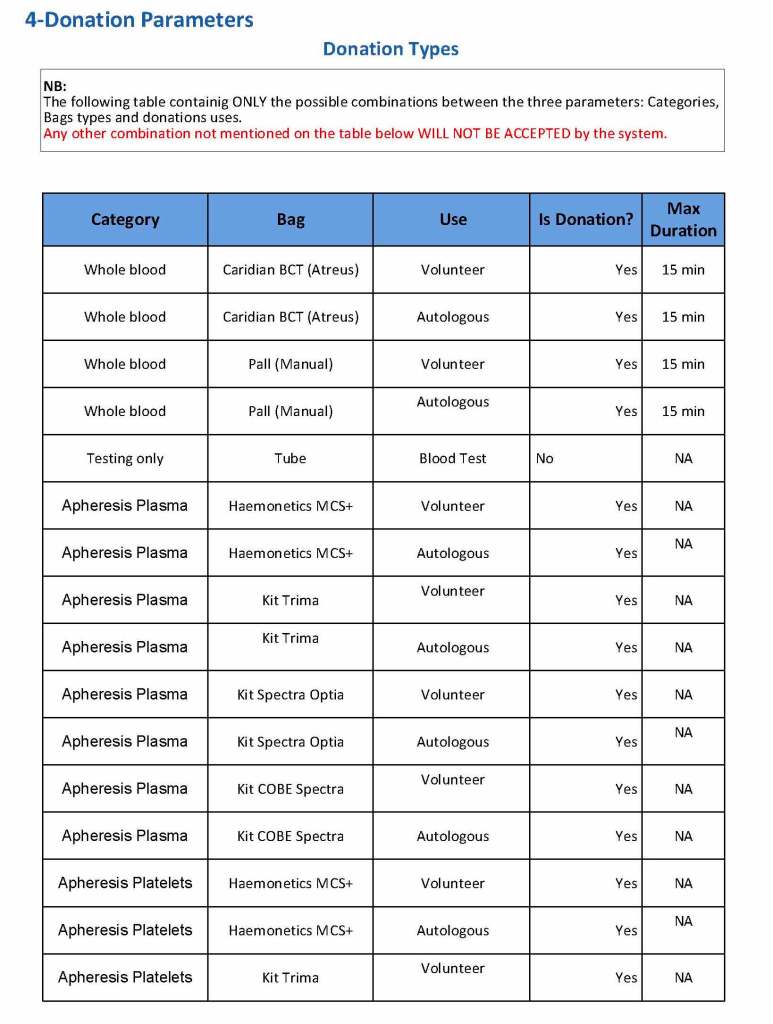
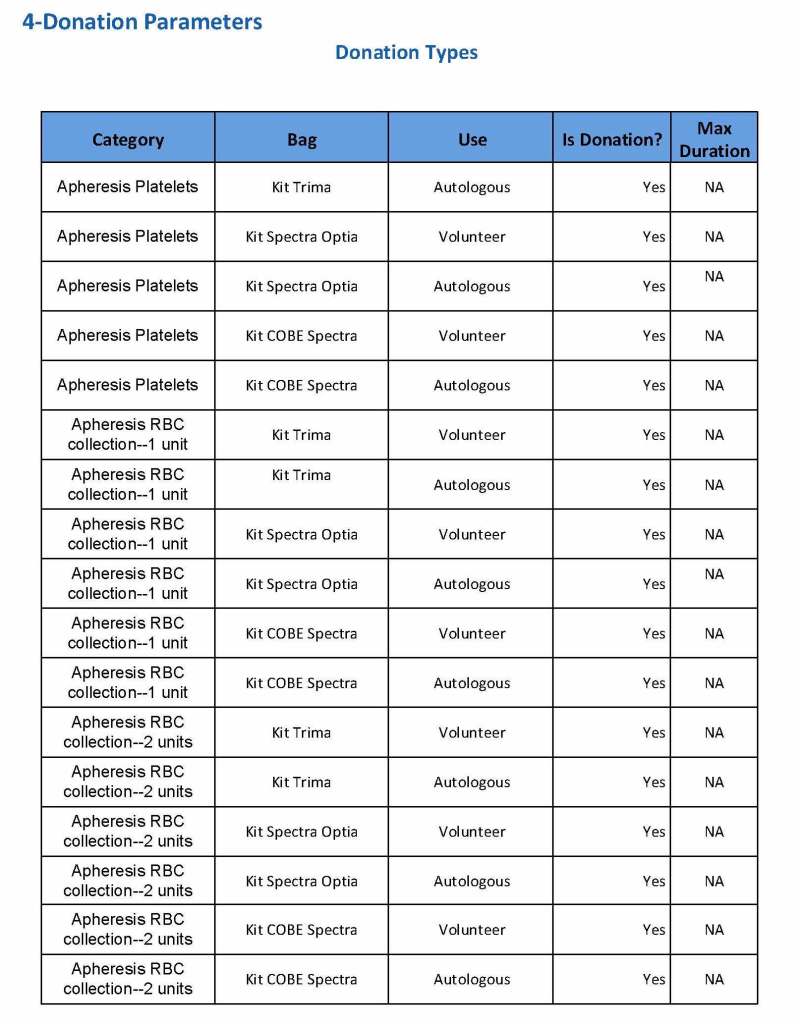
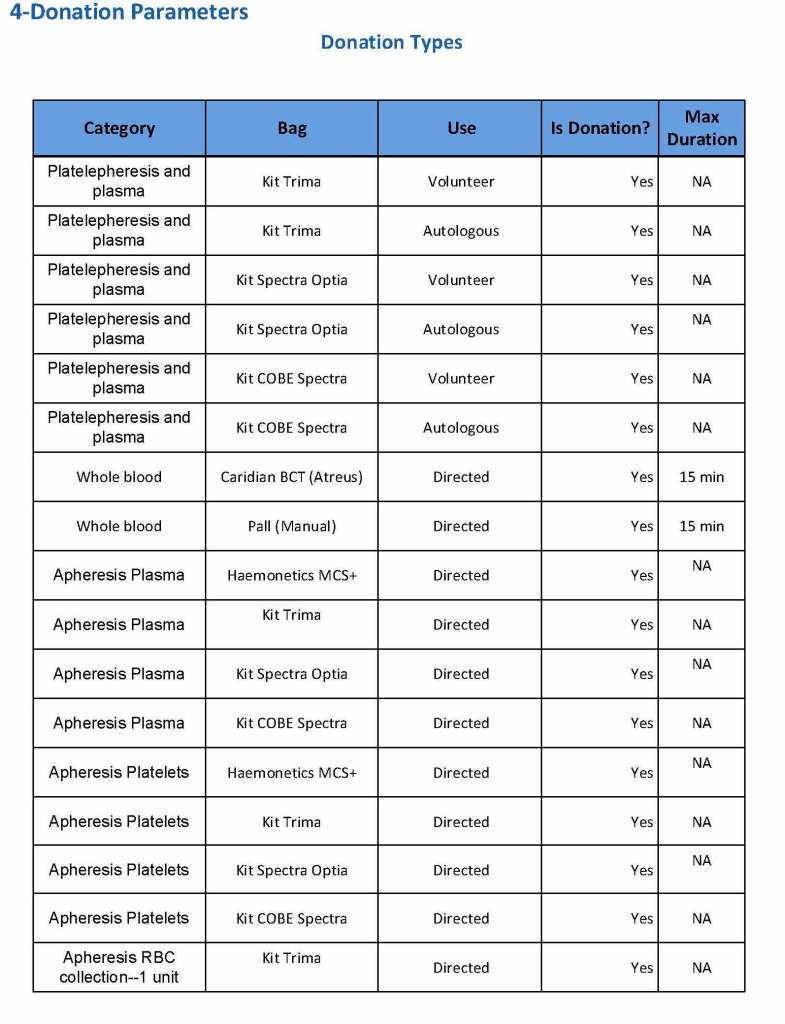
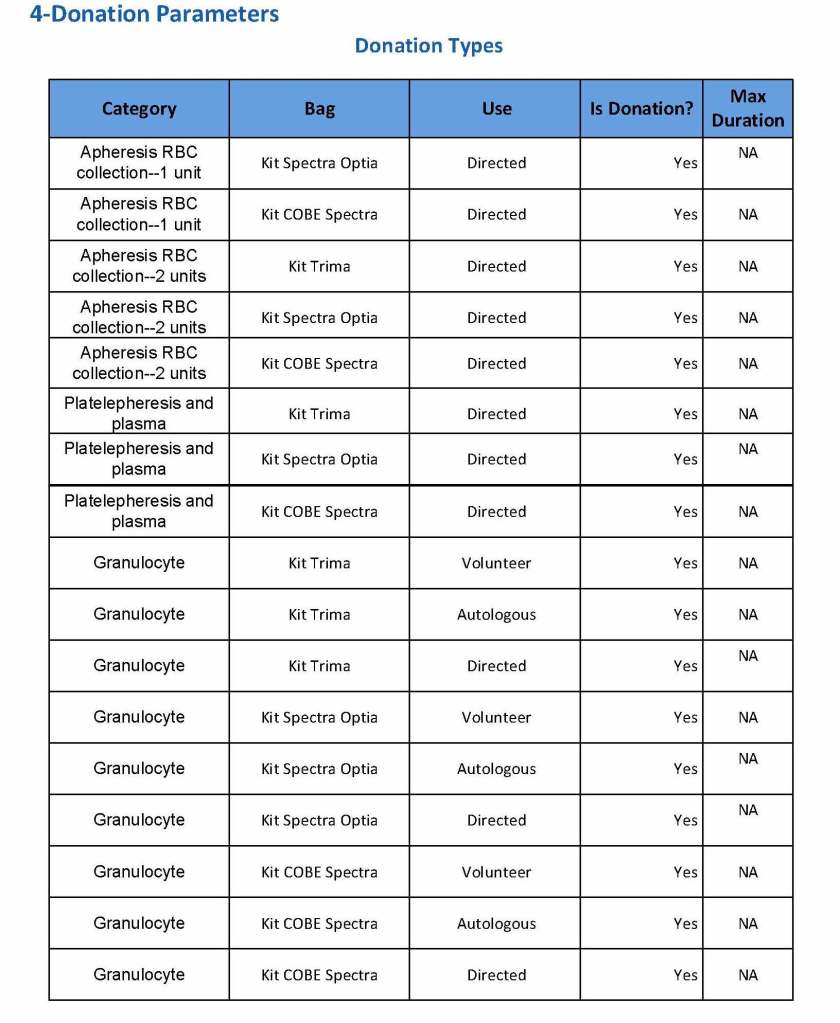
Yesterday’s post showed my active blood inventory management scheme for my previous position in Qatar. I thought today I would elaborate on how I adjust the inventory based on critical shortages and planning for disasters and other major events.
