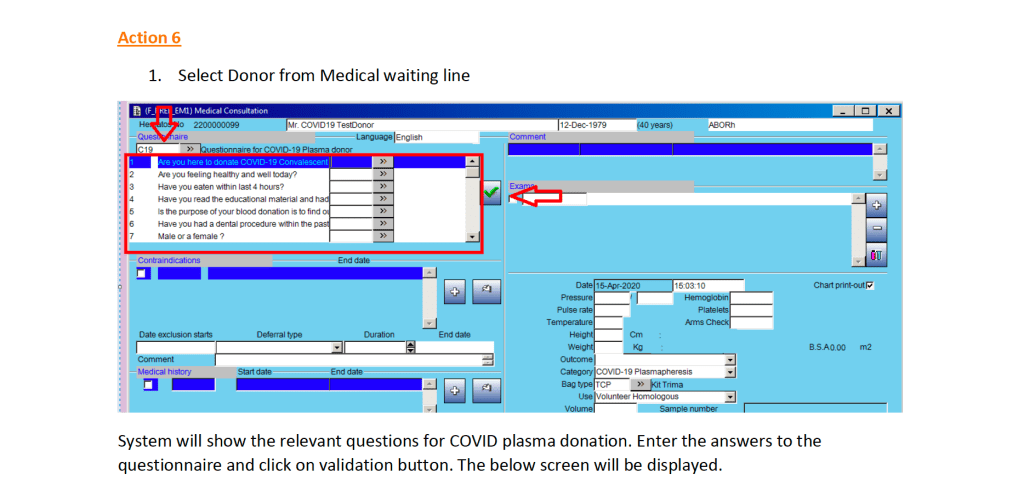
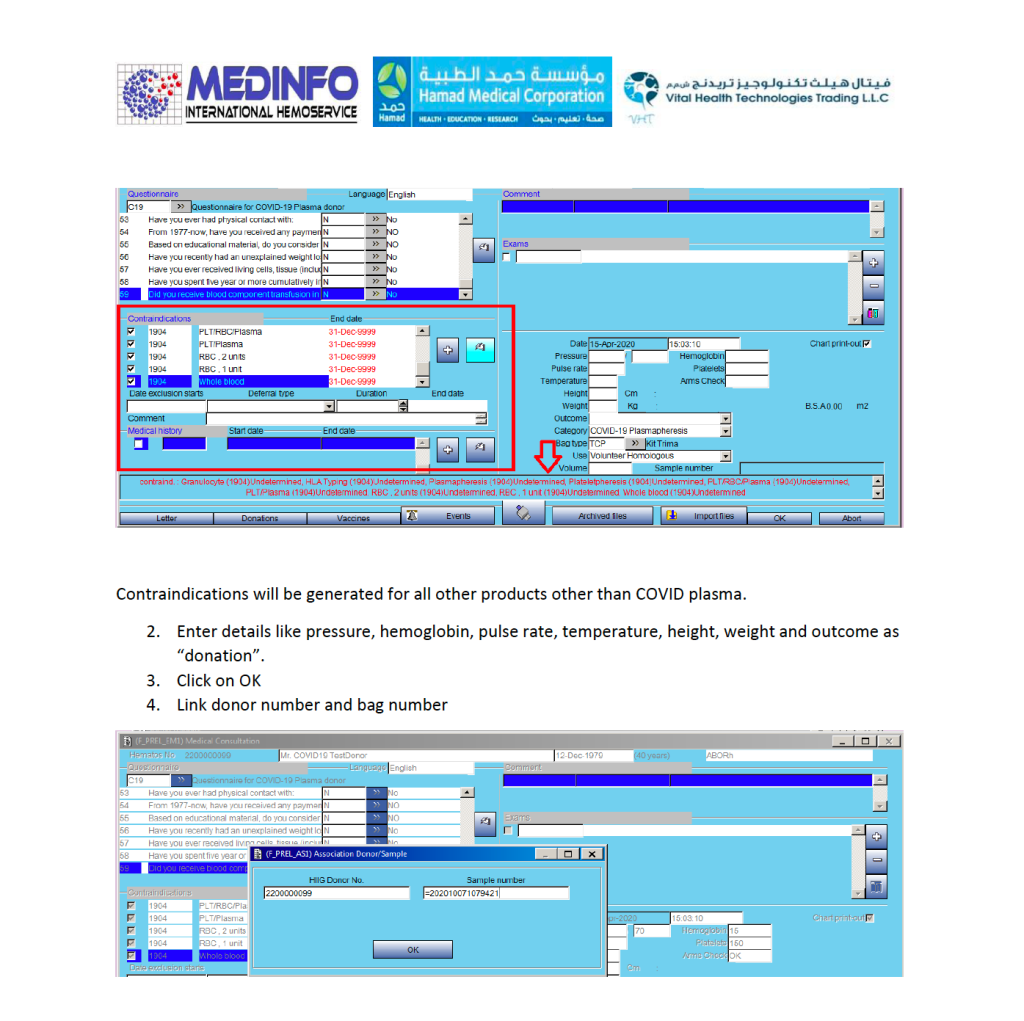
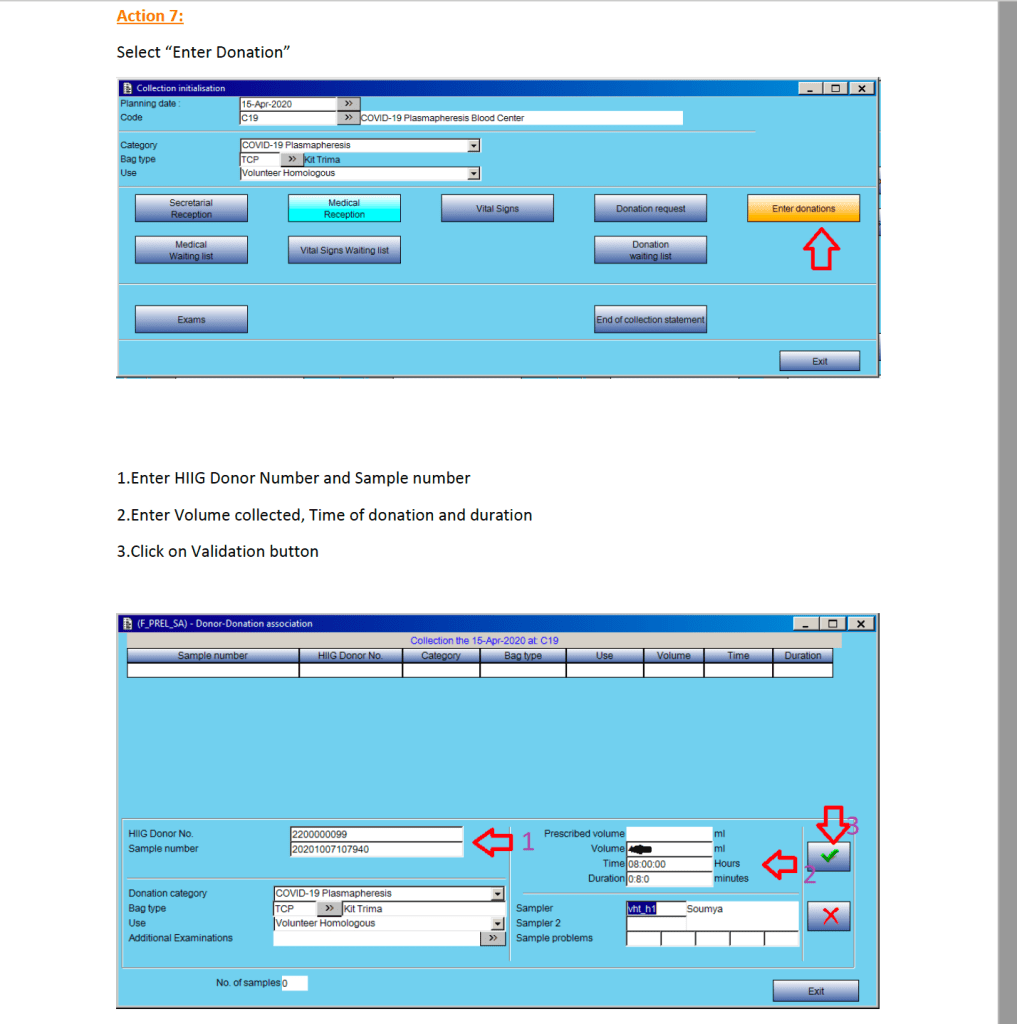
This is a teaching document for medical technology and transfusion fellows to explain the general structure of a validation.
