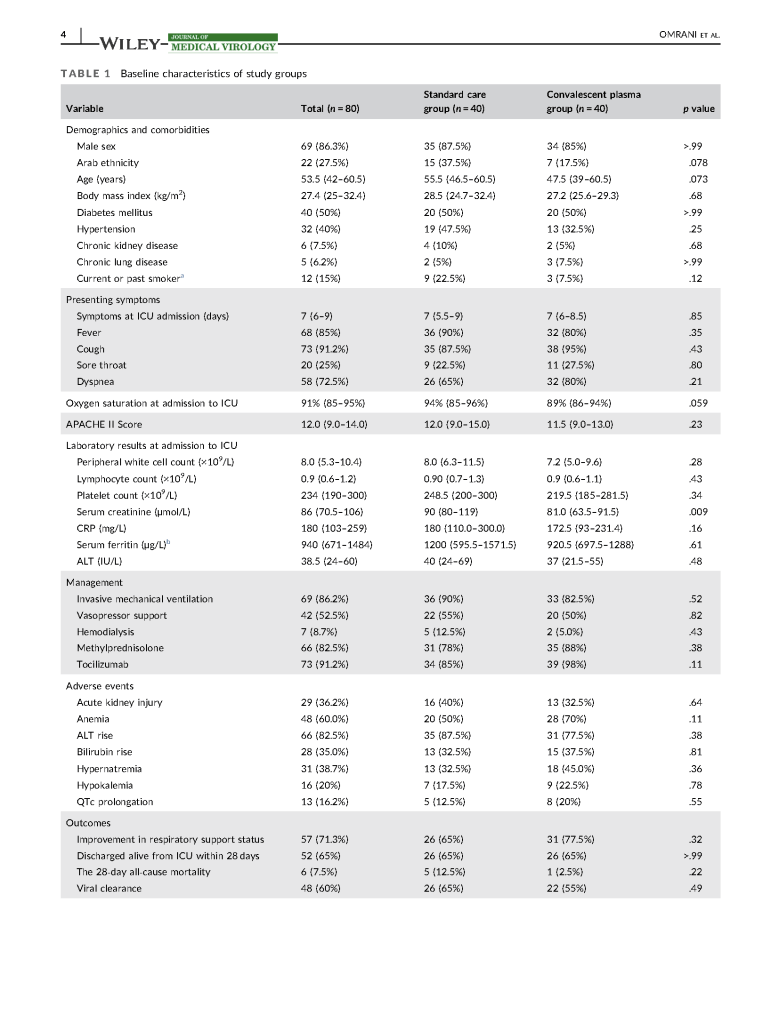
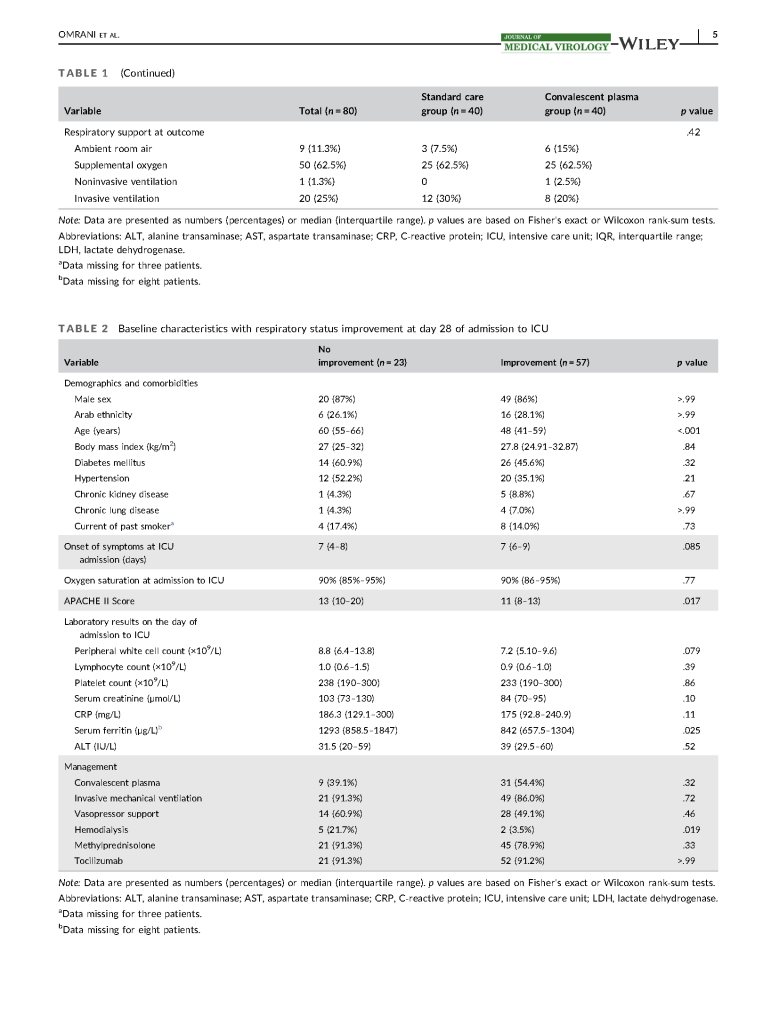
I am a co-author on this paper just being released titled The impact of COVID-19 Pandemic on Blood Supplies and Transfusion Services in the Eastern Mediterranean Region. It compares the responses made by different countries in the region.