I always review the critical shortages to check for atypical usage (e.g. a disaster situation) or production issues (equipment breakdown, shortage of donors during holiday period).
If it is due to increased utilization, I try to adjust the critical and desirable inventories upward to cover the shortfall for future events. However, it is not always possible if the event is a one-of-a-kind situation unlikely to recur. Also, I must take into account the available resources (supplies, kits, manpower, equipment) to see if I can cope with the increase.
If it is due to resource issues, I see if I can bolster those by recommending increases or improving utilization of what is available.
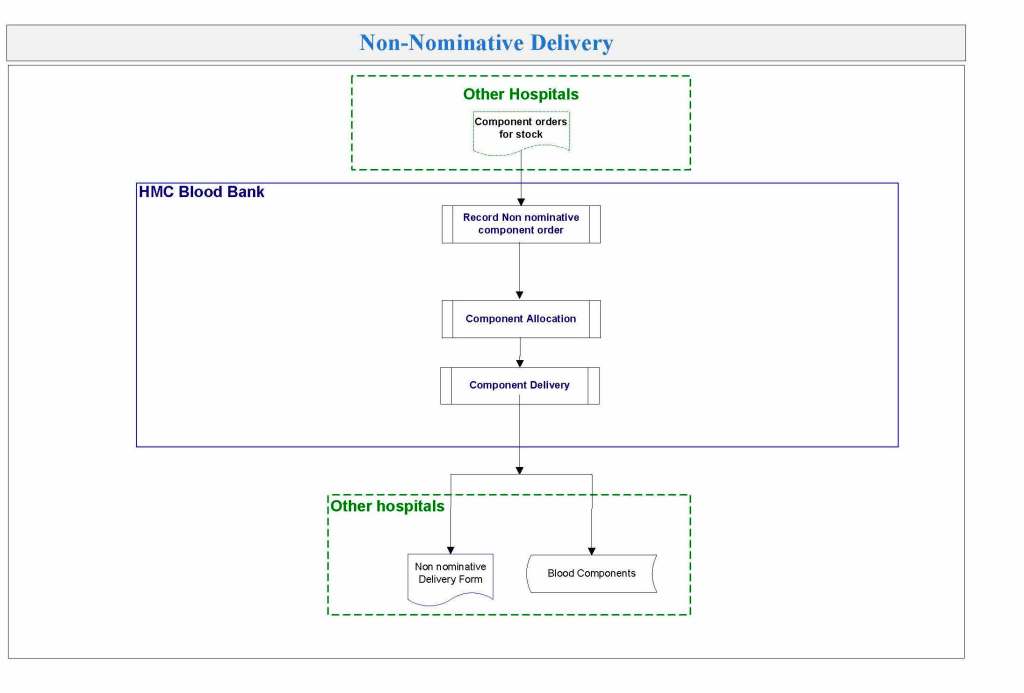
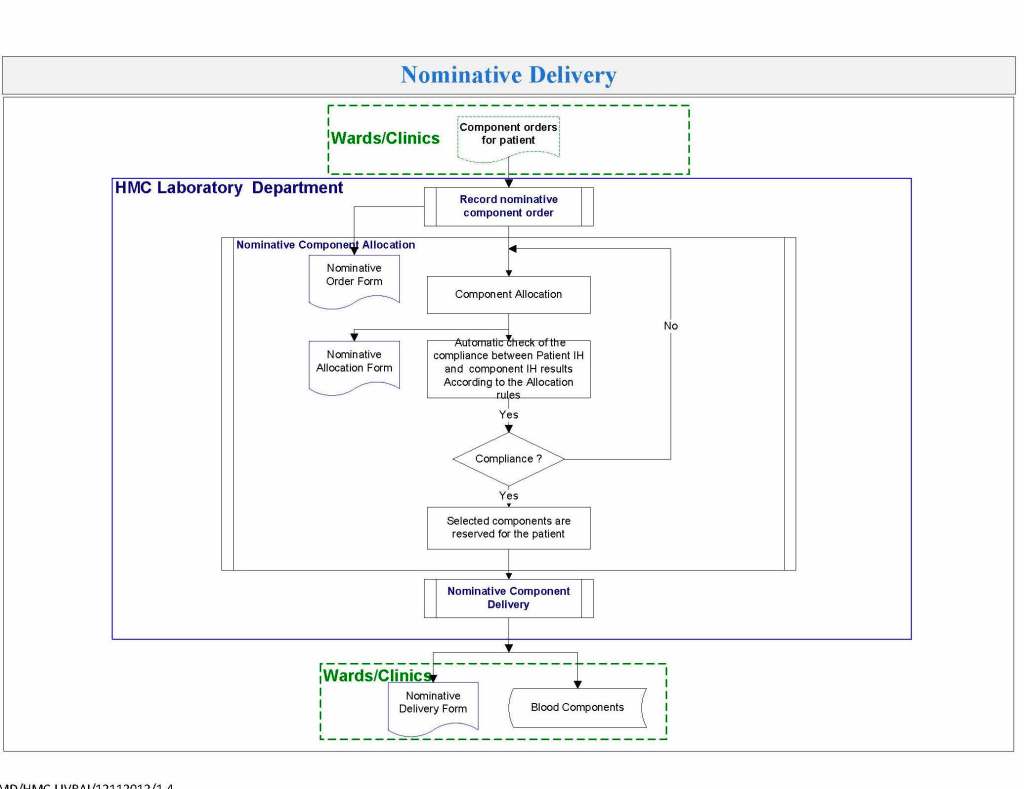
Very important is through-put: How quickly can I produce components from whole blood or apheresis components? This was one of the major reasons we shifted away from PCR to other NAT testing with single-well processes since to minimize the need to make additional runs (Grifols Panther System). Also, automated component processing can greatly speed production (one Reveos can process four whole blood units in about 23 minutes or about 12 units in 75 minutes.) Those staff can be busy with other tasks while the machines are working.
