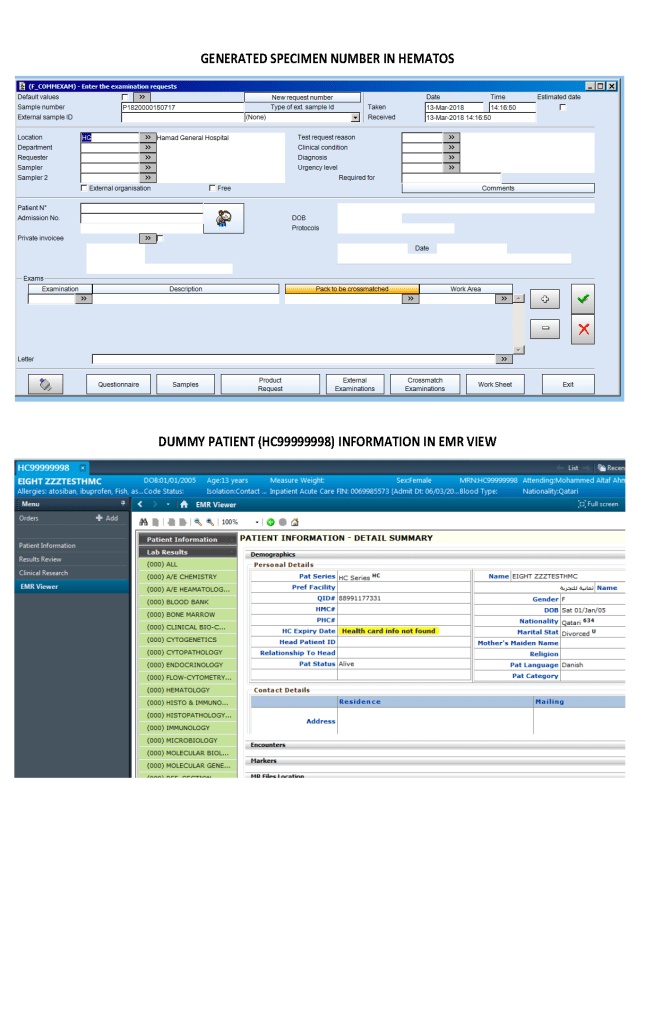
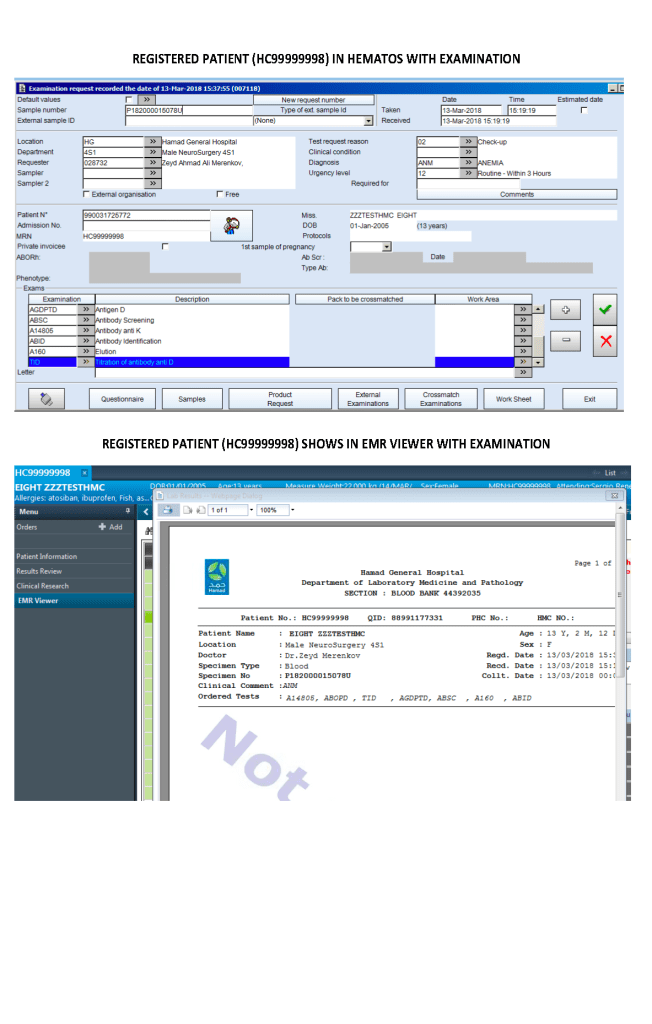
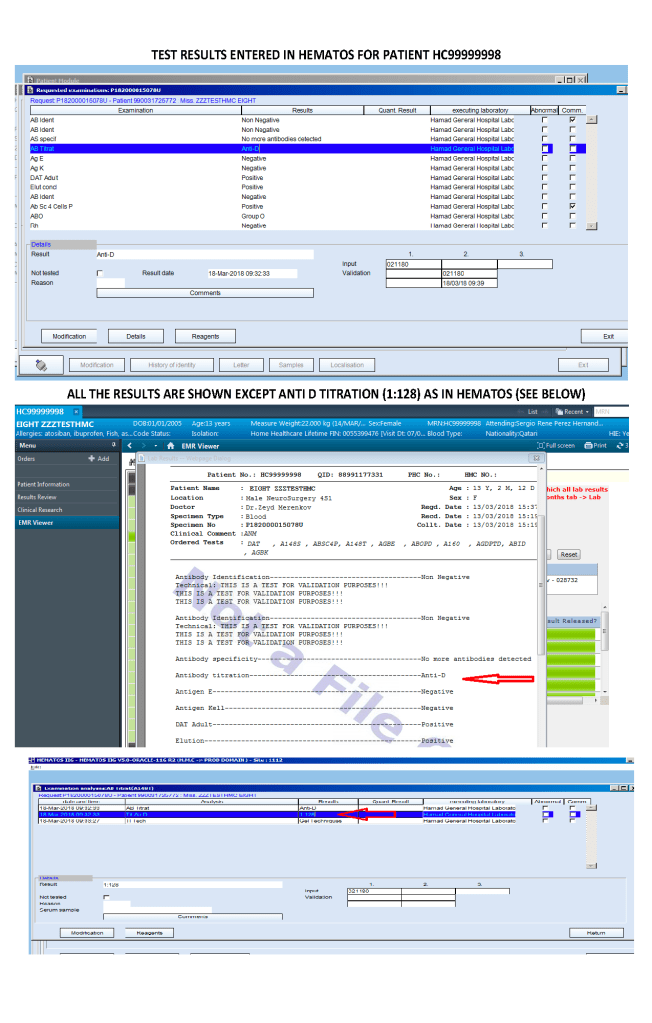
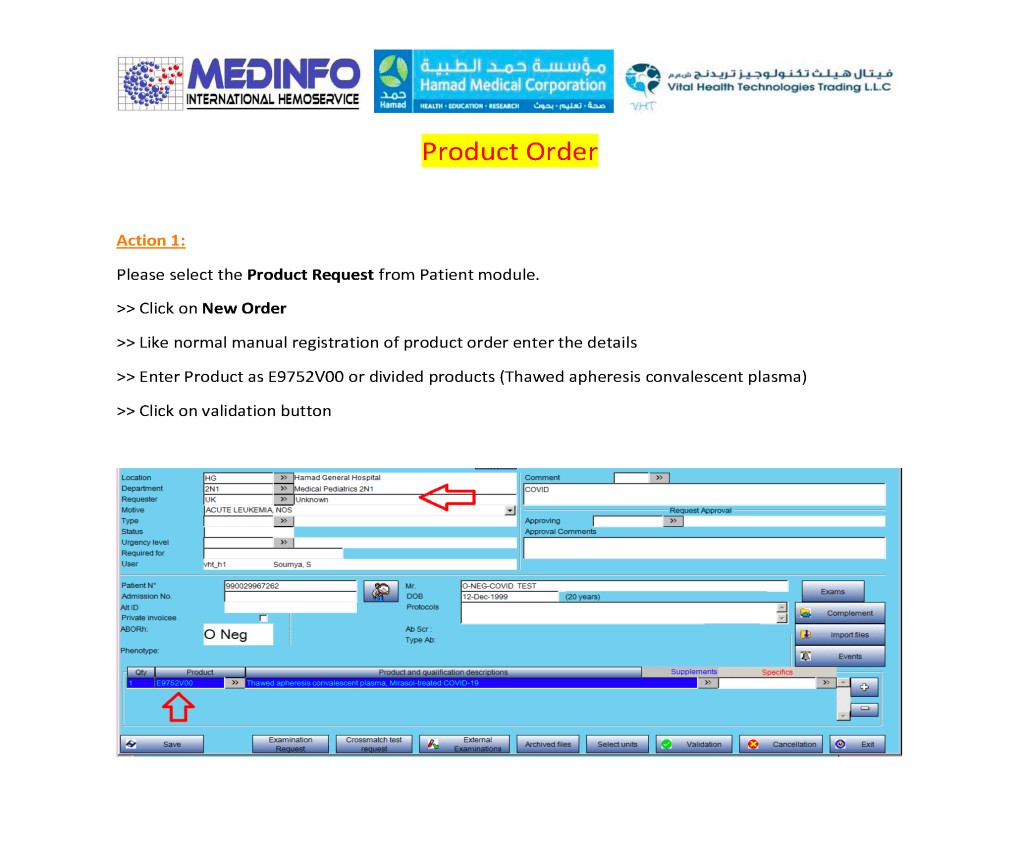
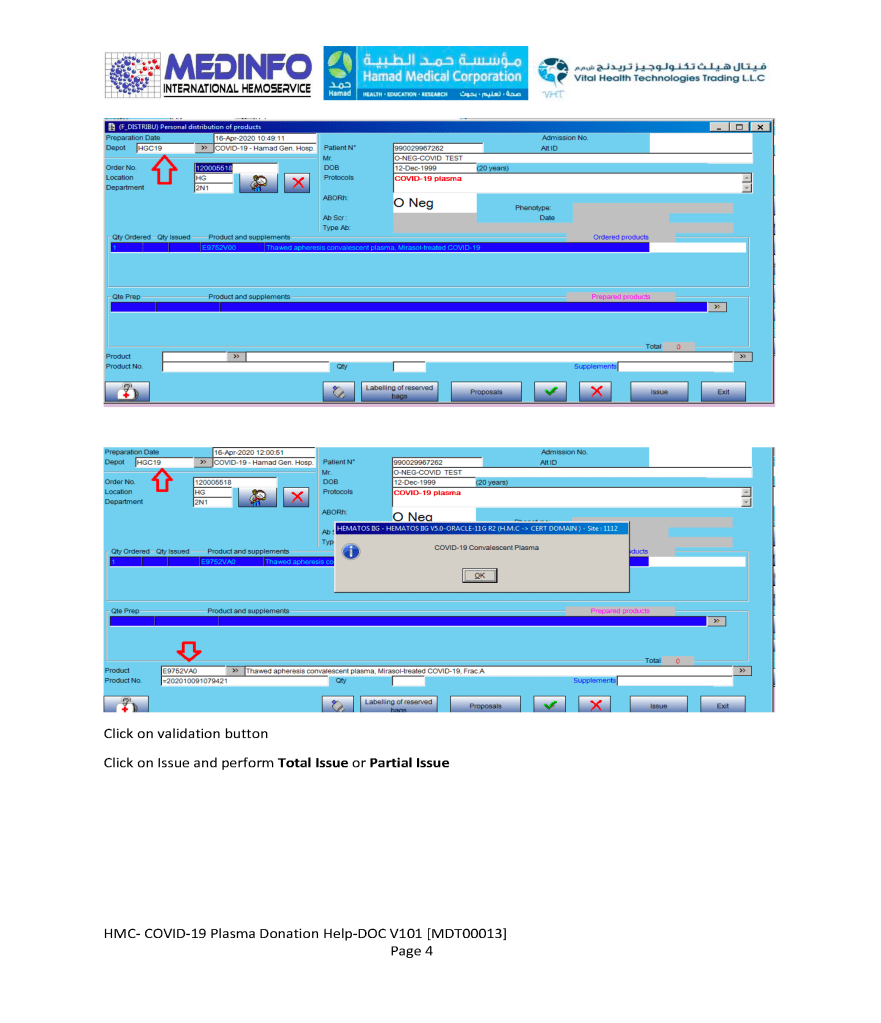
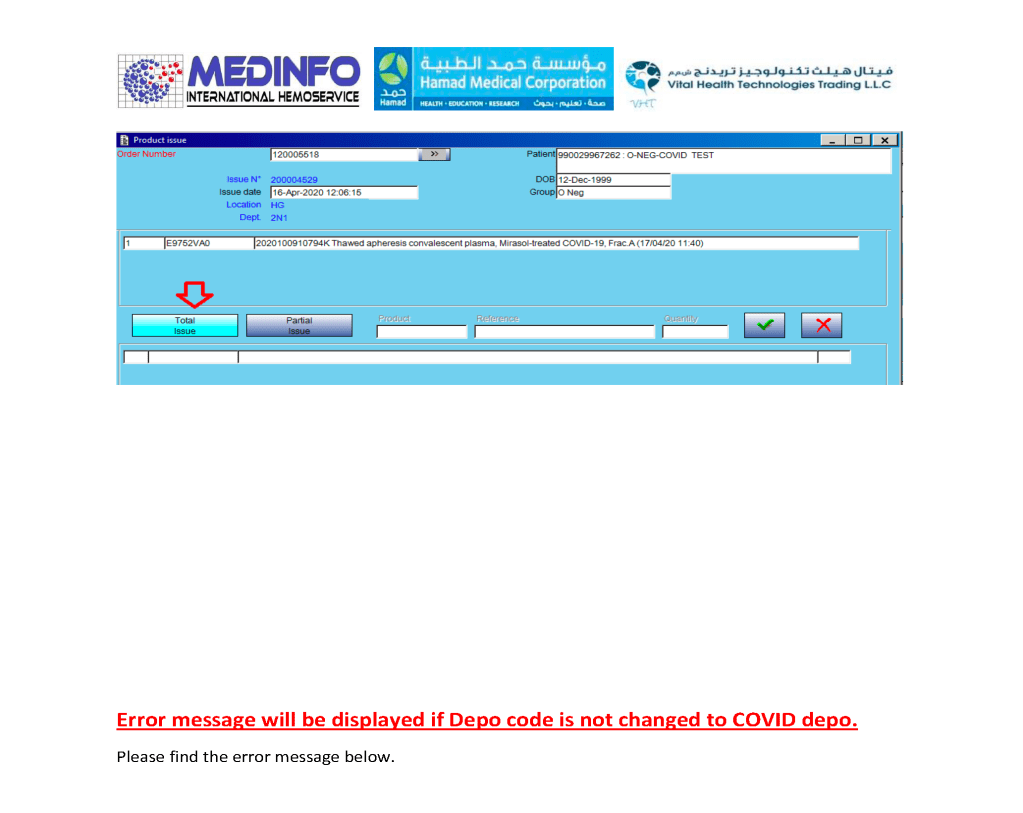
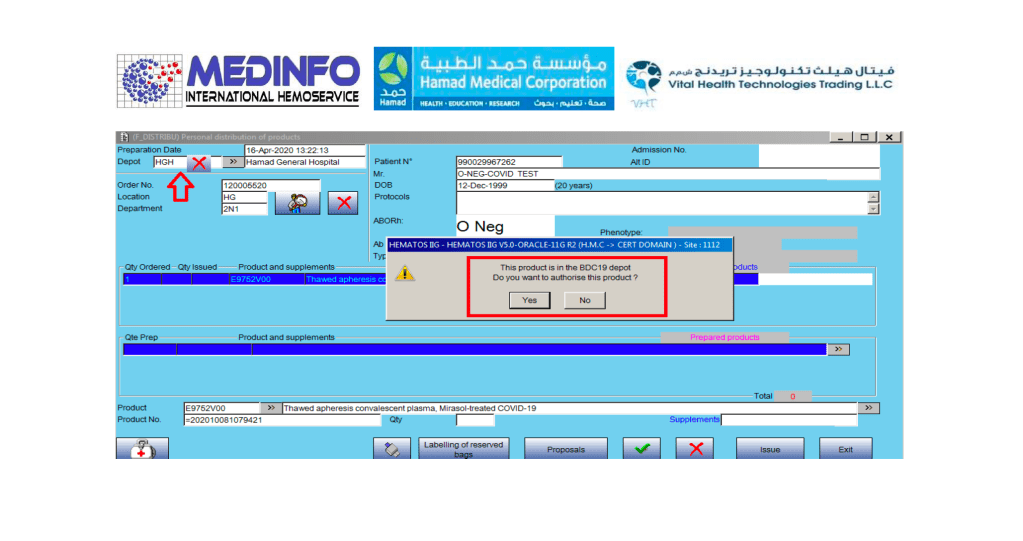
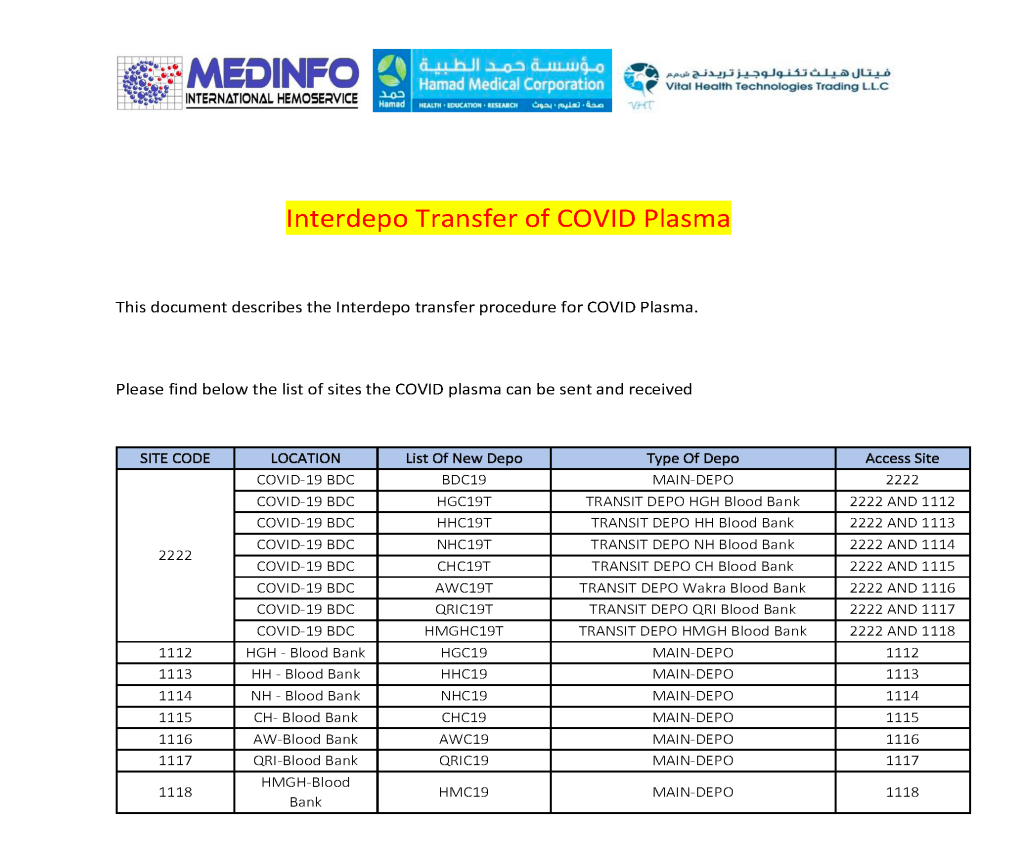
I designed my blood bank software Medinfo for use by my staff at all levels and positions to adhere to and facilitate compliance to the workflow processes. Blood bank staff were restricted to access only those functions needed for their job duties.
