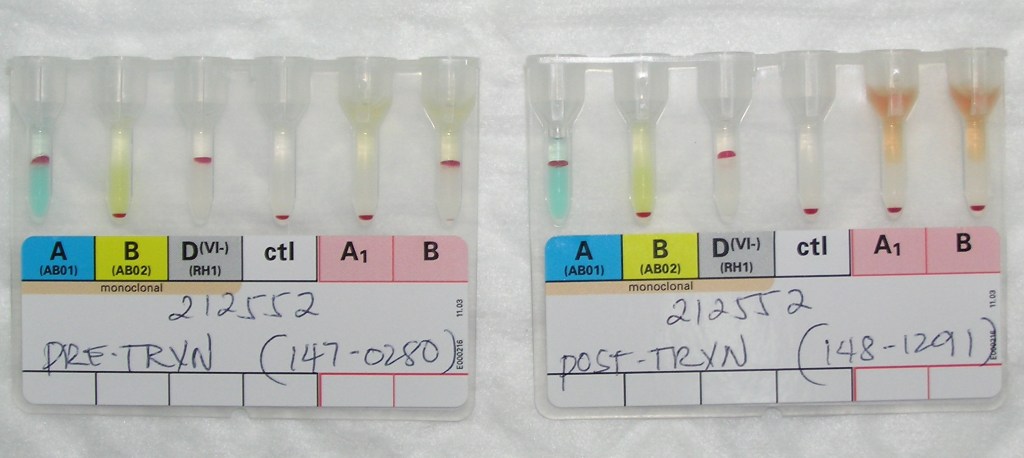
You can have the most sophisticated blood bank software, but if you can’t read the labels or if they fall off, you have a disaster. These are my thoughts from implementing our blood bank computer system back in 2013.
Check Digits:
Both ISBT specimen and product labels have an internal system to verify that they have been read correctly. Within the blood bank software, this should not be a problem. However, can your third party such as a hospital information system HIS read them?
ISBT Compatibility:
The institution’s HIS could not read the component labels. To this date, the problem has not been fixed. As a workaround, we sent them the ISBT label codes directly from the blood bank software. The only complete transfusion record was in the dedicated blood bank computer system, not the HIS. You could not rely on the bedside nursing entry at all.
The HIS did not use the ISBT database and had no values for the E codes. Again, we had no choice but to send an abbreviated E-code descriptor to them. We did not use their transfusion module at all, but one of our clients did. They had to hard code the list of E codes in use with their descriptors into their HIS.
Label Adhesive
We tested candidate labels at room temperature, 1-6, minus 18, and minus 80 C. We found that most of the labels’ adhesive were not sticking at minus 80. For some, you could literally blow on the blood bag and the label fell off. I imagined a scenario in which I opened a freezer and saw the blood bag labels all lying separately at the bottom.
RFID Tags:
Do you use an RFID tag integrally attached to the ISBT label OR do you stick a separate RFID tag? If the latter, how do you ensure that you put the proper tag on the proper bag?
Readability:
Readability: Can all your blood bank devices read your printed labels? Do you have to adjust the printers for this? Whose responsibility is it to do this?
Labels printed outside the blood bank:
If you receive patient specimens from outside the blood bank, can your devices read them? Who is responsible to adjust the printers in the wards and clinics?
Validations:
Who validated that the HIS prints the accurate complete label for the right patient? We discovered that this was not the case with our HIS and needed correction by them. Remember that any processes affecting Transfusion Medicine should be assessed by Transfusion Medicine. Do not accept verbal assurances from anyone, not even your HIS.
2/2/21