This is part 3 of a medical student lecture I gave at National Guard Health Affairs.













Immunohematology testing and processes
This is part 3 of a medical student lecture I gave at National Guard Health Affairs.













This is the second part of a lecture I gave to medical students at National Guard Health Affairs in Riyadh.







This is an updated version of a medical student lecture I gave at National Guard Health Affairs in Riyadh.











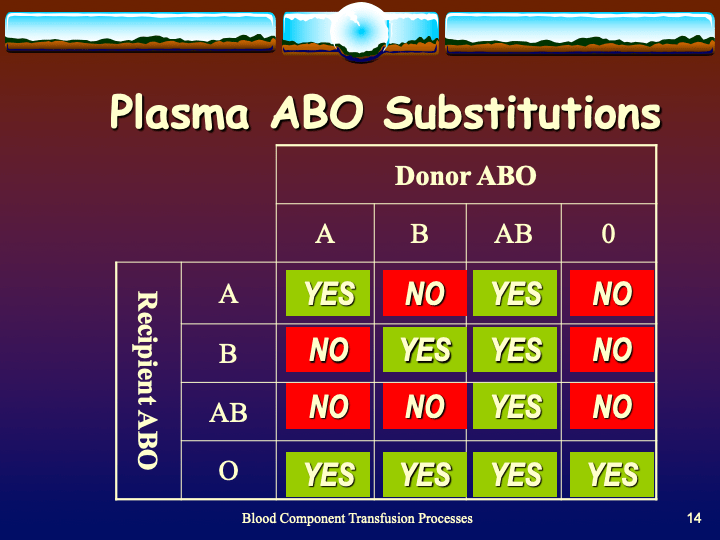
To be continued
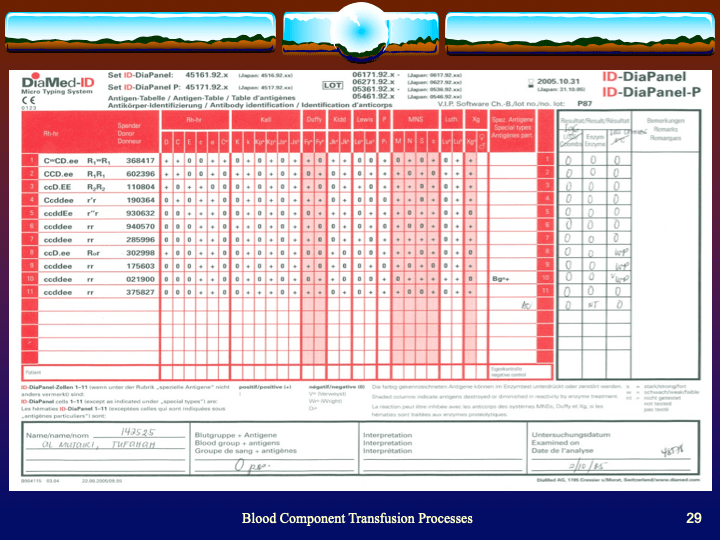
I am a strong believer in performing both AHG and enzyme panels together in routine antibody workups. I especially feel this is important when the patient is R1R1 since I always want to rule out anti-c. Sometimes, anti-c is only identified in the enzyme phase.
This is a case from my files of an R1R1 patient with the following results:
Antibody Screen:

AHG Panel:
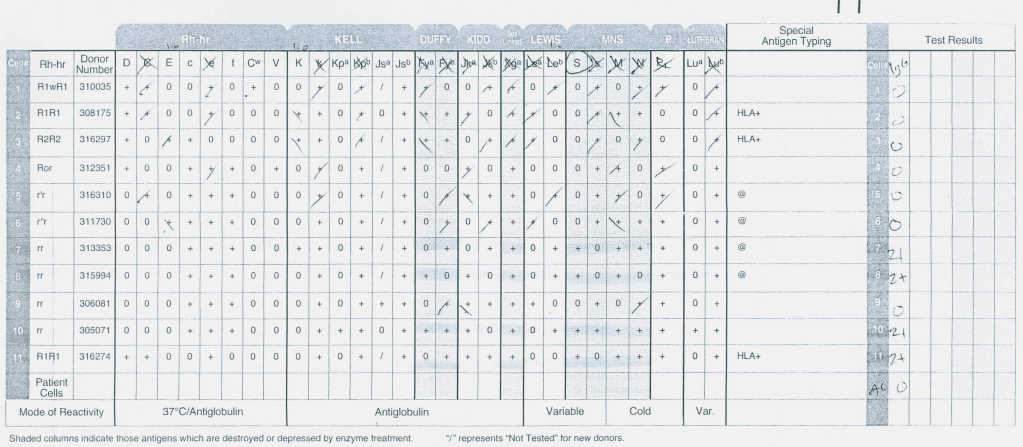
Enzyme Panel (Ficin):
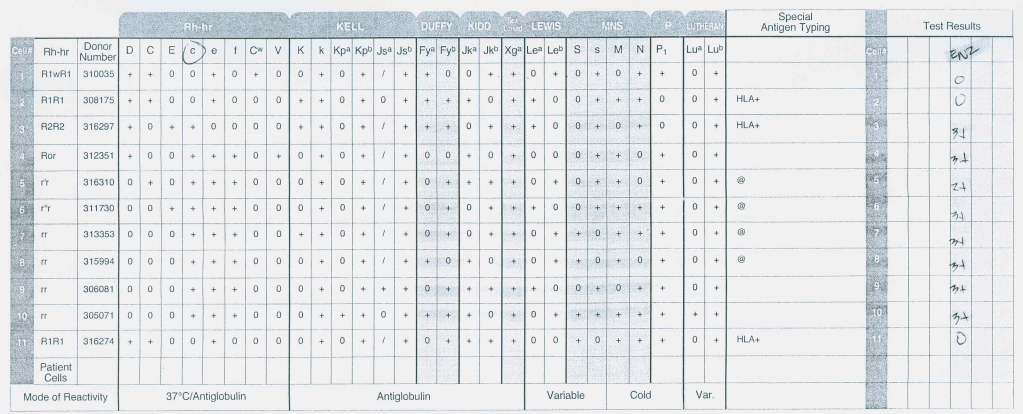
This anti-S is enzyme-labile but anti-c is revealed, only reacting at enzyme phase. The patient was Kell-negative so I selected S-negative, R1R1 K-negative RBCs for transfusion. Anti-c can be a dangerous antibody causing severe hemolytic disease of the fetus/newborn and sever hemolytic transfusion reaction. If only the AHG panel had been performed, the anti-c would have been missed.
Nowadays, if we have a multiply transfused patient with a complex antibody pattern, we might resort to RBC genotyping to help us resolve the antibody issues. Fortunately, there is one situation where we can quickly phenotype the patient by using hypotonic saline to lyse the transfused RBCs since the sickle cells are resistant.
The results can be quite clean and easy to interpret as in the following example using 0.4% saline:
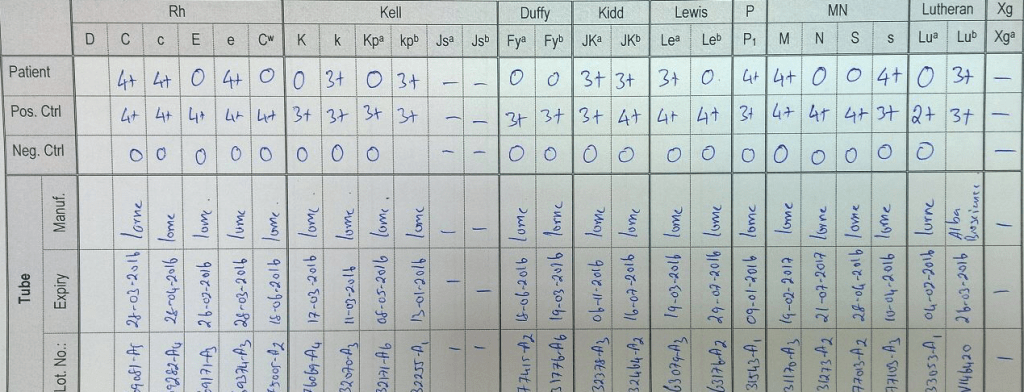
It is a lot cheaper to make dilute saline than an RBC genotype—and much quicker!
This was a teaching document for medical students and residents I made for NGHA Riyadh. I have updated it for leukodepleted components and platelet additive solution.
Immediate Steps for All Reactions:
After Transfusion is Terminated (except mild allergic, see below):
| Reaction Type | Signs and Symptoms | Etiology | Clinical Action |
| Allergic (mild) | Pruritus, urticaria (hives) | Antibodies to plasma proteins | Steps 1-3 above; administer antihistamines (PO, IM, or IV); resume transfusion if improved; if no improvement in 30 minutes treat as below. |
| Allergic (moderate to severe) | Hives, dyspnea, abdominal pain, hypotension, nausea, anaphylaxis | Antibodies to plasma proteins, including IgA (patient has anti-IgA antibodies) | Steps 1-5 above; administer antihistamines, epinephrine, vasopressors, and corticosteroids as needed; avoid future reactions by premedication and consider use of washed red cells if refractory. |
| Febrile (mild to moderate) | Fever, chills, rigors, anxiety, mild dyspnea | Antibodies to leukocyte antigens, (mostly HLA): cytokines | Steps 1-5 above; mild—administer antipyretics as needed; avoid future reactions by premedication and use of leukodepleted red cells and platelets |