In the system I developed in Qatar, we could complete processing into components (RBCs, buffy coat platelet pools, leukodepleted plasma)–Reveos 3C Program, all marker and immunohematology testing, leukoreduction of the pools and RBCs, Mirasol pathogen inactivation, and platelet additive solution in as little as five hours!! There is great need for speed in a place that must be 100% self-sufficient in all blood components. We could even further reduce the total processing time if we only made RBCs and plasma, Reveos 2C Program
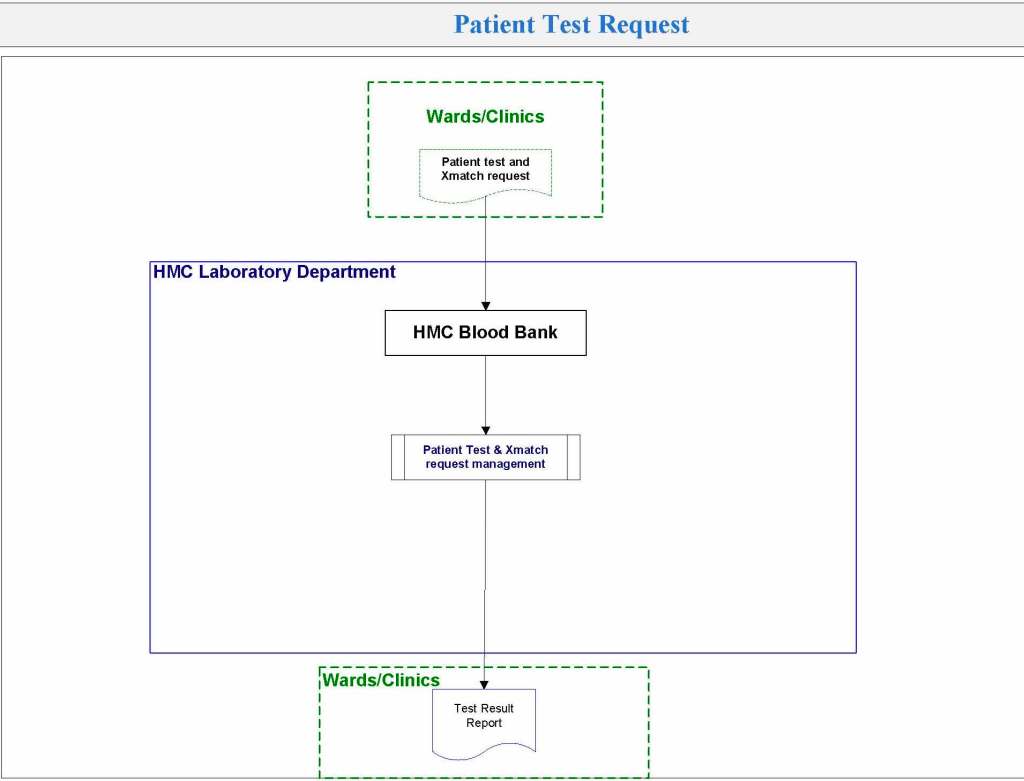
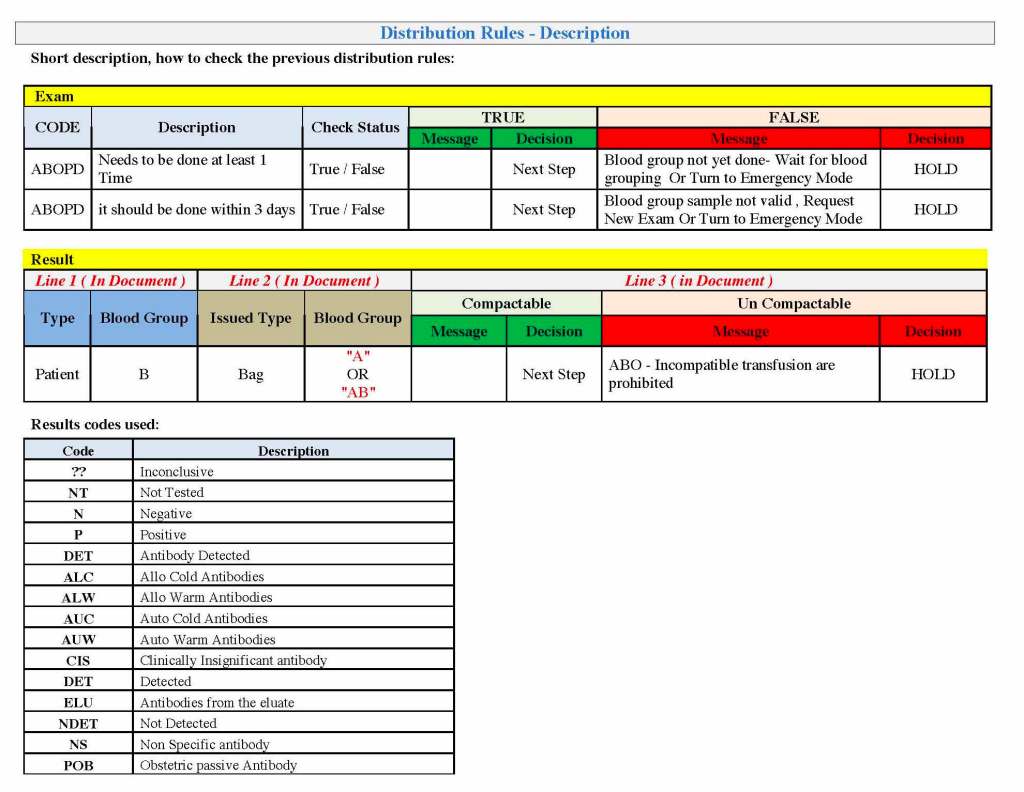
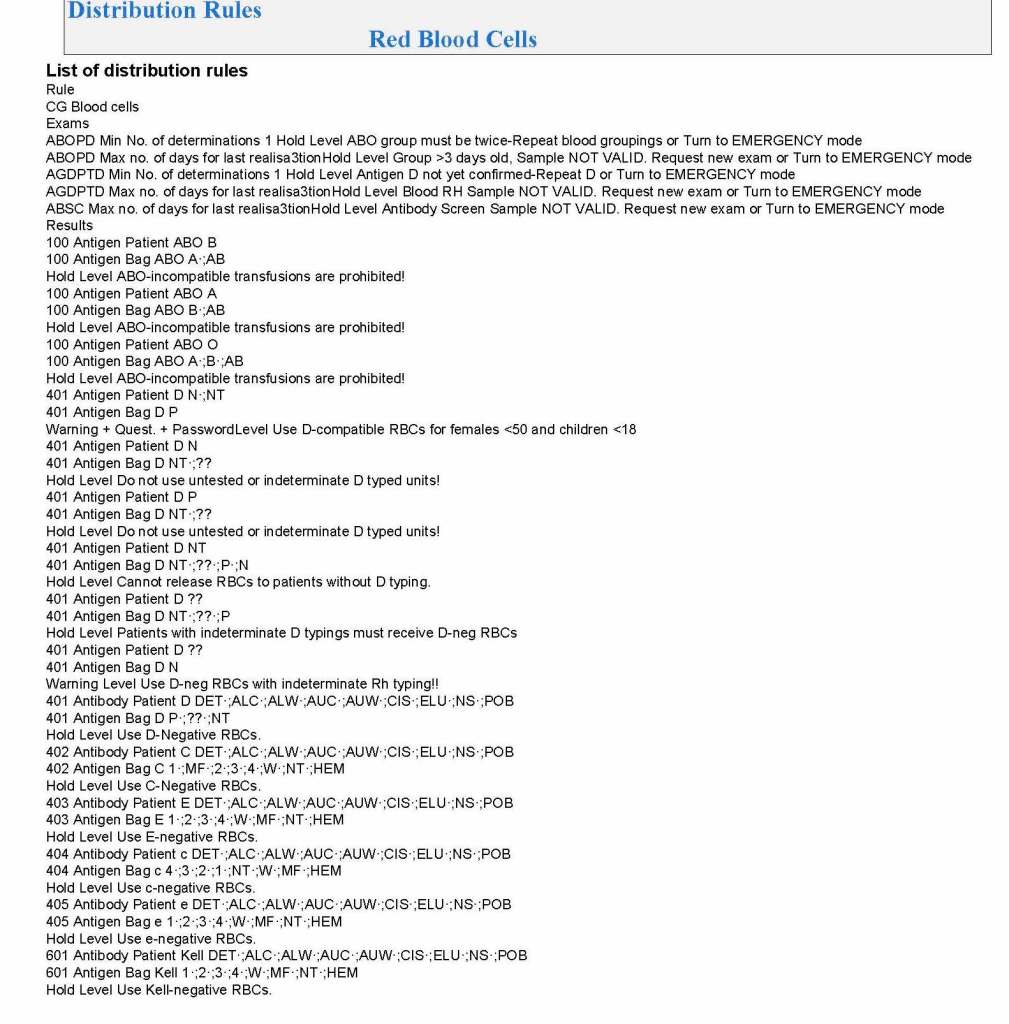
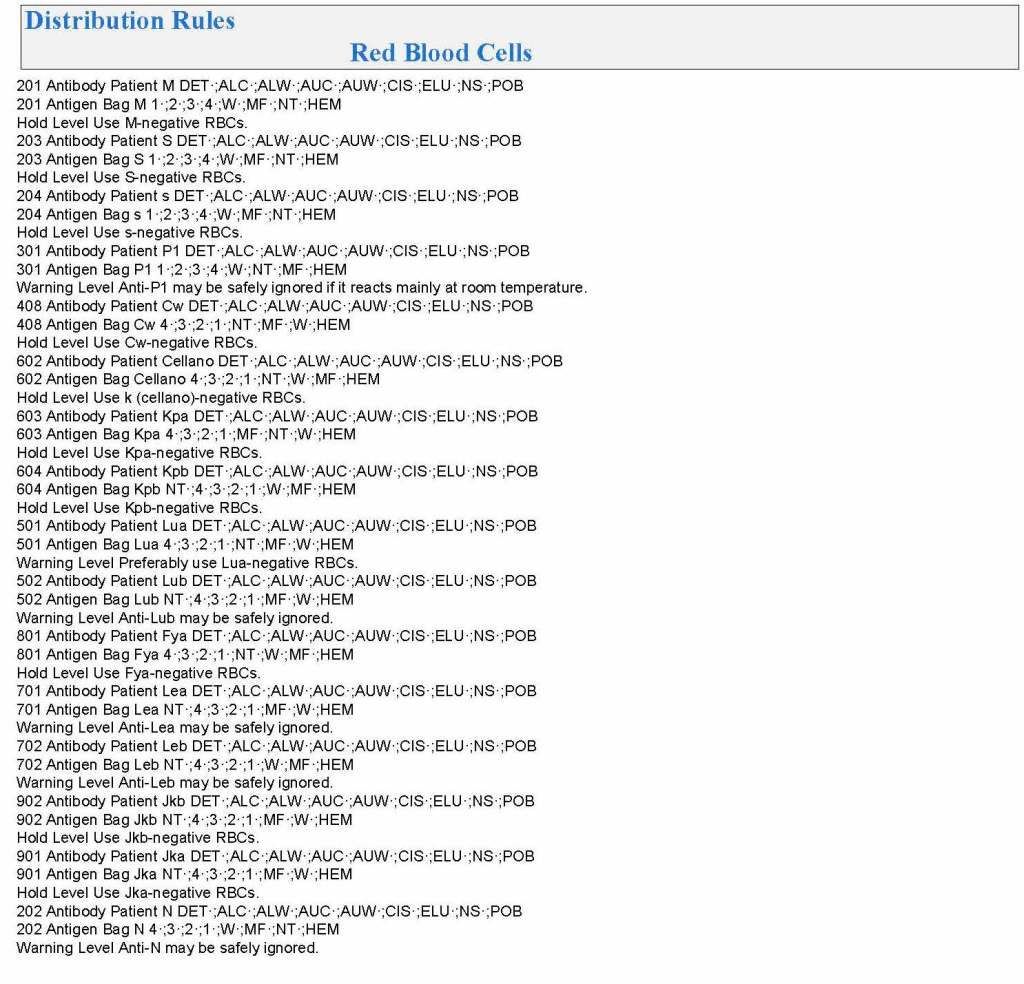
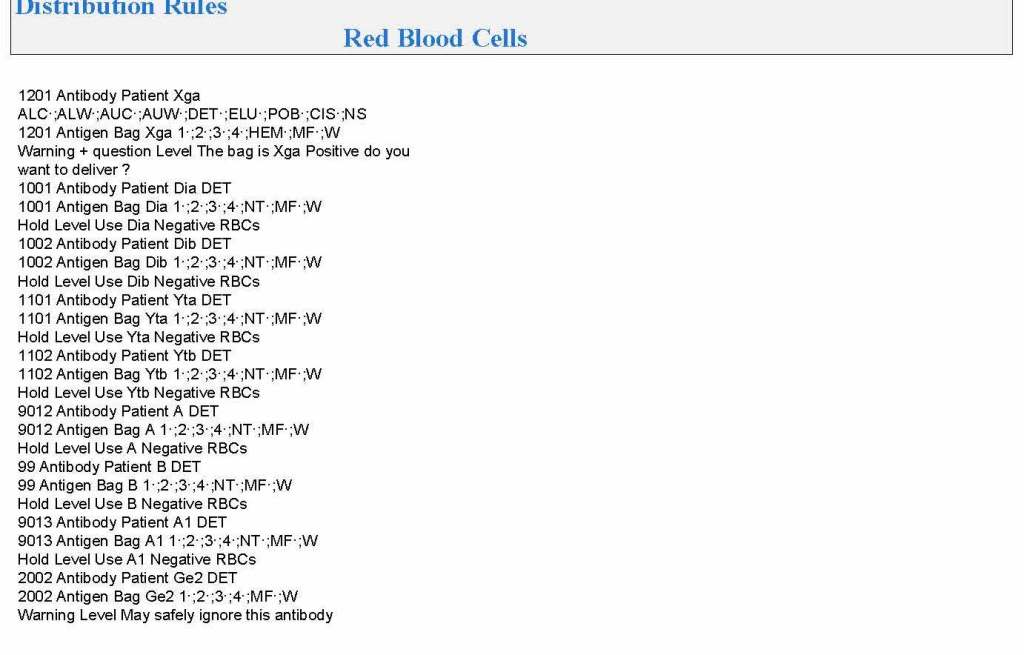
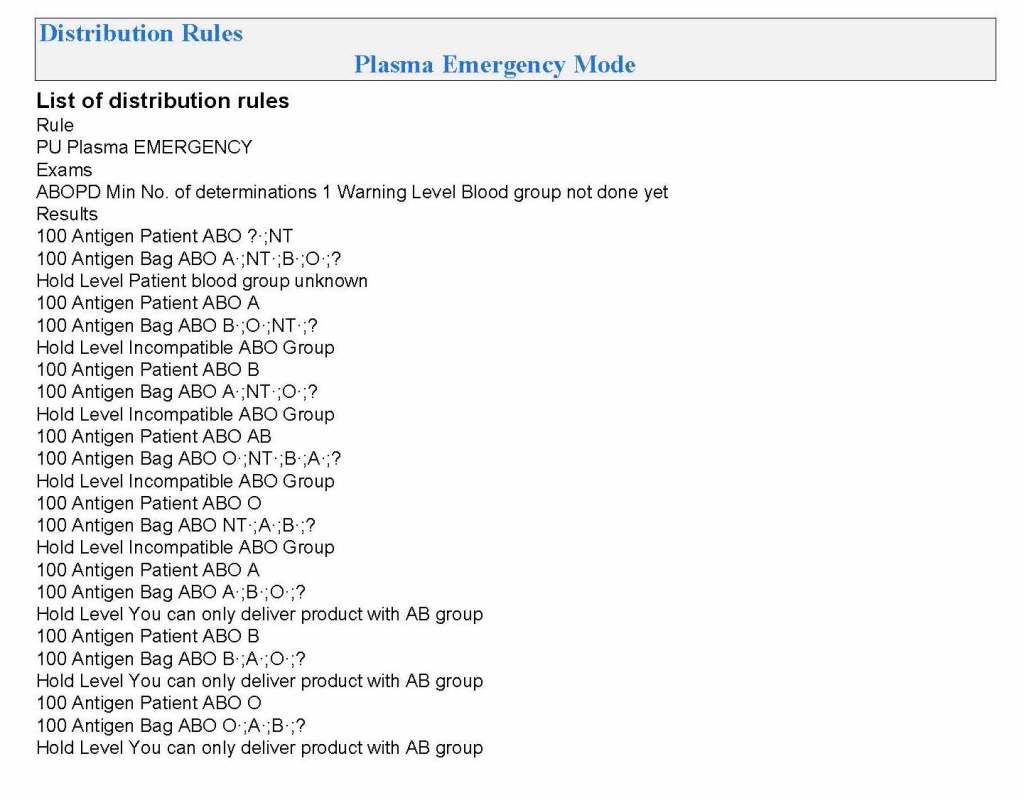
In rapid turn-around events, it is most helpful to have a robust blood bank computer system that can scale to the challenge. Also, it must mercilessly enforce all the rules starting with donor qualification, screening, collection through testing and production. At times of emergency, it is difficult to meet Good Manufacturing Processes manually.
After each major shortage, I recommend a “post-mortem” analysis of the situation with senior donor and quality staff to analyze our processes and see if we can further optimize them for the future. A report is prepared and reviewed by me as the Division Head/Medical Director of the Blood Bank. If possible, we implement our recommendations. If not, I request additional resources from the Administration.
As regards Disaster Planning, I always asked Administration how many victims did they want to save? When I got the response, I always try to adjust inventory by two extra RBCs and one adult platelet dose (> 2E11) per salvageable victim. This may come at the expense of increased wastage, especially in a region that cannot export the excess, unused stock.
The exasperating issue is that I didn’t get a clear answer on this last point. What number should I use? I made a spreadsheet showing calculations for a variety of endpoints, e.g. 100, 500, 1000 treatable victims and sent this to Administration to consider.