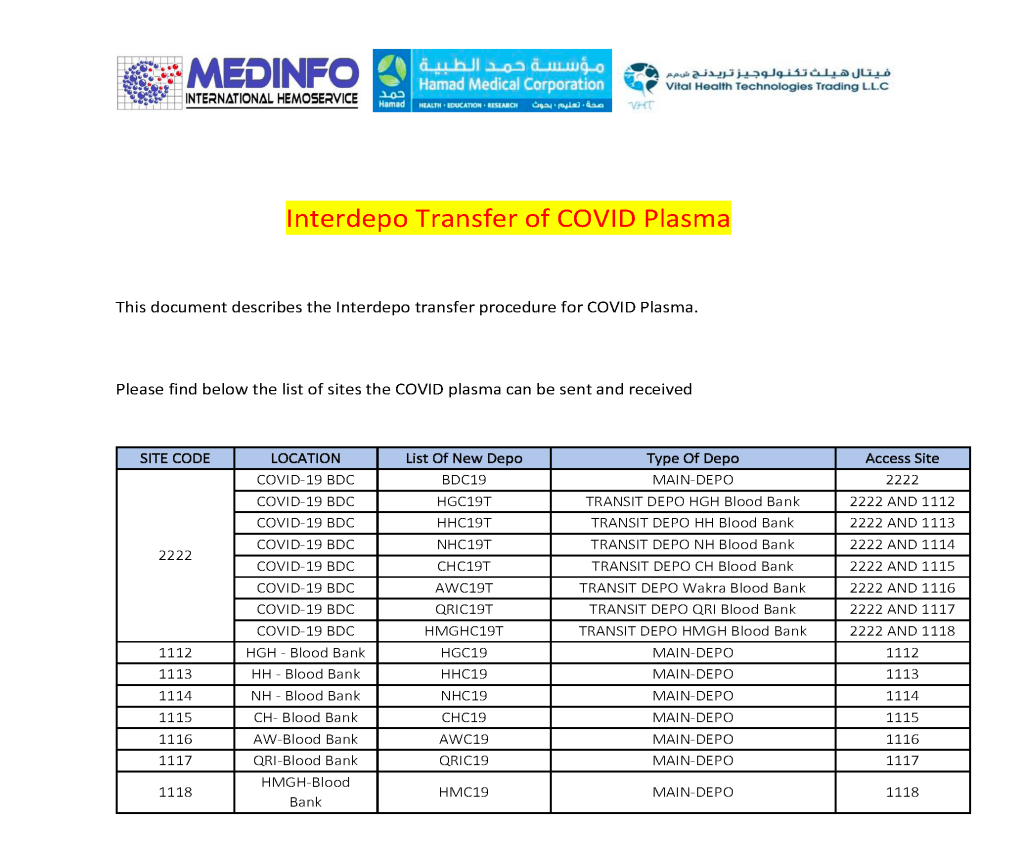
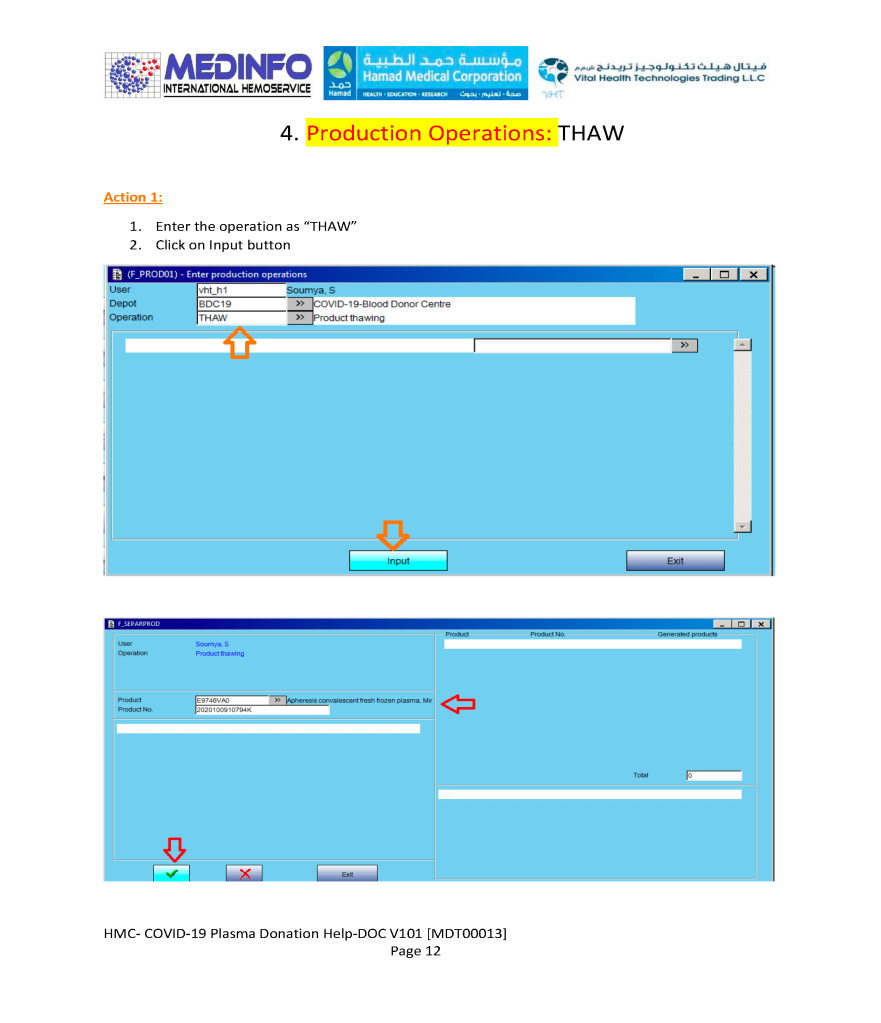
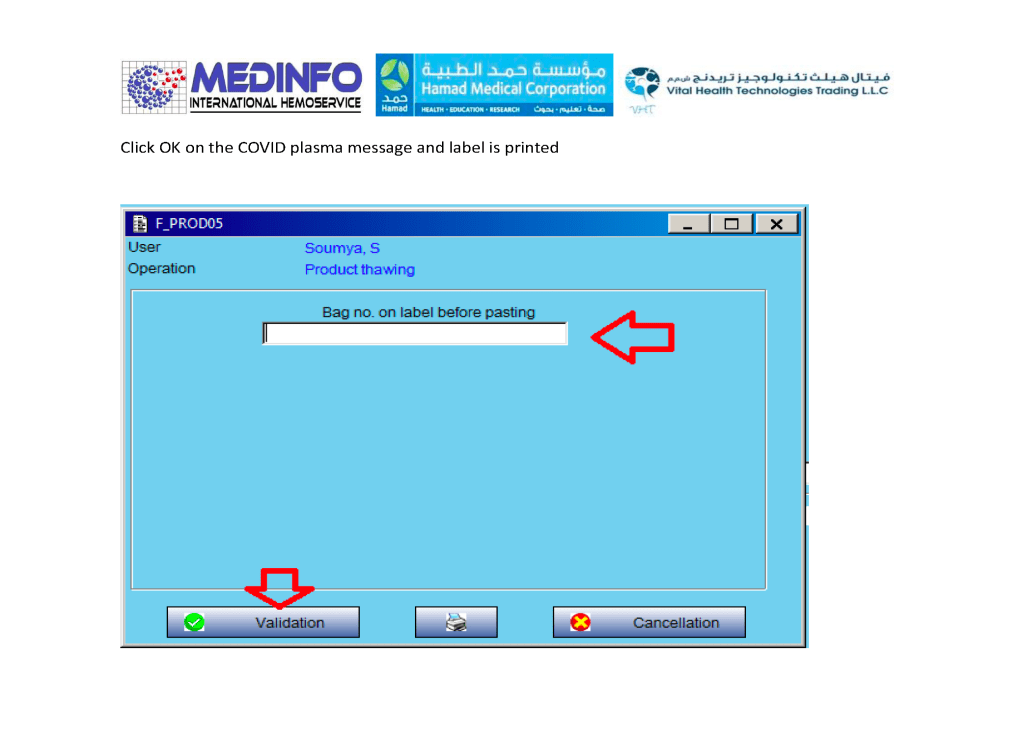
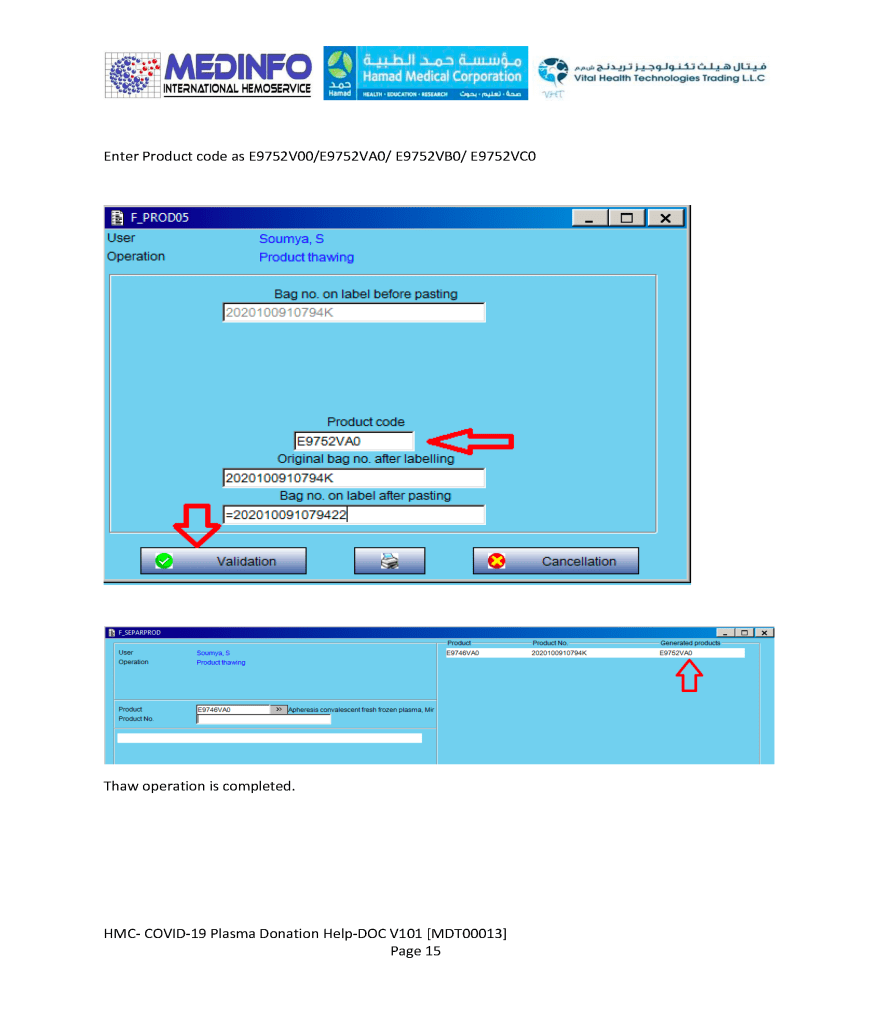
The COVID-19 pandemic imposed new challenges to our system. In general, these could be divided into:
- Decreased donors
- COVID vaccine effects
- Decreased available staff
- Shortages of supplies
- More demands on donor apheresis staff—CCP
- More demands on donor processing staff—CCP
- More demands on hospital transfusion service/blood bank staff—CCP
There were fewer donors in the early phase and the nurses also had to add a large number of donor plasmapheresis collections for COVID convalescent plasma CCP. Still they had to maintain all donor and therapeutic apheresis services with no increase in staff. Although elective procedures had been cancelled, there were still obstetrical, oncologic, and trauma services in full action.
Many of our staff were on leave when the borders were closed. Some had to wait months before they could return to work. Others had COVID-19 infection and were quarantined for several weeks. This further reduced staffing. We could not just hire outside staff since considerable training is involved in these processes.