Tag: COVID-19 Plasma
COVID-19 convalescent plasma
COVID-19 Convalescent Plasma CCP Donor Questionnaire and Collection
This is a part of a series of posts on the actual Medinfo design of the CCP donation and release processes. The site and donor registrations were covered in a recent previous post.
Donor Questionnaire and Physical Examination:
After registration, there is the online CCP donor questionnaire and vital signs entry.
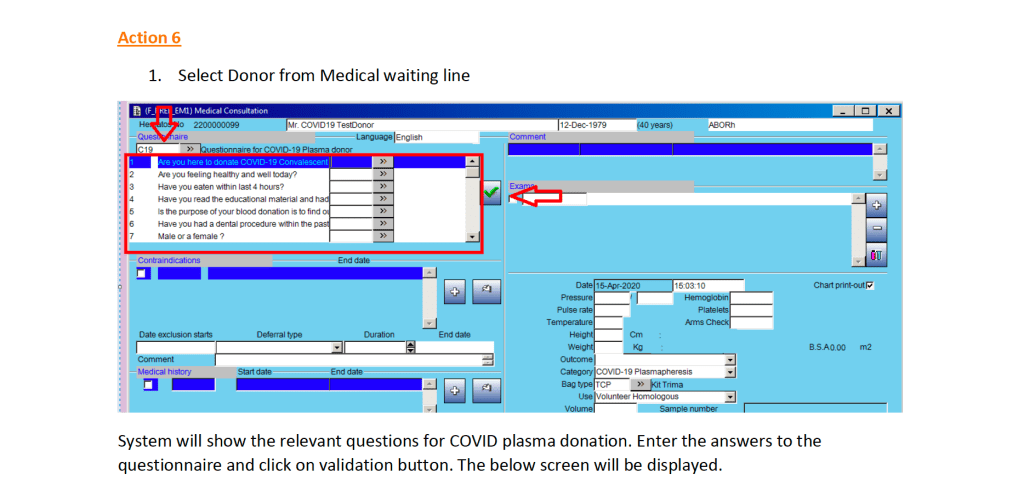
Note that the CCP donor will automatically be excluded from other types of donation. All other types will appear as contraindications in RED below.