Principle:
All validations must be planned. A validation protocol must be prepared with specific criteria for acceptance. All validations with attached evidence must approved by the Head, Transfusion Medicine.
Policy:
- A written validation protocol must be prepared in the advance and at least including the following:
- Specific parameters and number of iterations to be performed
- Designated staff to perform validation
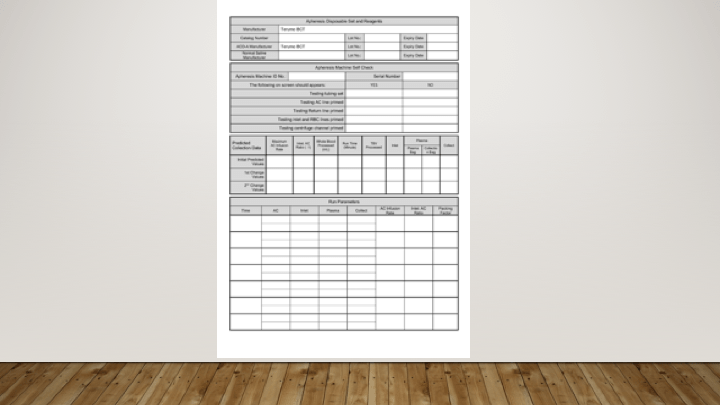
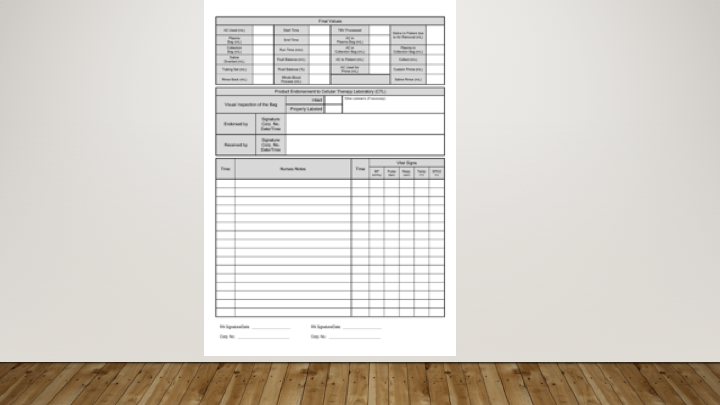
- Documentary evidence of the testing
- Specific acceptability criteria
- The completed validation protocol must be submitted to the Division Head, Transfusion Medicine, or designee for review.
- Once the validation plan has been reviewed, it must be performed by the designated staff.