I dedicated a separate donor collection space for the CCP program away from the regular donors as well as a quarantine processing area. Similarly, the CCP plasma was kept segregated from the regular plasma supply and a specially designed location was identified for release of this product. Working for this program diverted resources from blood collection to this special project, again without increasing resources.
With disruptions to shipments of supplies, including the Reveos whole blood kits and Trima donor apheresis sets, we had to rely on our large in-home inventory until the situation stabilized. We prescreened the CCP donor candidates before we would collect them to avoid wastage of kits.
Fortunately, our throughput was minimally affected because our equipment and processes had always stressed speed. We used single-well NAT testing to minimize the need of additional runs. Also, we used Reveos automated component processing to greatly speed production (one Reveos can process four whole blood units in about 23 minutes or about 12 units in 75 minutes.) One technologist could operate all 4 of our machines simultaneously and perform other tasks while the machines were working.
In the system I developed in Qatar, we could complete processing into components (RBCs, buffy coat platelet pools, leukodepleted plasma), all marker and immunohematology testing, leukoreduction of the pools and RBCs, Mirasol pathogen inactivation, and platelet additive solution in as little as five hours.
In rapid turn-around events, it is most helpful to have a robust blood bank computer system that can scale to the challenge. Also, it must mercilessly enforce all the rules starting with donor qualification, screening, collection through testing and production. At times of emergency, it is difficult to meet Good Manufacturing Processes manually.
I had built parallel separate donor collection, donor processing, and transfusion service/hospital blood bank processes specifically for CCP and had to staff them with available personnel, limited our capability to process regular donors. The blood bank computer software restricted CCP use to designated physicians and transfusing locations. For those interested, there is a separate series of posts about the CCP project and its implementation in the dedicated blood bank Medinfo HIIG.
COVID-19 vaccinations should have minimal effect in donor qualification since mRNA or antigen-based ones do not cause donor deferral. Live attenuated COVID vaccines will defer donors for 2 weeks by current rules—the same as other live vaccines. Donors who had previously received CCP will be deferred for three (3) months after last receiving this product.
In summary, the COVID pandemic reduced staffing and affected donor recruitment. We had production mitigations to maximize throughput. The system was stressed by the reduced staffing and special demands to produce CCP. However, the extent of our automation allowed us to maintain throughput throughout the crisis.