Blood Donation records could not be viewed by outside staff for confidentiality reasons. Blood donor records were not linked to patient records at all.
We did not allow access for Medinfo to non-Transfusion Medicine staff since the screens were designed to maximize efficiency of the work processes, not the viewing by outsiders of results. Outsider access was made through the hospital information system HIS.
The separate hospital information system HIS interfaced to Medinfo for the following functions:
- Ordering blood components
- Ordering a limited number of tests from which algorithms would be generated on the blood bank side for further testing
- Querying the status of the test or component orders (e.g. ordered, collected, in blood bank being processed, completed)
- Viewing of completed tests and component requests
Even within the HIS ordering capabilities, there were additional restrictions:
- Blood components: outside doctors could order base blood components could be ordered, but special processing such as washing or irradiation followed internal blood bank rules. Outside physicians could state their preferences in an order comment, but blood bank rules applied. Any disagreements had to be discussed with the transfusion medicine physician.
- Testing: Only base tests such as ABO/D typing, antibody screen, direct antiglobulin test, transfusion reaction workup, and cord blood testing could be directly ordered by outside physicians, but further testing depended on the results of these tests as allowed by internal blood bank algorithms. An outside physician could not directly order other tests but had to discuss his concerns with the transfusion medicine physician.
Results viewing in the HIS were subject to additional conditions as well:
Only certain results, not all results were viewable directed in the patient’s chart since showing all results may be confusing to the outside physicians and nurses. The selected results were sent back into the HIS for viewing. All these non-viewable results were retrievable for blood bank staff in Medinfo.
Another option, one I did not use at either HMC Doha or NGHA in Saudi Arabia, was to order tests and components by physicians directly into Medinfo. Likewise, they could view test results directly in the system. Special screens could be constructed to offer ordering and results retrieval.