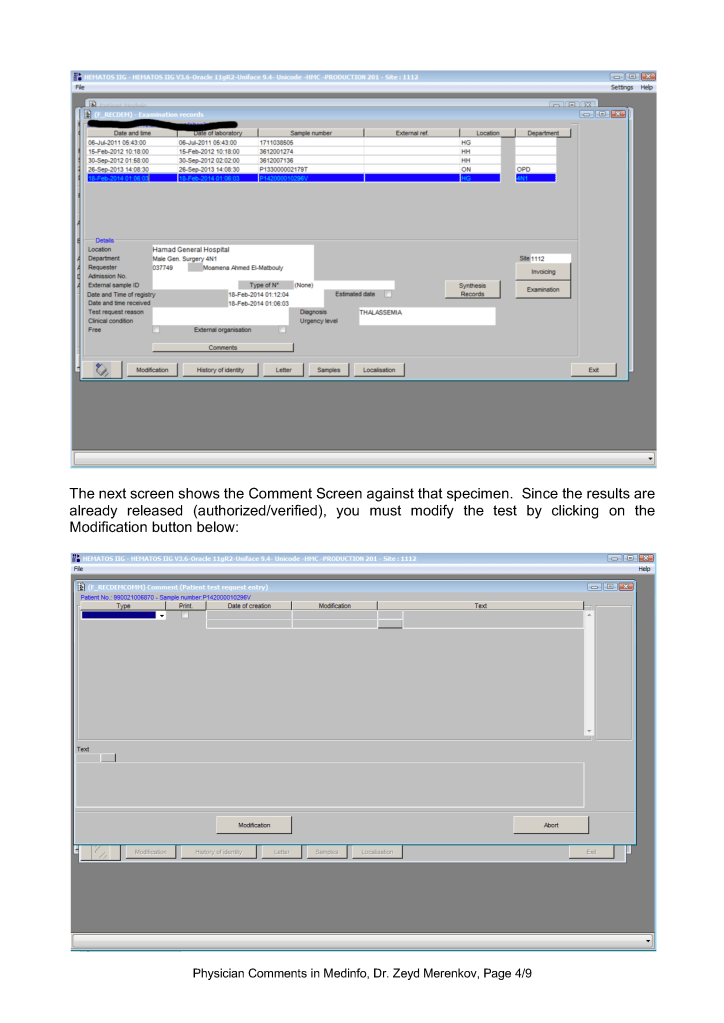
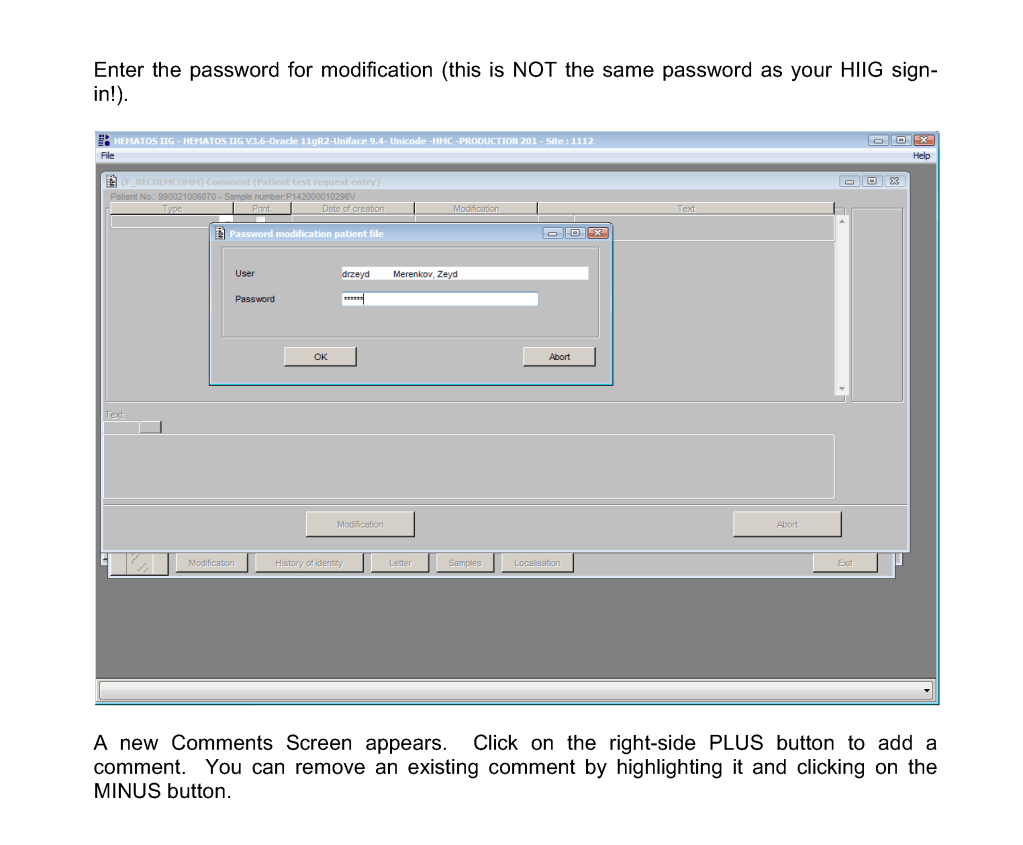
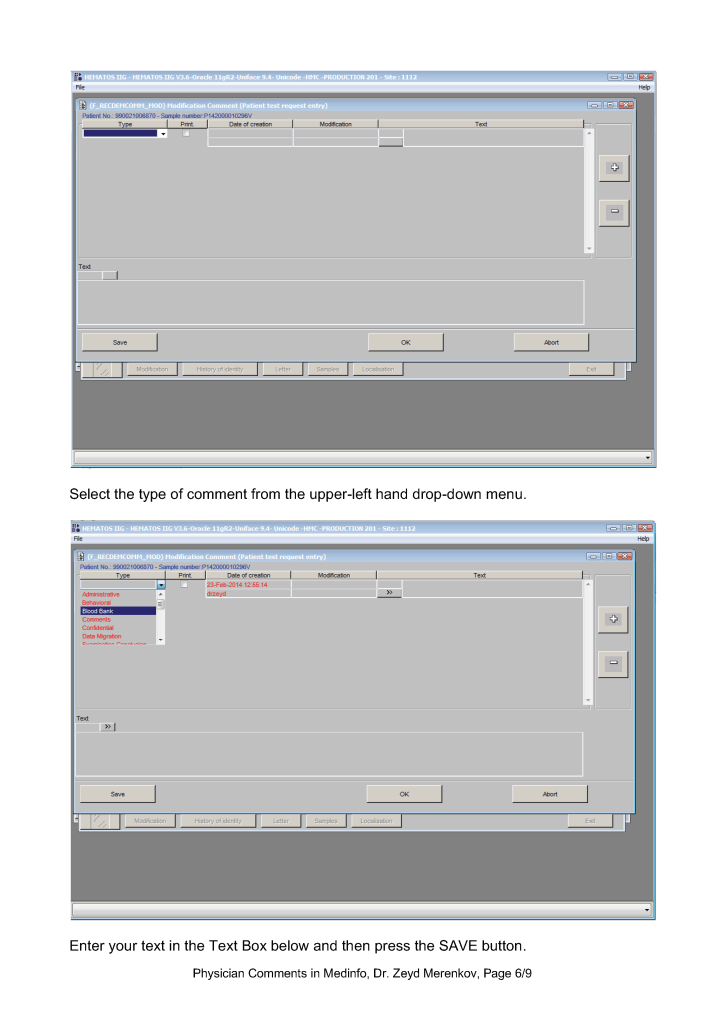
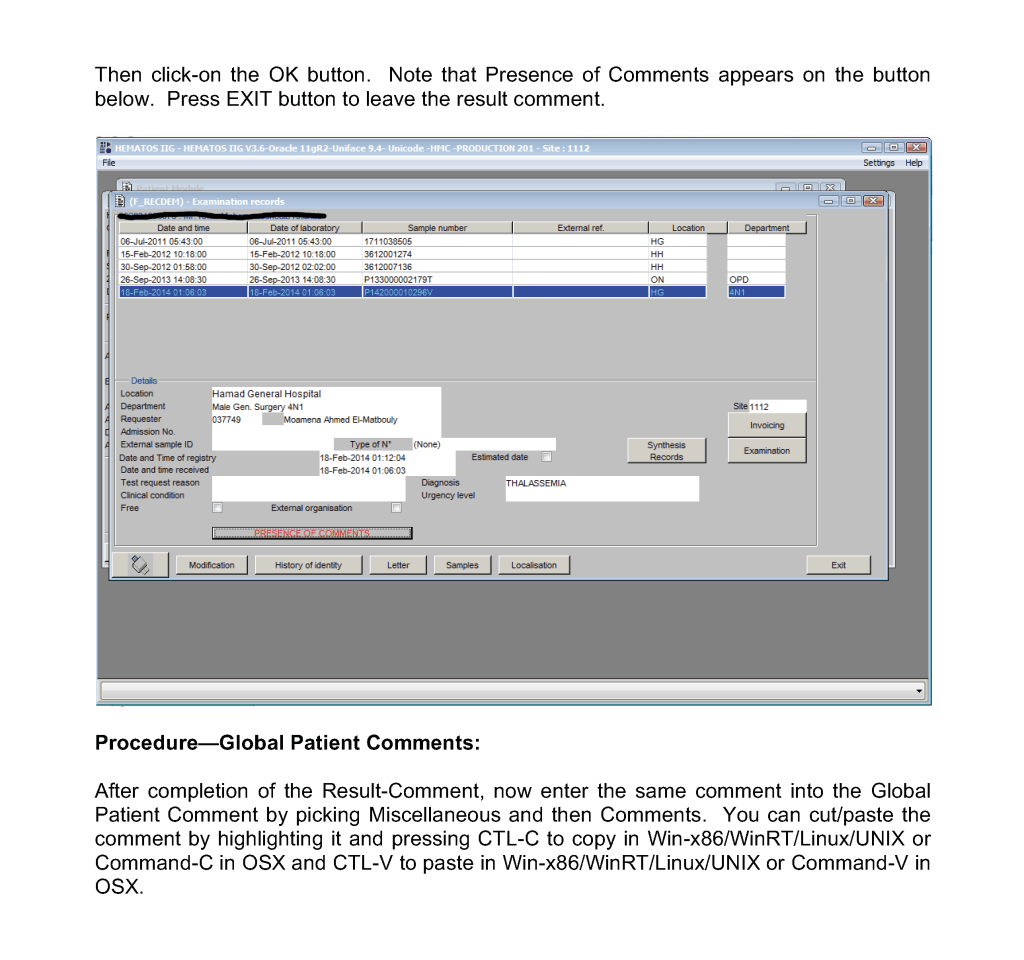
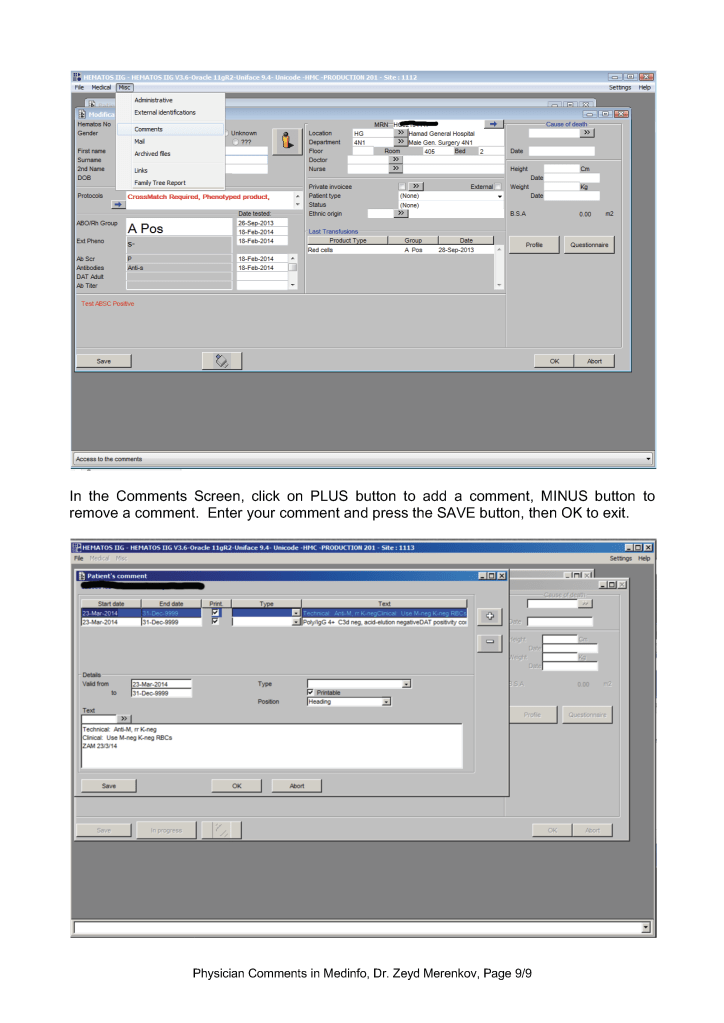
This is a teaching document of a process to irradiate RBC components when our irradiators are not functioning. In our system, we used Mirasol pathogen-inactivation so our RBC units were affected. The number of units for advance irradiation was based on our historical usage of irradiated units across our system. The workweek is Sunday-Thursday so on Thursday special effort was made to have the minimum number of irradiated units available.
