This is a competency assessment for non-physician, non-nursing staff to transport blood from the hospital transfusion services/blood bank to the emergency department and operating rooms. This was designed by my Quality Manager, Ms. Edith Durante.

Teaching medical and technical staff transfusion medicine
This is a competency assessment for non-physician, non-nursing staff to transport blood from the hospital transfusion services/blood bank to the emergency department and operating rooms. This was designed by my Quality Manager, Ms. Edith Durante.

Below is a sample document specification for a document control process. It was originally designed but never implemented due to changes in the administrative structure in that organization. I want to thank Ms. Editha Durante, Quality Manager for preparing this.






Principle:
Blood components must be maintained at specified temperatures to avoid hemolysis, bacterial contamination, and maintain full efficacy (e.g. coagulation factor activity.)
Policy:
Note the different temperature ranges for refrigerated components stored in the blood bank (1-6C) versus transport (1-10C).
References:
Section 5.6.5, Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
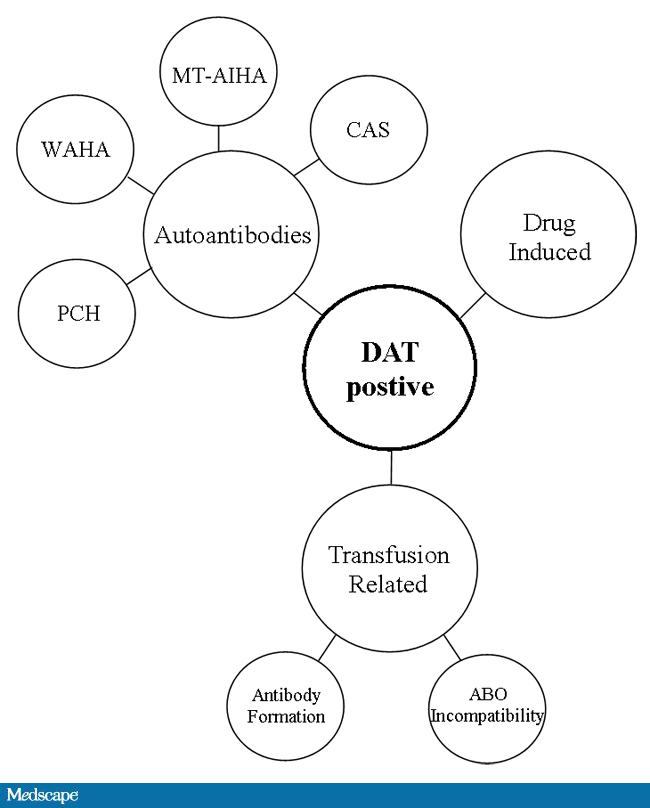
This is a helpful Medscape diagram showing causes of direct-antiglobulin-test-positive hemolytic anemias. Note that not all cases of the conditions may consistently be DAT-positive, especially the ones that fix complement. Again, a negative result means non-detected, not necessarily not present.
In my own test algorithm, if hemolysis is suspected, I would perform monospecific IgG and C3 DATs. I might also include a special DAT card detecting both C3c and C3d and heavy-chains mu (IgM) and alpha (IgA) if the initial DAT is negative. C3c positivity would tell me that active complement activation is occurring.
This a revised version of a previous post for the processes of autologous transfusion that I developed at HMC Doha. It can serve as a template for other sites and was also a teaching document for the Transfusion Committee members.
Background:
There are four basic types of autologous transfusion: preoperative, perioperative hemodilution, intraoperative, and postoperative drainage/collection. The use of all of the above techniques can significantly decrease the need for homologous blood and as an added benefit reduce the risk of the disease transmission and immunosuppressive effects of such homologous transfusions.
Preoperative collection can make available packed red blood cells, whole blood, platelets, FFP, and/or cryoprecipitate. However, at most two units of blood per week can be collected. RBC’s can be stored for up to 42 days in the liquid state, frozen RBC’s up to ten years, platelets up to five days, and fresh frozen plasma and cryoprecipitate up to one year. The last collection cannot be less than 72 hours prior to the surgery time. Units can be collected as long as the patient’s hematocrit remains above 33%. Supplemental iron and erythropoietin can increase the number of units harvested. The biggest obstacle to using this service is the coordination of the patient scheduling for this procedure. The blood bank does not have the resources to prospectively analyze the surgical scheduling and make the various appointments, contact the attending physician, etc. Thus, this service is vastly underutilized.
PHD or Perioperative hemodilution (also called acute normovolemic hemodilution) is useful in cases when the anticipated blood loss is at least one liter and the initial hematocrit is at least 34%. This includes essentially all types of surgery, but in particular cardiac, vascular, orthopedic, and urologic cases. The patient’s hematocrit Hct. is lowered to the range of 20-25% and the blood is replaced by crystalloid in a ratio of 3:1–i.e. three times as much fluid as blood, or in the case of colloid replacement, a 1:1 ratio of colloid plus 0.5 to 1.0 ml. of crystalloid. Crystalloid has the advantage of being readily removed by diuretic use. However, this technique should not be undertaken when vascular access is inadequate or appropriate monitoring devices are lacking. The physician performing PHD must be familiar with the compensatory mechanisms normally invoked when the hemoglobin is acutely lowered.
Another new twist to PHD is the perioperative collection of platelets by a special attachment to a cell-saving machine. This could allow collection of a typical apheresis load, about 6 to 10 units of fresh platelets for potential use. There are currently studies underway to determine if this has particular clinical advantages to warrant the additional cost.
Intraoperative salvage may be performed with a number of canister or automated devices. The latter is usually used when there are large volumes (usually 3 or more units) of blood to be salvaged. Depending on the body site, the recovered material is at least filtered and may or may not be washed. Care must be taken to collect the blood at a low suction rate and with minimal turbulence to minimize hemolysis.
Postoperative drainage collection of certain sites such as post-knee replacement surgery or chest wounds involves a canister collection device. This blood may or may not be filtered before reinfusion.
Note that perioperative and intraoperative material can only be transfused up to six or eight hours at room temperature or 24 hours if refrigerated at 1-6 degrees (depending on the method used) post collection to minimize the risk of infection. Intraoperative collection may be contraindicated in cases of cancer and if the bowel has been violated.