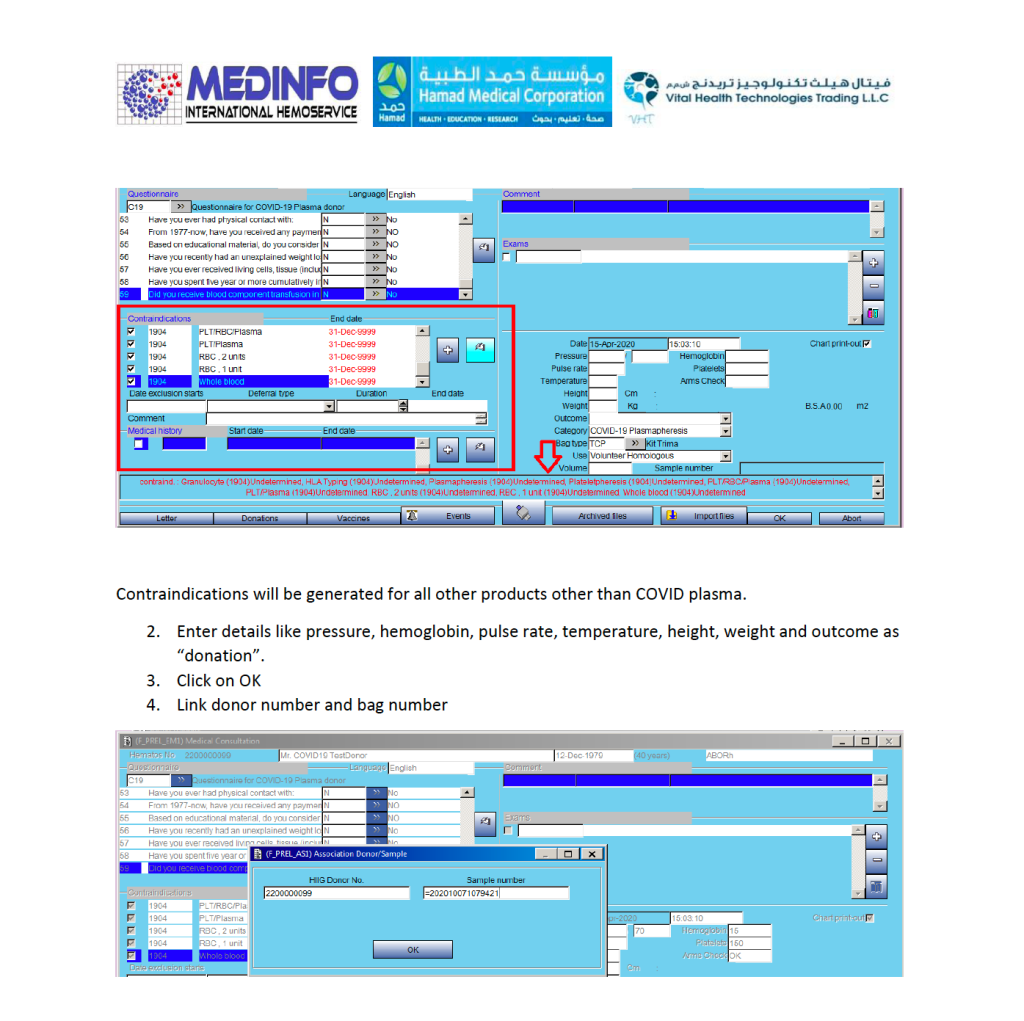
Donor Apheresis Collection:
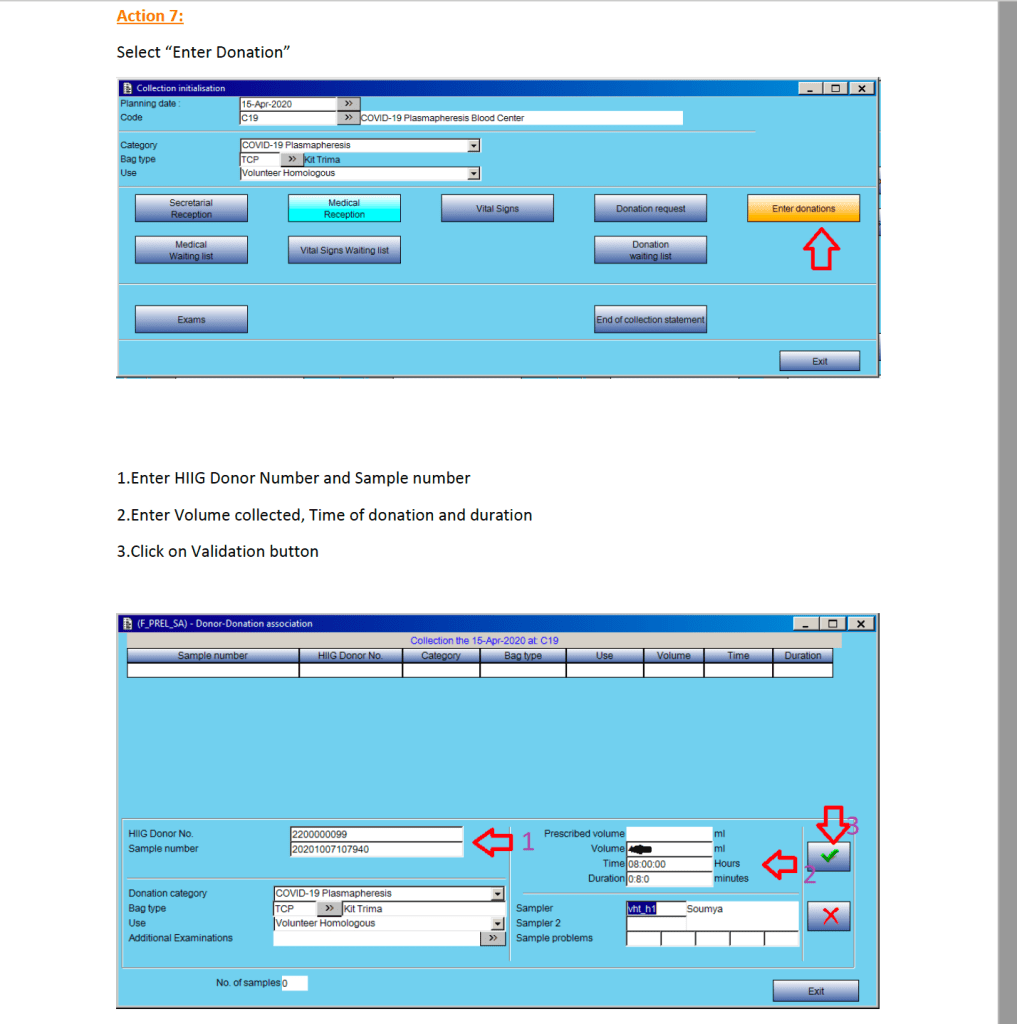
The actual donation process is the same as for plasmapheresis donors:

Specifications for the CCP Blood Bank-Hospital Information System Interface
At HMC Doha, we had a limited bidirectional interface between Medinfo Hematos IIG blood bank software donor and patient modules and the hospital information system HIS Cerner Millennium for blood component and limited blood bank testing ordering and return of the blood component ordering statuses and all blood bank test results.
For the purpose of COVID Convalescent Plasma CCP, the following specifications applied:
- Ordering of CCP was either by number of units (average 220 ml each) or volume in ml (up to 200 ml).
- Maximum order was 2 units.
- Only designated physicians could order CCP.
- A valid type and screen (72 hour maximal validity) had to be in effect to place an order for CCP.
- If not valid, a new type and screen must be ordered before ordering CCP.
- Selection of the ABO type of CCP was at the discretion of blood bank using its standard algorithms.
- Status of order (ordered, received in blood bank, in process in blood bank, or released) would show in the HIS.
Note: Bedside documentation of the component transfusion was the NOT the responsibility of Transfusion Medicine or Medinfo Hematos IIG.
Specifications for Medinfo COVID Convalescent Plasma CCP Processes
These are my specifications used in April, 2020 at HMC Qatar for setting up the CCP processes in Medinfo Hematos IIG:


COVID-19 Convalescent Plasma CCP Donor Registration
I designed a completely quarantined process for collection, processing, and release of CCP at HMC Doha. This document shows the Medinfo process for donor registration as a separate donor center code.
