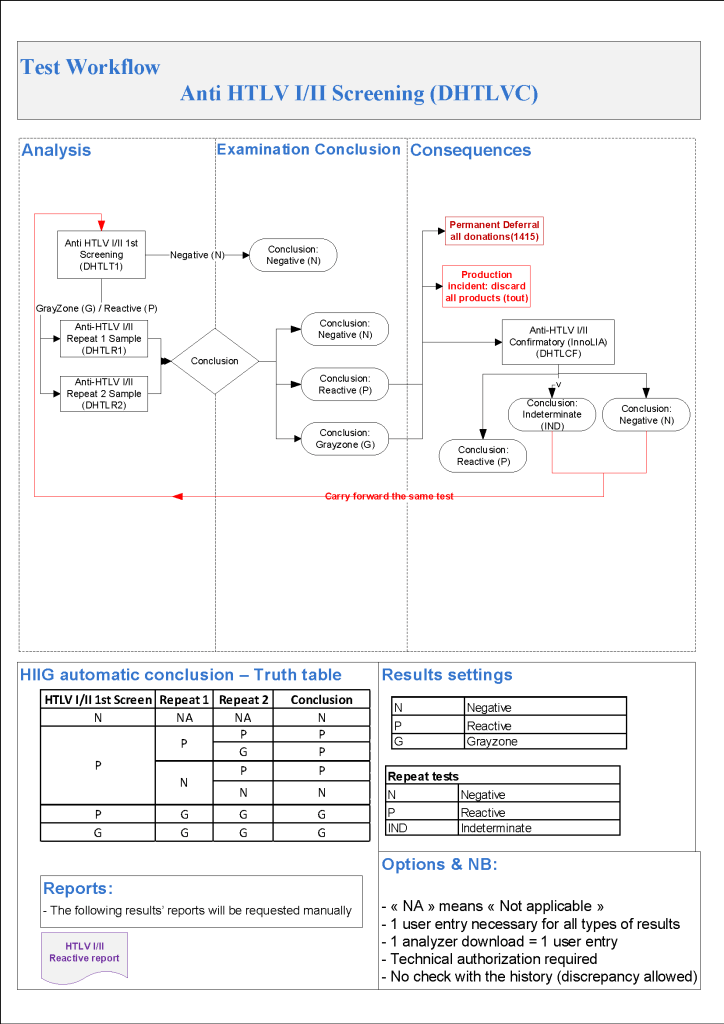
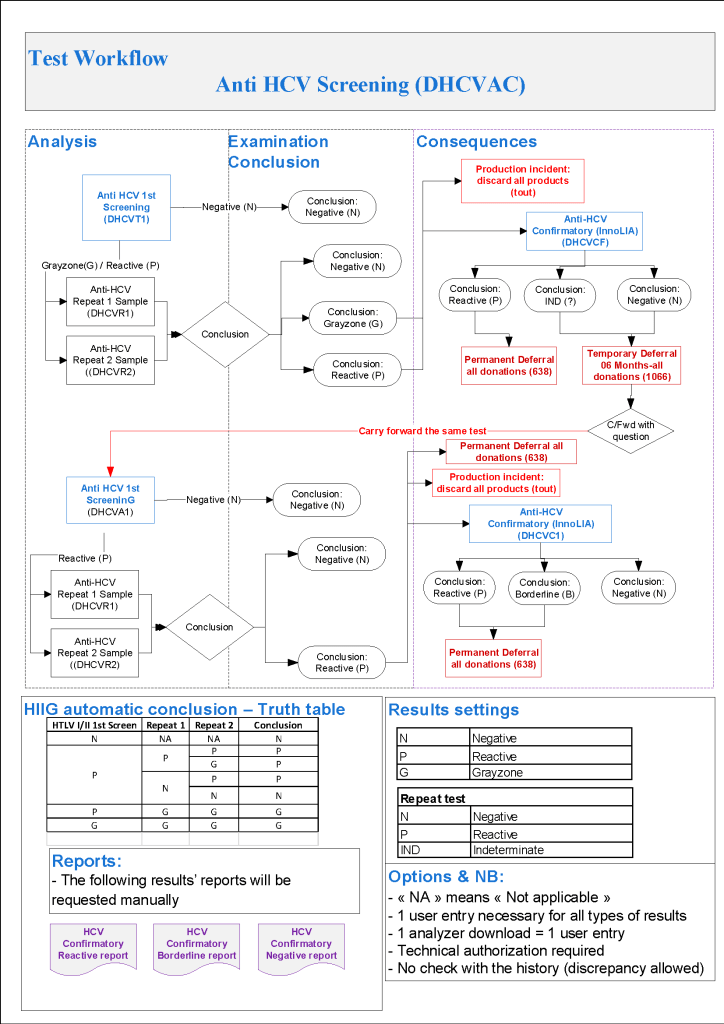
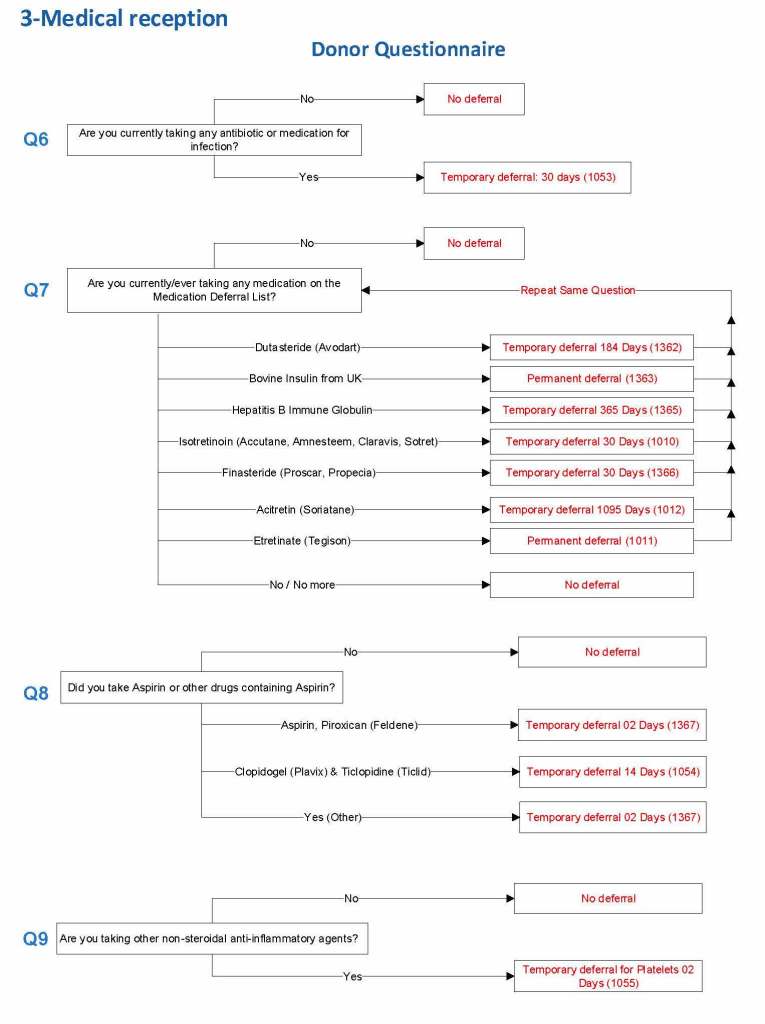
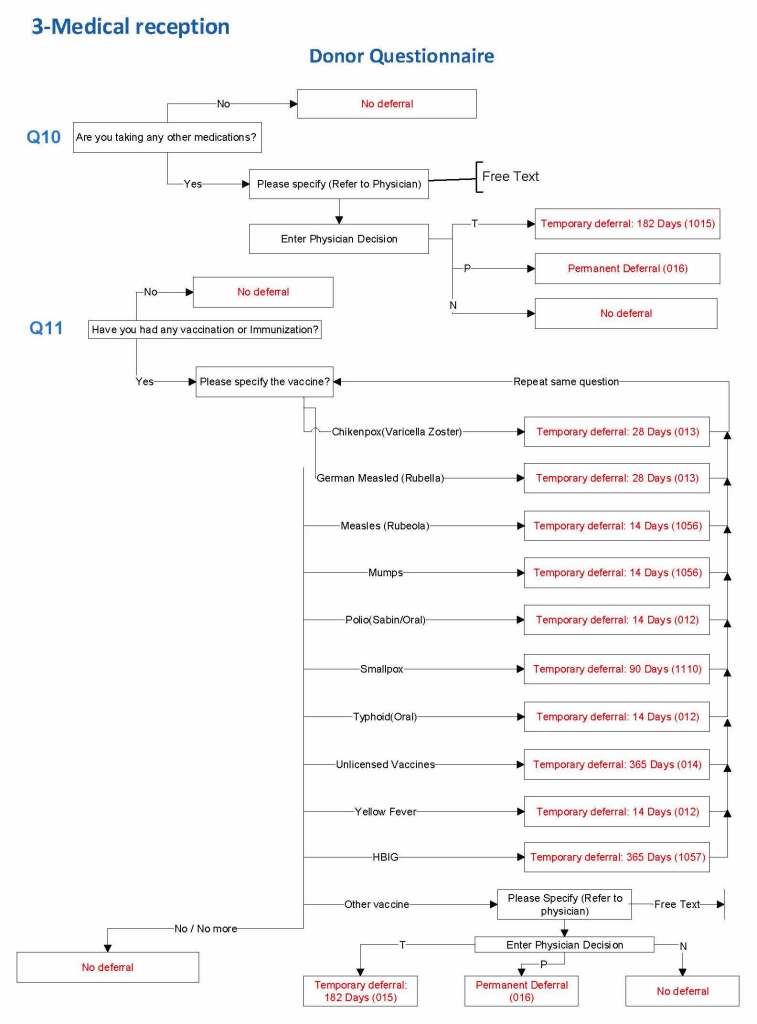
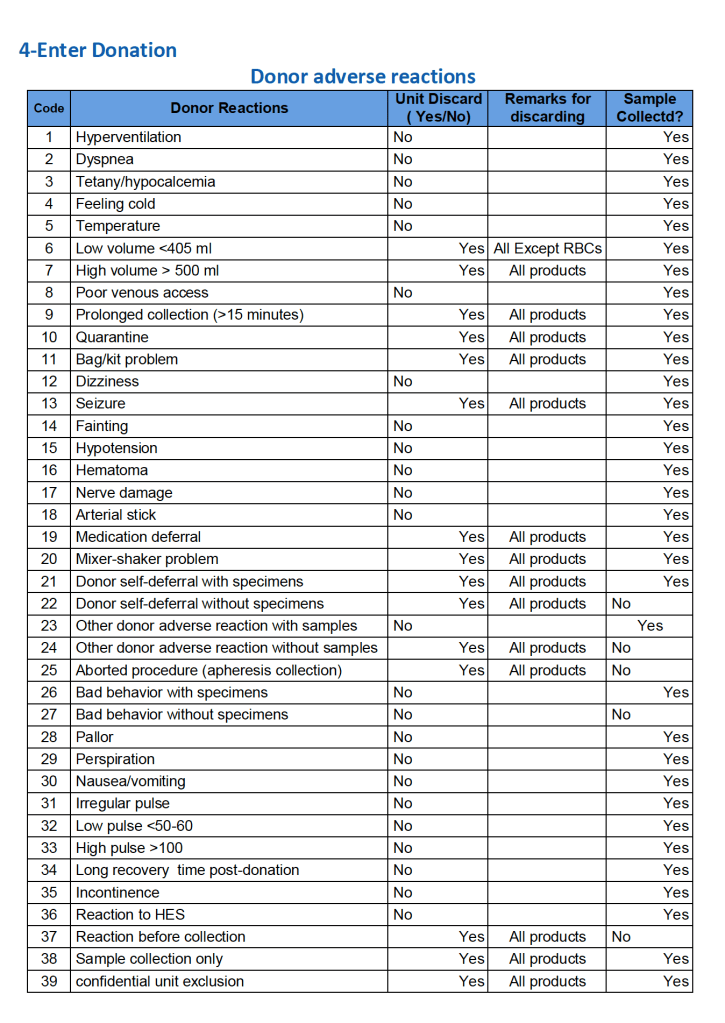
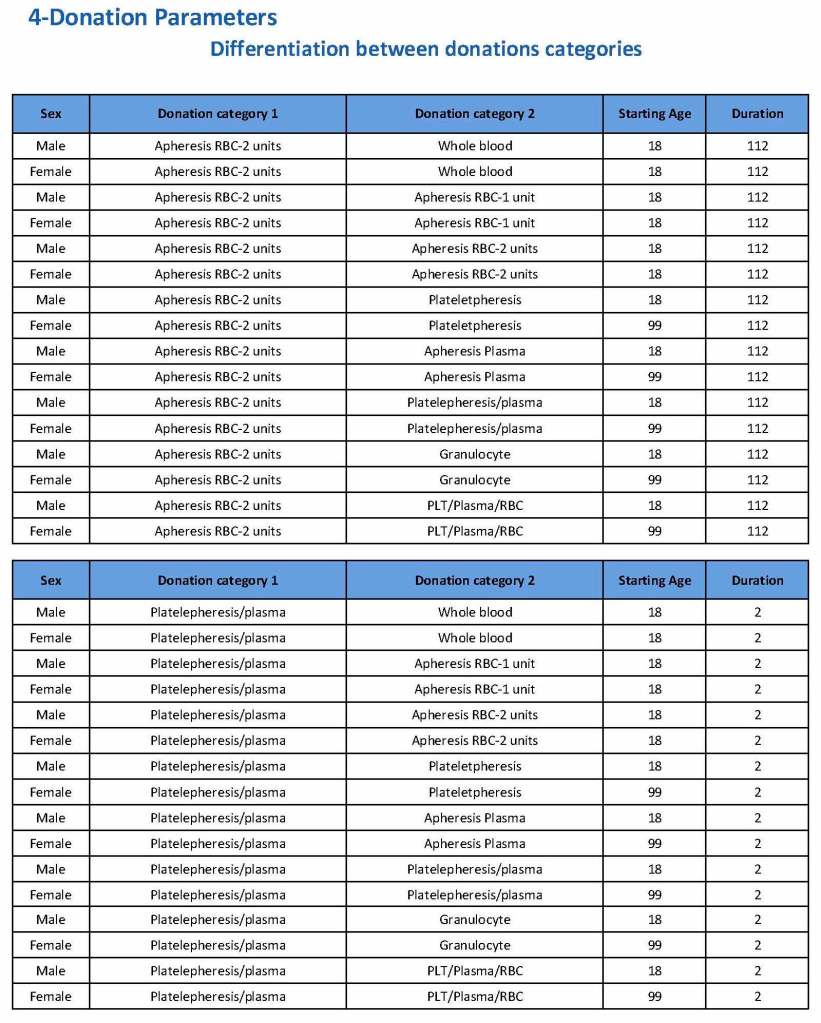
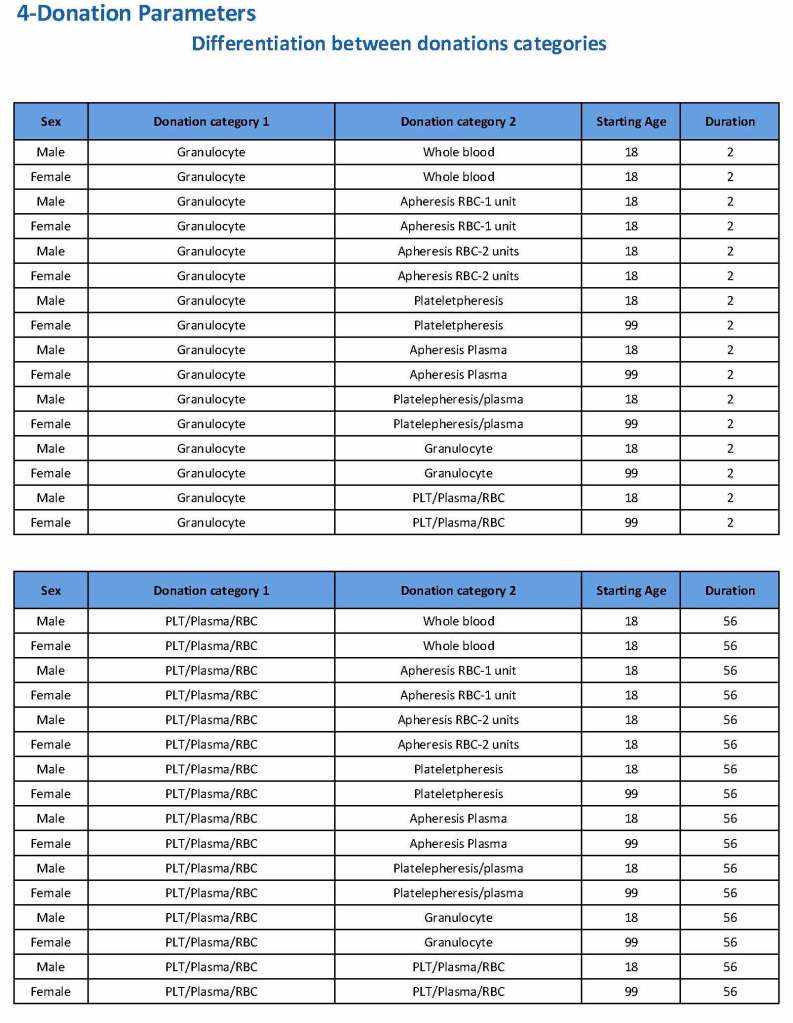
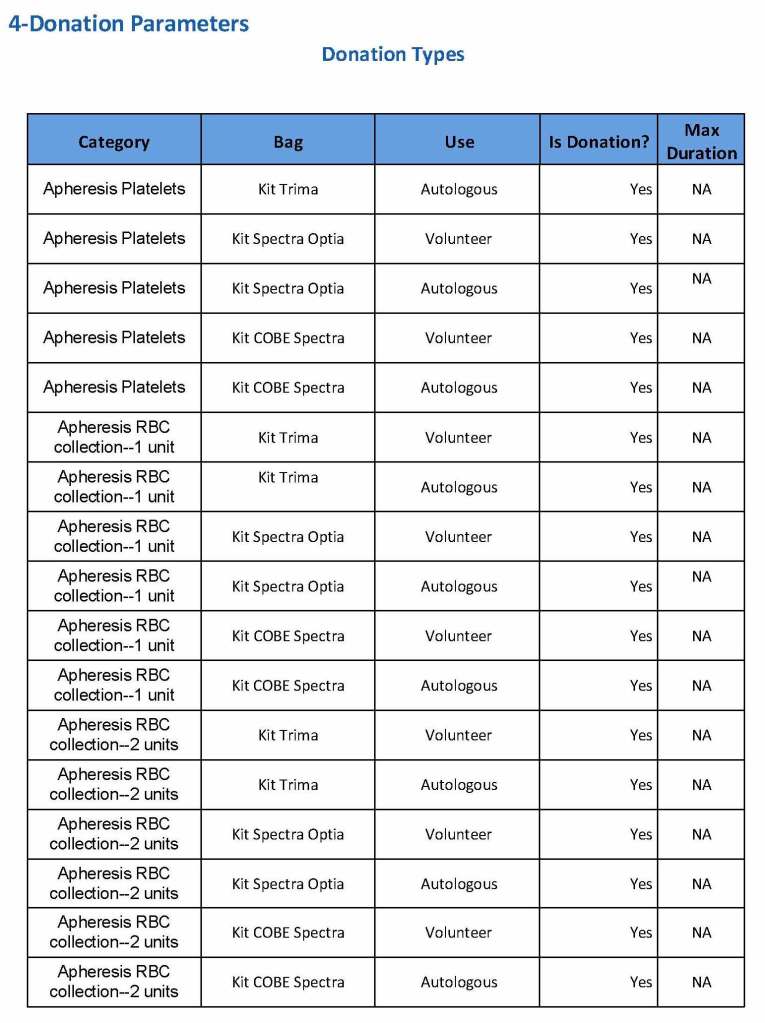
HMC Doha 2011-2020: Set-up of both donor and patient modules including inter-depot transfer:
Donor:
Implemented COVID-19 convalescent plasma CCP production at HMC Doha over two-week period, February-March, 2020 including full integration with software
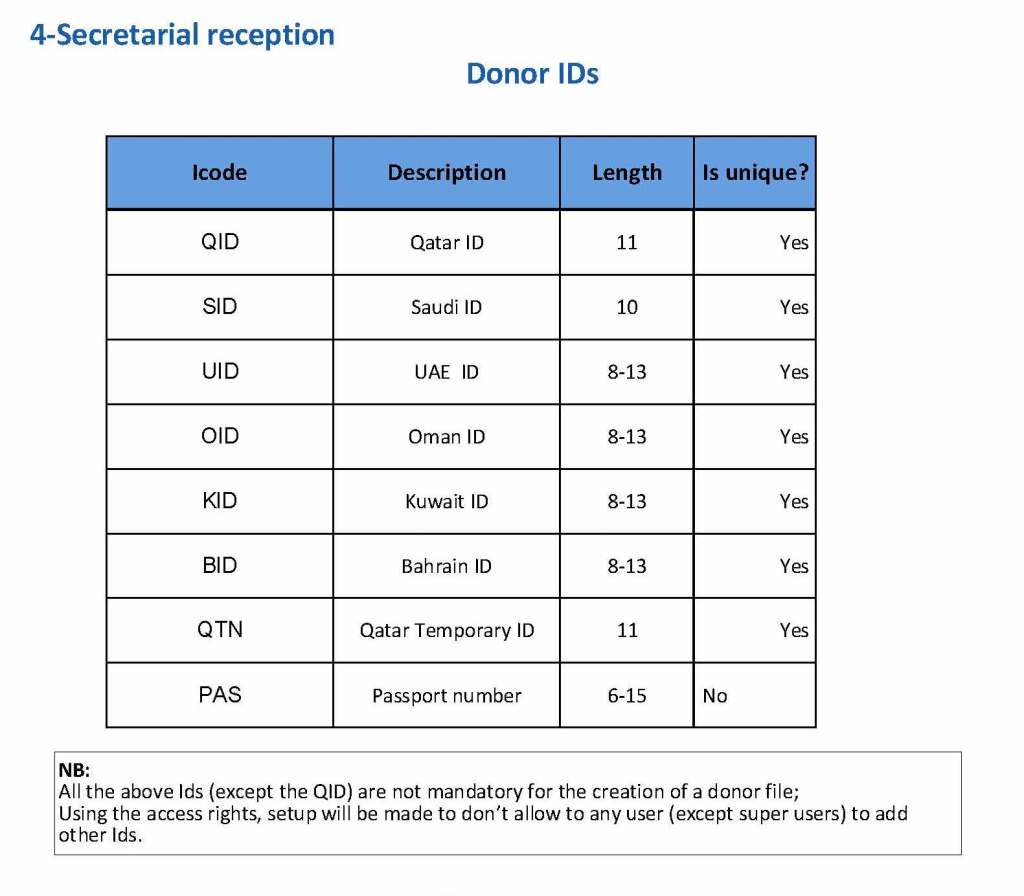
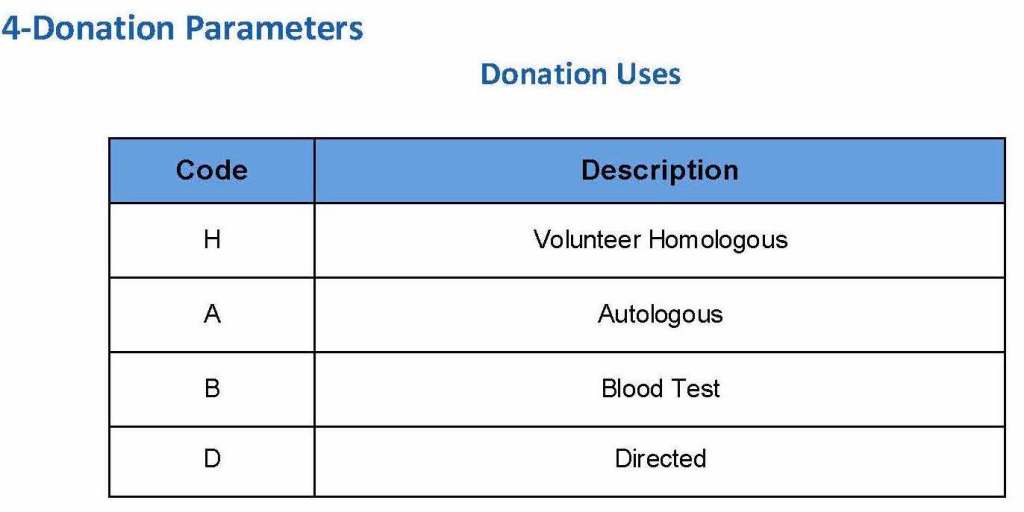
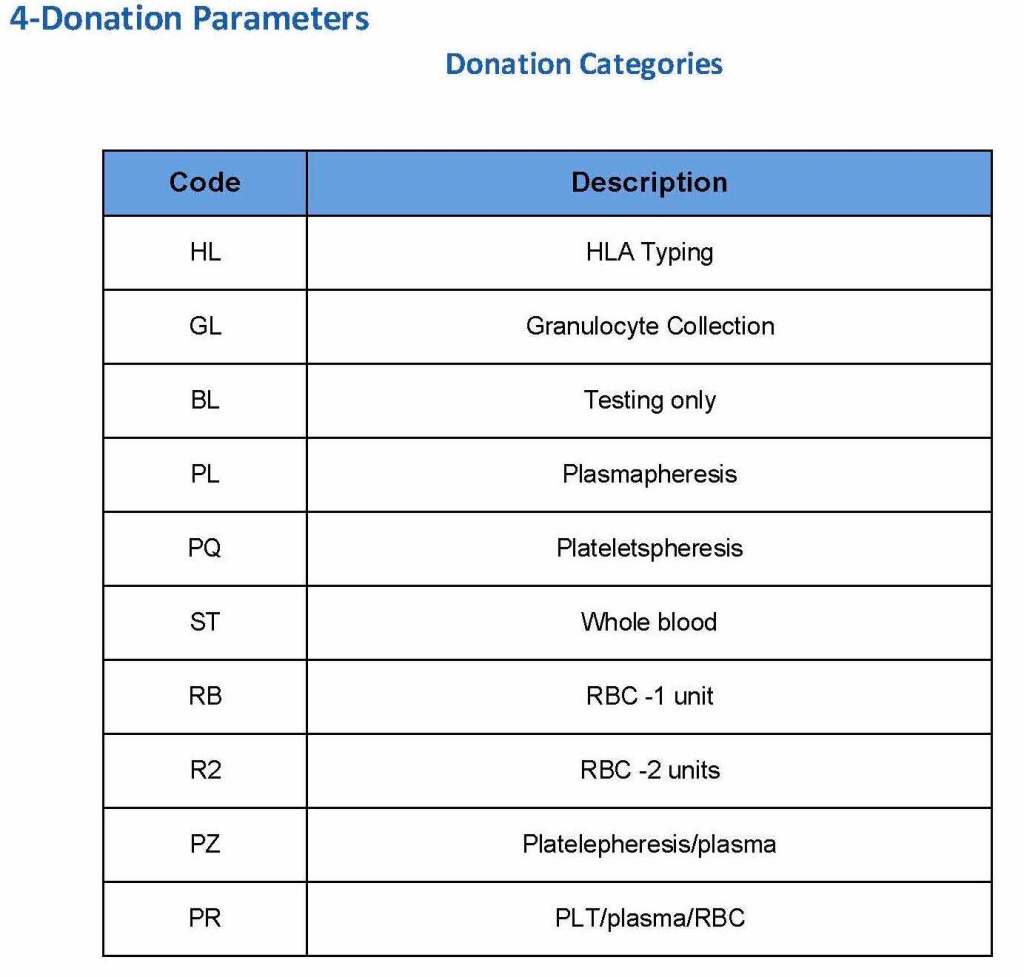
Donor collection, donor marker testing, donor immunohematology testing, inter-depot transfer between production site and hospitals and hospital-hospital transfers, ISBT labelling and specimen
