When I first moved overseas from the United States, I brought the perspective of my American training and experience. I saw everything in my new blood bank through those eyes.
Yet, most of my staff were not American or even North American. Few were even native in English, and most of those were not American. They had different qualifications, many of which would not have been accepted by the American schemes. Still, they functioned well.
I also worked with the US military technologist staff during Gulf War One. Some did not even have a Bachelor’s degree; yet, they performed the work well.
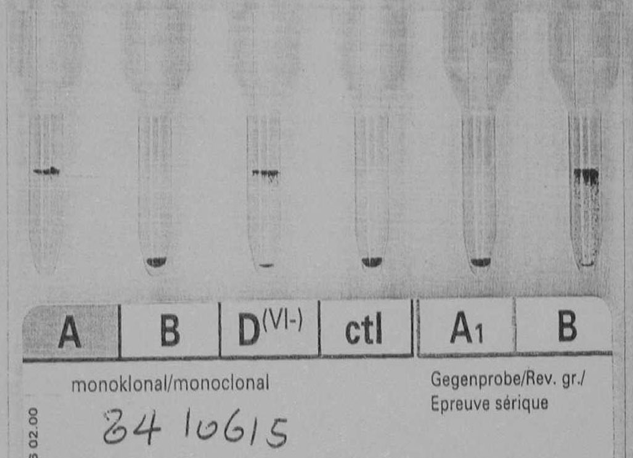
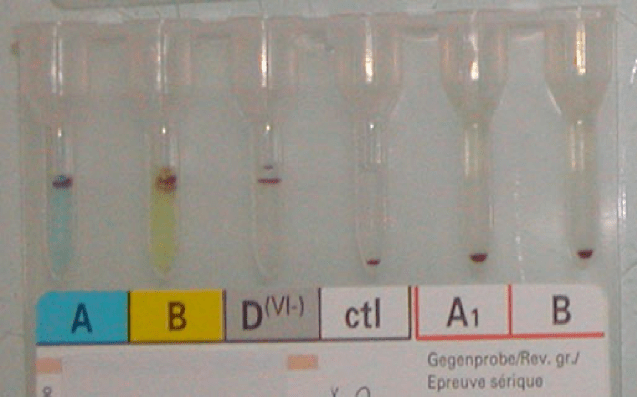
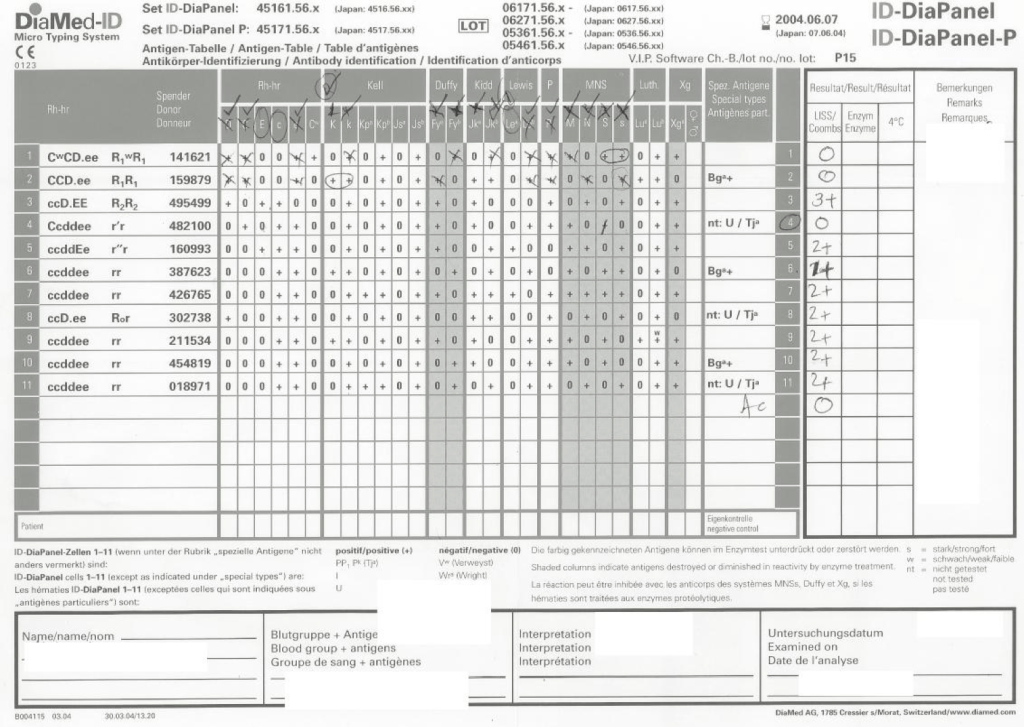
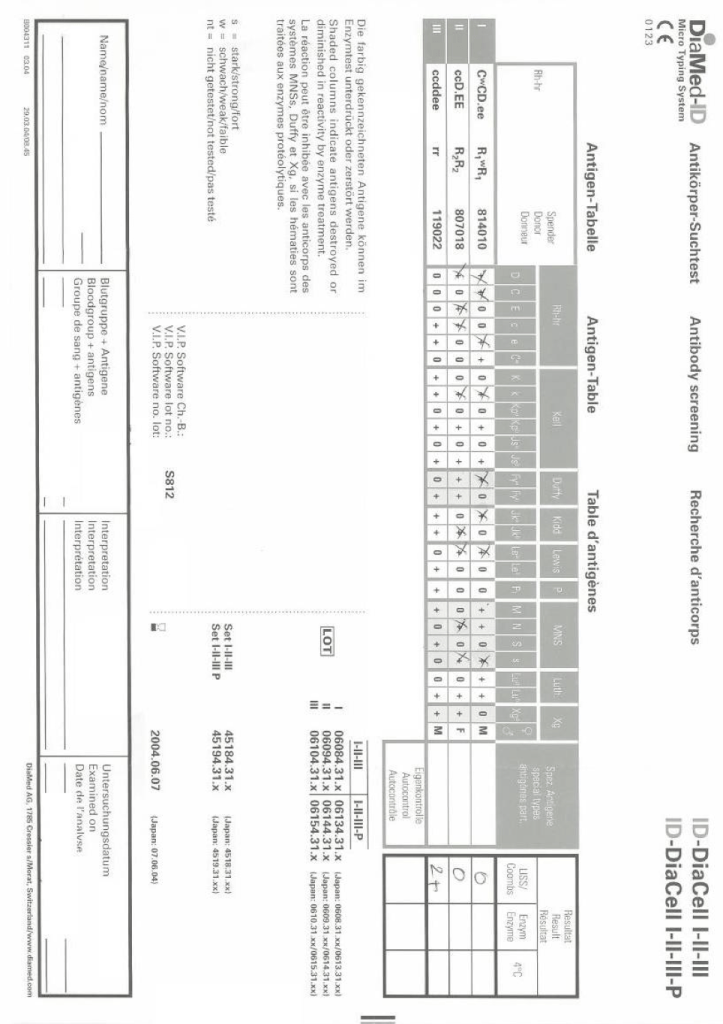
I used many technologies that were not yet (or never) US FDA approved such as gel or glass bead typings and pooled buffy coat platelet production. There were rare reagents I could buy off the shelf (e.g. anti-Tja/PP1Pk).
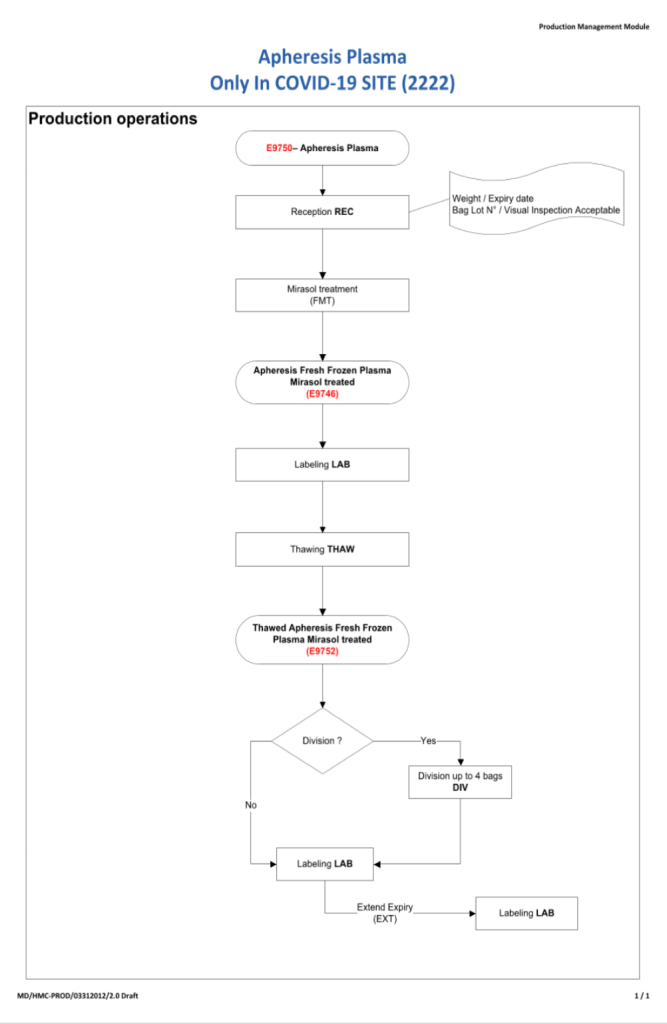
Later, I adopted pathogen-reduction technology (Mirasol), automated component production (Atreus then Reveos), and platelet additive solution. I achieve a level of good manufacturing practice that would have been difficult to achieve by the FDA-approved methods.
My perspective had changed. In the Middle East, I studied many frameworks and came to the conclusion that the best approach was to customize them to our local needs. My particular experience was to start with one framework, i.e. Council of Europe CE, and then localize it.
To do this, I could not use an American turnkey blood bank software for either the donor or patient operations. I needed a flexible system that could be customized to my needs. Again, I chose a CE-marked system, Medinfo Hematos IIG that had already been adapted to many frameworks.
It is much easier to work solely within one system such as FDA. However, if I had done that, I would have lost so much flexibility and not had a system optimized for local conditions. I would not have used Mirasol, Reveos, Diamed, and many other reagents.
One big disappointment at such international meetings is the perspective by one country’s regulatory agency that they feel its regulations and framework will work well overseas. I would wager that those people were not well acquainted with international conditions.
Another frustration was attending another international meeting in which the presenters apologized for the source of information since it came from a foreign country (France) and not their own (United States).
No country has a monopoly on what is best for everyone. To share our experiences and compare is so valuable. No one assume his way is the best. In my career, I have had the richest experiences studying other perspectives and my organizations have benefited greatly from the exchange. We can all learn from each other. We are citizens of the world.