Check donor history and donor deferral database. If there is no previous encounter, generate a new donor file:

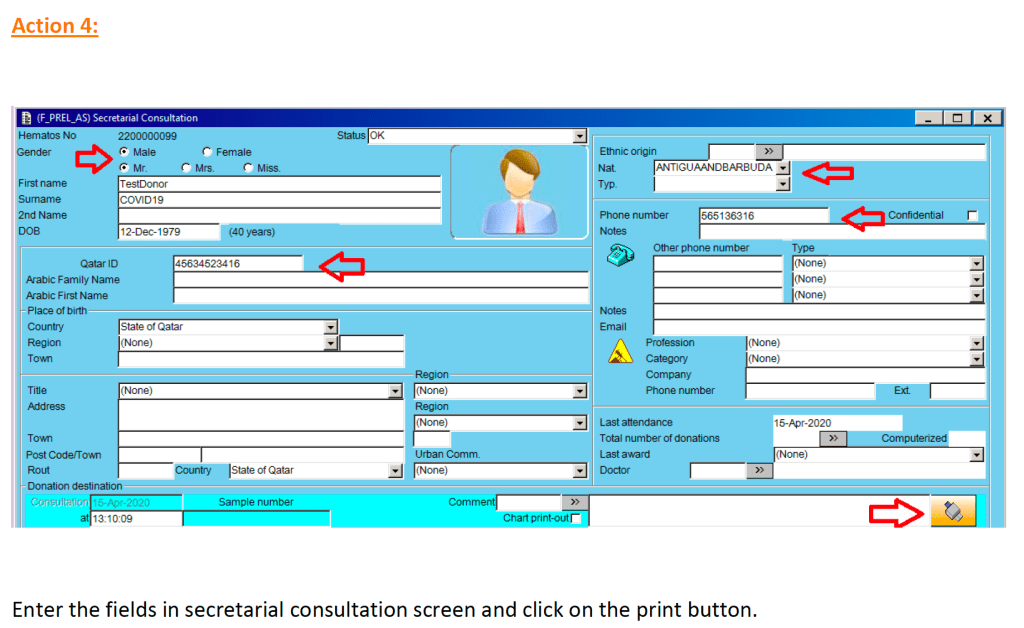
At the completion of this action, the Blood Donation Record with the donor unit number (in this example 2200000099) and consent form in English and Arabic is generated.

CCP could only be collected at this special site and only the apheresis bag could be used for collection. Regular donation options were not available at this CCP site nor was CCP collection an option at the regular donation sites.
8/1/21
COVID-19 Convalescent Plasma CCP Site Registration
I designed a completely quarantined process for collection, processing, and release of CCP at HMC Doha. This document shows the Medinfo process for site registration as a separate donor center code.
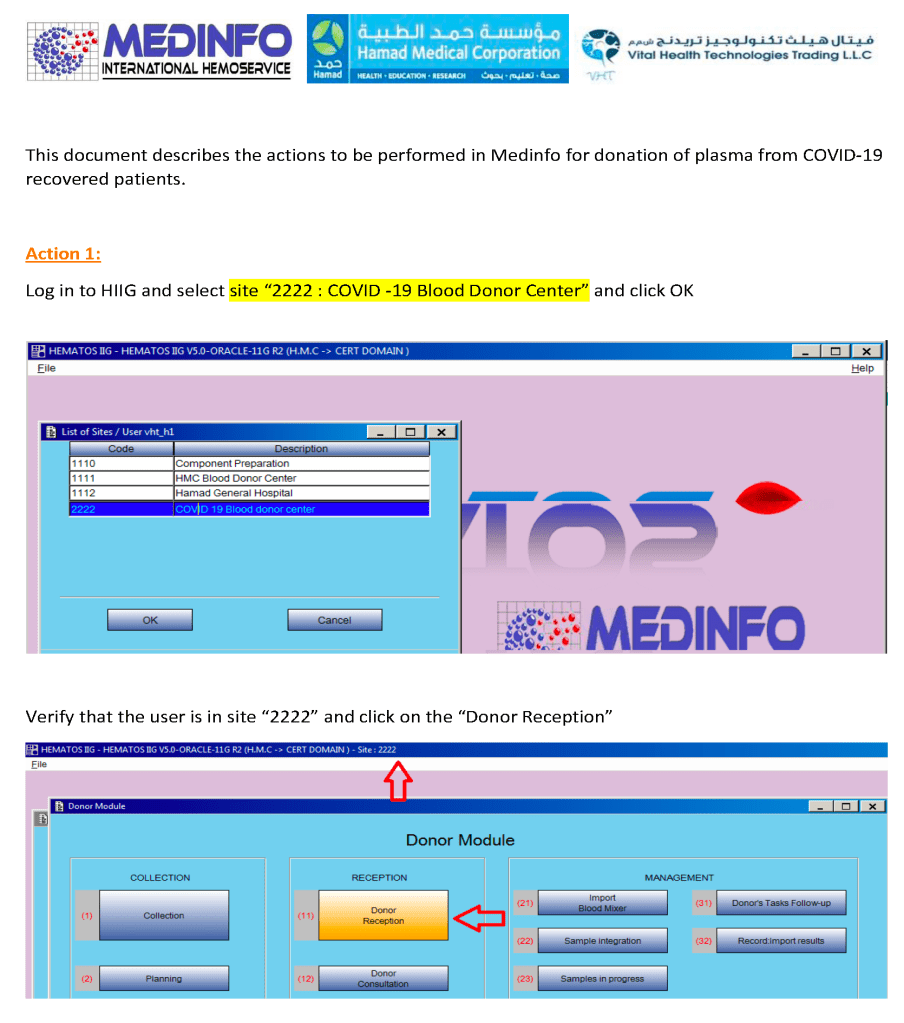

CCP could only be collected at this special site and only the apheresis bag could be used for collection. Regular donation options were not available at this CCP site nor was CCP collection an option at the regular donation sites.