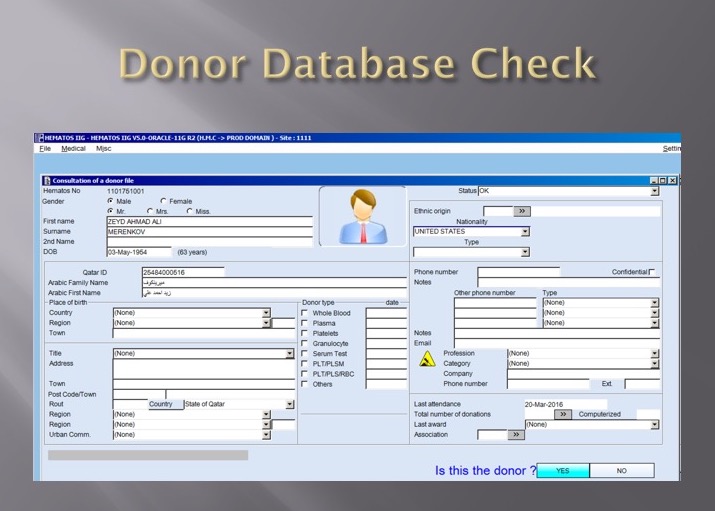
Collection Interface (read-only) with Qatar Ministry of Interior to obtain both English and Arabic demographic information from donors
Establishing world’s first interfaces (bidirectional as required) with Terumo BCT Atreus and Reveos automated blood processing equipment, Mirasol pathogen-inactivation/platelet additive solution, Terumo Trima Accel donor apheresis machine, Terumo mixer-shaker donor collection device