- Software validations will be performed in a specific test environment, not in the live, production system.
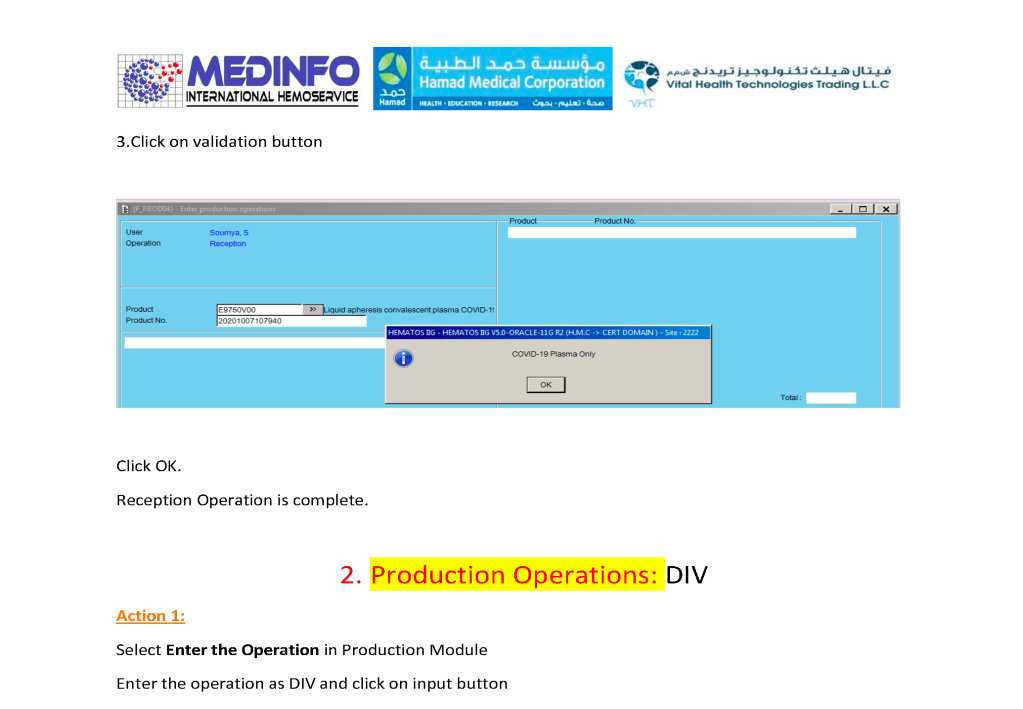
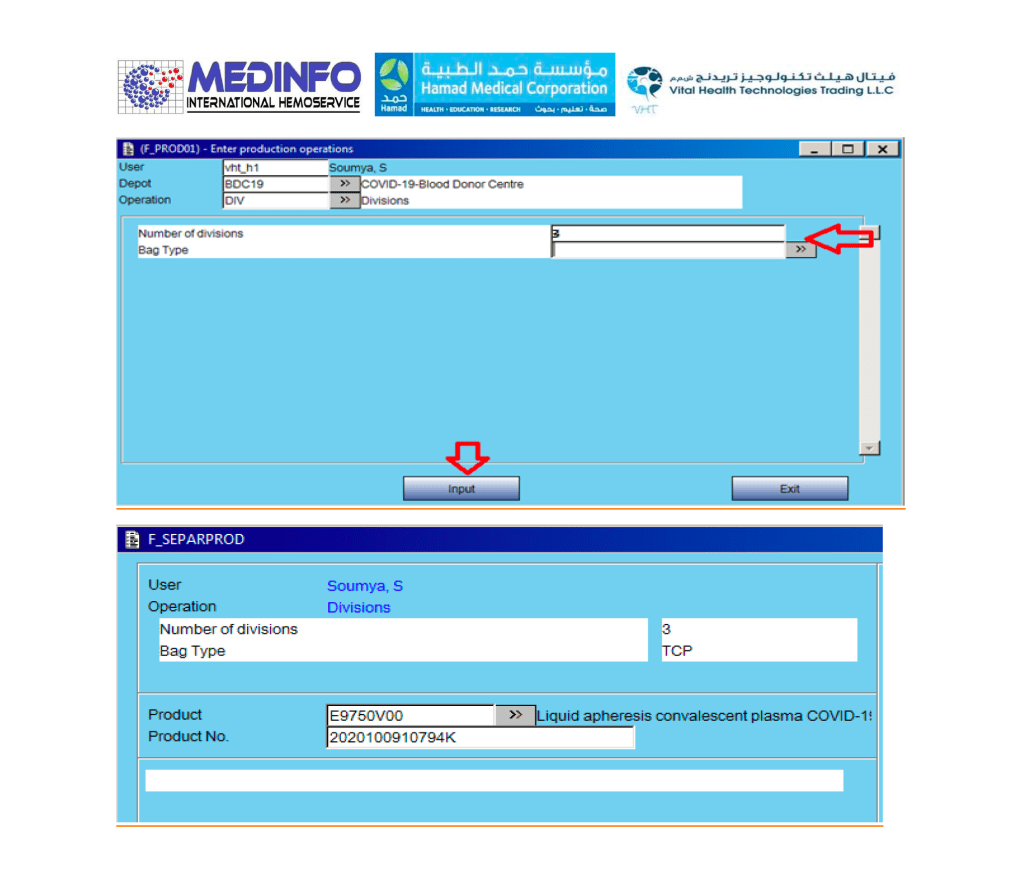
- The completed validation document, including screenshots of the software functionality if applicable, must be submitted to the Division Head, Transfusion Medicine for review.
- The equipment or software may only be used if the acceptability are met AND the validation is approved by the Division Head, Transfusion Medicine or designee.
- The completed validation protocol will be stored in the document control system.
Reference:
Standards for Blood Banks and Transfusion Services, Current Edition, Bethesda, MD, USA