4/1/21
COVID-19 Convalescent Plasma CCP Series Introduction
I will be posting a detailed series about the manual and software-enhanced COVID-19 processes that I set up in Qatar at HMC Doha in March-April 2020.
In this series I will provide you with screen shots of my Medinfo Hematos IIG software design for each step in the process: collection, processing, testing, inter-depot transfer, and hospital transfusion service/blood bank release.
This GMP-compliant software-enhanced system is based on the manual system I set up in early March 2020 at HMC.
I want to thank Medinfo Hematos IIG for their rapid response to building this parallel system based on my standard processes in so short a time (two weeks) and my special thanks to the software engineering team at Vital Health Technologies, the agent for Medinfo in Qatar.
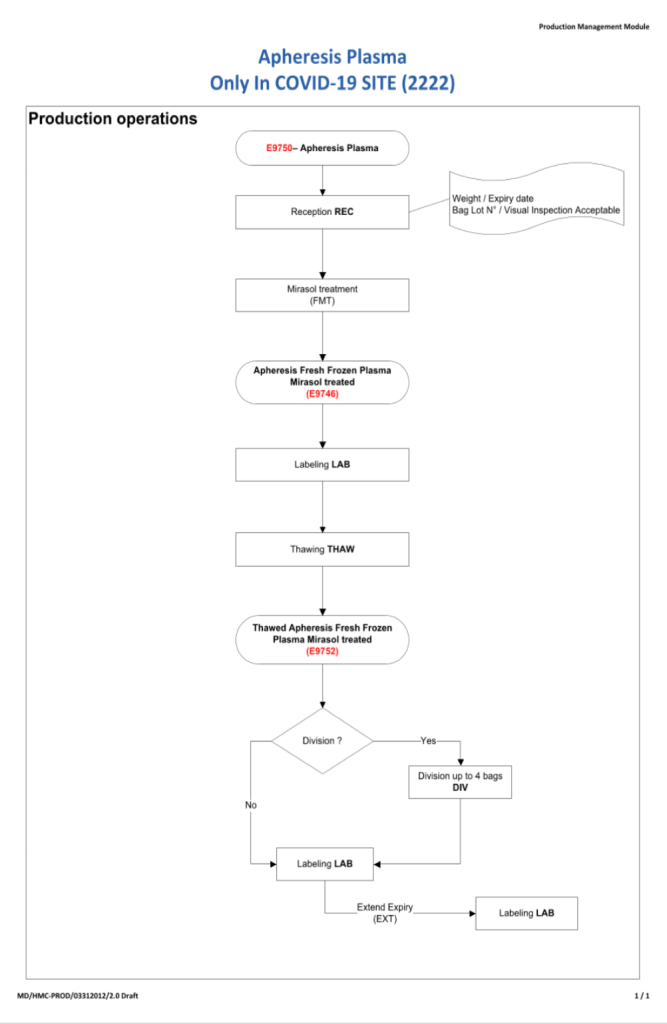
To start the series, I am providing the basic workflow for the system. As is normal in Medinfo software design, a full mapping of the processes are made. This workflow shows the new CCP ISBT codes and the quarantine collection and processing steps. The donor testing (marker and immunohematology) processes are similar to those for regular donor units.
This is basically the same process both manually and in the software. I always say:
A good software process is based on a good manual process!!
Please note the following workflow for our initial discussion.
Pre-Screening for CCP Patients
This is a review/update of this document prepared early in the course of our COVID-19 Convalescent Plasma CCP collections. It now includes testing of specimens not only for donor marker testing but also COVID-19 antibody titers.
All blood components are considered medications and are subject to Good Manufacturing Practices as mandated by international accreditation standards. The whole process must be done reproducibly and precisely by specific personnel trained and documented to be competent. This includes collection of convalescent COVID-19 plasma.