We used irradiated units for compatibility testing to avoid the possibility that the unit would be released without irradiation.
Emergency Interim Procedure:
- Verify the patient’s diagnosis/location: All Hematology-Oncology patients should receive irradiated blood. Refer to the list of diagnoses for which irradiation is indicated (attached).
- Only PRBCs need to be irradiated. Mirasol-treated (pathogen-inactivated) platelets can be used directly without irradiation in accordance to Council of Europe CE regulations.
- Attach radiation indicator labels to the selected units as per the irradiation procedure.
- Send the units for irradiation to the Radiation Oncology department.
- Verify that the proper dose of irradiation was received while in the Radiation-Oncology department.
- Keep a minimum stock of 20 group O-positive units irradiated at the start of each day AND before the start of the weekend on Thursday afternoon. Irradiate that number of group O-positive units plus any other specific requests for blood.
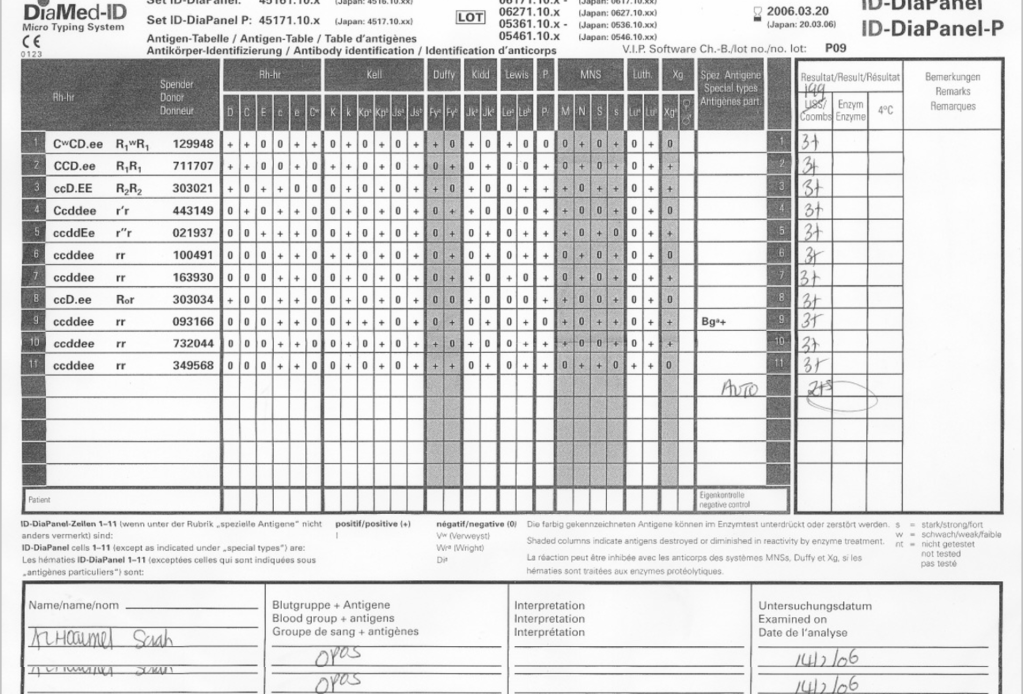
- You should use the irradiated units for crossmatching. DO NOT CROSSMATCH FIRST BEFORE IRRADIATING!! If the antibody screen is positive, refer the specimen to the Transfusion Service for further processing and selection of units.
- Note: If a special antigen typing is needed, we may have to irradiate after selecting the antigen-matched unit if it is not found in the group of irradiated units.
- If a unit is required for a child < 20 kg, it must be washed before release if the unit was irradiated more than 24 hours previously according to our irradiation policy.