Patient:
Implemented a CCP quarantine patient blood bank separate from regular hospital blood banks) for thawing and releasing of CCP plasma
Patient module including all compatibility testing, ABO/D typing, extended antigen typings, direct and indirect antiglobulin testing (antibody screens), antibody identifications, eluate, component modifications (thawing, pooling, aliquoting), interfacing of Diamed automated gel testing
Establishing algorithms for emergency release, electronic crossmatching, automated patient specimen titration with Ortho Vision MAX, prophylactic antigen matching, ensuring irradiation of blood components or use of pathogen-inactivation as required, allocation rules by algorithm, required and optional antigen matching in presence of antibodies
Development of bidirectional interface between Medinfo patient module and Cerner Millennium laboratory module (permitting order of transfusion tests and blood component orders in Cerner, transmission to Medinfo patient module for testing and allocation of components, and then sending test results and component status back to Cerner)
Common:
Developing current and future states to develop the workflows to prepare software processes
Validation testing, initial/user acceptance testing, follow-up validations
NGHA Riyadh 2009-2010: Set up of both donor and patient modules including inter-depot transfer:
Donor:
Collection, marker testing, immunohematology testing, component production, inter-depot transfer between production site and hospitals and hospital-hospital transfers, ISBT labelling and specimens
Patient:
Patient module including all compatibility testing, ABO/D typing, extended antigen typings, direct and indirect antiglobulin testing (antibody screens), antibody identifications, eluate, component modifications (thawing, pooling, aliquoting), interfacing of Diamed automated gel testing
Please refer to my website https://drzeydbloodbank.com for specific posts on Medinfo software process building. On the right hand side from the bottom TAGS menu, pick Medinfo Hematos IIG to see those articles.