Transfusion Medicine will provide staff who are deemed competent for the entire process of the collection, manufacture, and release of this unlicensed, emergency-contingency component.
It will help greatly if all candidates are prescreened to exclude the following candidates:
- Administrative:
- Donors must come with a valid Qatari identity card: no ID means no screening
- Sex:
- Males only to minimize the risk for transfusion-associated lung injury TRALI
- Donor Feeling:
- If the donor does not feel well, he should not come for screening/collection.
- Food/Drink:
- Donor must have eaten/drunk fluids within 4 hours of arrival for screening/collection.
- Medication exclusions:
- Antibiotics within the past 14 days
- ACE inhibitors in the past 48 hours
- Beta blockers
- Anticoagulants
- Anti-anxiety or other psychotropic medications
- Other medications on the Unified Donor Questionnaire Deferral List
- Medical exclusions:
- Stable vital signs
- History of seizures
- History of dementia or other chronic neurologic disorder
- Family history of dementia or other chronic neurologic disorder
- Significant cardiac arrhythmias
- History of hepatitis B, hepatitis C, HIV, brucellosis, Ebola
- Travel history:
- 5 years cumulative residence in Europe including Ireland and France 1980-2001
- 3 months cumulative residence in the UK (and/or all its territories) 1980-1996
- Any visit(s) to West Africa
- Testing:
- Antibody titers should be performed to exclude candidates with low-titer or absence of antibodies.
- Regular donor marker testing (excluding malaria and HTLV 1/2)
All processes will continue to be performed in the dedicated blood bank computer system. The COVID-19 antibody titers will be part of the donation record.
This is NOT a complete list of criteria. Transfusion Medicine personnel will screen according to the full donor criteria. Thus, donors passing the pre-screening may still be otherwise disqualified based on the detailed process.
8/11/20
COVID-19 Convalescent Plasma Revisited:
In February, 2020, I developed a program for convalescent COVID-19 plasma at Hamad Medical Corporation in Doha. In early March, 2020, our program started collecting CCP by apheresis. We started before the software modifications were completed since there were urgent requests by the clinicians for the product.
I proposed the software specifications and our vendor Medinfo Hematos IIG implemented them within 2-3 weeks, after which they were implemented/validated
Thus, now we have 8 month experience has been 8 months since starting manually and more than 7 months using a specific modification of our blood bank software Medinfo.
A complete manual system was implemented with quarantined registration, screening, collection, processing, and release. Only the donor marker testing was shared with the regular donors. This was built into the computer system.
Upon review, these are my current thoughts on our processes:
- Actively monitor supply requests: Keep good communication between ordering/treating physicians and apheresis unit to optimize the stock according to patient needs.
- Collect/process/release separately from regular donations.
- Use dedicated quarantine equipment (apheresis, processing, storage refrigerators)
- Collect manufacturer’s recommended maximum of plasma based on body weight.
- Use pre-donation screening to allow quick release of components and avoid wasting apheresis kits.
- Repeat testing on the new specimen collected at the time of apheresis donation.
- Process units by same processes used for normal donations, including pathogen-inactivation.
- Use standard processes for release of blood components to end-users.
- Restrict ordering to designated treating COVID-19 physicians (enforce in computer system)
- Restrict release of CCP to designated non-blood bank staff from the quarantine storage location (enforce in computer system)
Notes:
- Include COVID-19 antibody testing and establish a threshold level (e.g. 1:128 titer) for donor qualification. Do not collect if low-titer or absence of COVID-19 antibodies. Store titer information with donation record. Add antibody results to donation records that occurred before the assay was available.
- Review of donor criteria: are there increased risks using these recovered donors: cardiac or respiratory risk? Is there a way to continuously monitor CCP donors’s vital signs during the donation?
- Collect apheresis components only in pre-screened donors: Apheresis kits are expensive, use them only if the donor is prequalified, continue to retest when actual apheresis donation occurs
- Allow use of units directly after collection/processing as long as the other donor processing steps have been completed (allow blood bank computer system to use pre-donation specimen for marker testing criteria).
Covid Convalescent Plasma Treatment in Qatar 2020
In February 2020, I established COVID-19 plasma collection and distribution for the state of Qatar. Here is a research paper prepared in conjunction with the Qatar Communicable Disease Center about using it for patients there.