Other Issues:
The transfusion criteria for autologous blood is the same as for allogeneic units.
The same compatibility testing algorithm applies both the autologous and allogeneic units.
Policy:
References:
Medinfo Hematos IIG has an underlying framework of functionality. It is flexible since it acts upon rules based on parameters (e.g. sex, age, diagnosis, test results, etc.) You can change the processes in the system by changing the parameters without upsetting the underlying structure of the software. This means you can make changes very simply and quickly without having to “hard-code.”
Rules are based on parameters which are entered into the system by the user or the results of previous action. I am listing here some examples of parameters used to define processes in my Medinfo installations:
Based on these parameters, the system may require:
Examples:
Overriding rules:
One can also define if the rules can be overridden by someone with appropriate credentials:
On the other hand, rules can be specified to prohibit overriding:
Principle:
In accordance with AABB Standards, all actions contrary to the standard operating procedures and policies of Transfusion Medicine must be specifically approved by the Head, Transfusion Medicine or designate.
Documentation of variances must be organized in a system for ready retrieval for analysis. They should not be entered into a system that is cumbersome to find the entered variances.
Examples include but are not limited to: Rh(D)-incompatible transfusions, least-incompatible crossmatch, extension of expired rare reagents, etc.
If the same variance is occurring frequently, it should be determined if modifications in the underlying documentation (policies, processes, procedures) should be made.
Policy:
References:
I prepared this policy for HMC Qatar, which did not perform genotyping.
Principle:
Weakened expression of the D antigen may be due to either quantitative or qualitative defects. Qualitative defects (partial or mosaic D) are uncommon.
Patients with quantitative defects have the whole D molecule and will not make anti-D upon exposure to D-positive RBCs.
Patients with partial or mosaic D may form antibodies against the portion of the D molecule they lack. However, not all partial D types may make anti-D upon exposure. Since we cannot specifically test for this condition, we shall consider females of child-bearing age with weak D as potentially partial D and use D-negative RBCs.
Genotyping for the D gene may define better which partial D types need to receive RhIG or D-negative transfusions.
Definition:
Weak D is defined as weak (1+ or weaker) reactions with one or more routine reagents or reactions only at antiglobulin phase (i.e. Du testing).
Policy:
References:
Technical Manual, Current Edition, American Association of Blood Banks, Bethesda, Maryland, USA
Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
You can have the most sophisticated blood bank software, but if you can’t read the labels or if they fall off, you have a disaster. These are my thoughts from implementing our blood bank computer system back in 2013.
Check Digits:
Both ISBT specimen and product labels have an internal system to verify that they have been read correctly. Within the blood bank software, this should not be a problem. However, can your third party such as a hospital information system HIS read them?
ISBT Compatibility:
The institution’s HIS could not read the component labels. To this date, the problem has not been fixed. As a workaround, we sent them the ISBT label codes directly from the blood bank software. The only complete transfusion record was in the dedicated blood bank computer system, not the HIS. You could not rely on the bedside nursing entry at all.
The HIS did not use the ISBT database and had no values for the E codes. Again, we had no choice but to send an abbreviated E-code descriptor to them. We did not use their transfusion module at all, but one of our clients did. They had to hard code the list of E codes in use with their descriptors into their HIS.
Label Adhesive
We tested candidate labels at room temperature, 1-6, minus 18, and minus 80 C. We found that most of the labels’ adhesive were not sticking at minus 80. For some, you could literally blow on the blood bag and the label fell off. I imagined a scenario in which I opened a freezer and saw the blood bag labels all lying separately at the bottom.
RFID Tags:
Do you use an RFID tag integrally attached to the ISBT label OR do you stick a separate RFID tag? If the latter, how do you ensure that you put the proper tag on the proper bag?
Readability:
Readability: Can all your blood bank devices read your printed labels? Do you have to adjust the printers for this? Whose responsibility is it to do this?
Labels printed outside the blood bank:
If you receive patient specimens from outside the blood bank, can your devices read them? Who is responsible to adjust the printers in the wards and clinics?
Validations:
Who validated that the HIS prints the accurate complete label for the right patient? We discovered that this was not the case with our HIS and needed correction by them. Remember that any processes affecting Transfusion Medicine should be assessed by Transfusion Medicine. Do not accept verbal assurances from anyone, not even your HIS.
2/2/21