| Acute lung injury | Fever, chills, dyspnea, respiratory failure | Antibodies form donor plasma to recipient WBCs; less commonly recipient antibodies to donor WBCs | Supportive therapy for respiratory failure, oxygen, mechanical ventilation, leukodepleted blood components, consider use of solvent detergent plasma, minimize plasma transfusions (use platelets in additive solution and leukodepleted RBCs in additive solution). |
| Acute hemolytic | Anxiety, chest pain, flank pain, dyspnea, chills, fever, shock, unexplained bleeding, hemoglobinemia/ hemoglobinuria, cardiac arrest | Hemolytic transfusion reaction; usually due to ABO incompatibility | Steps 1-5 above; treat shock with vasopressors, IV fluids, corticosteroids as needed; maintain airway; increase renal blood flow (IV fluids; furosemide); maintain a brisk diuresis; monitor renal status for acute renal failure. Monitor coagulation status for DIC; administer blood components as needed after etiology is clear. |
| Septic / toxic | Chills, fever, hypotension | Bacteria in contaminated blood | Steps 1-5 above; treat shock with vasopressors, IV fluids, culture patient and blood bag,antibiotics. |
This is an updated version of a lecture I gave to medical students at National Guard Health Affairs in Riyadh. I have included new blood component types such as universal low-titer group O whole blood, universal low-titer group A plasma, and refrigerated platelets.













This is a revised version of a previous post.
Principle:
Daratumumab is a monoclonal antibody that binds to CD38 antigen, which is expressed weakly on the surface of all RBCs. It may thus cause a positive direct antiglobulin test DAT and so interfere with compatibility testing if an antiglobulin phase is required.
This effect may persist up to 6 months after discontinuing the drug. The monoclonal antibody does not interfere with routine ABO/D typing.
Special techniques (neutralization of CD38 antibodies by CD38 anti-idiotypic antibodies, or soluble CD38 antigen) may remove the panreactivity but may not be generally available. DTT, a sulfhydryl reagent may denature the native CD38 antigen on RBCs but it should be used under a biologic hood.
Kell antigens will be denatured so Kell antibodies cannot be detected after treatment so Kell-negative RBCs should be used. In the Gulf Area, this is about 72% of RBCs.
Policy:
References:
Trick or Treatment, Anti-CD38 Reactivity and How to Treat It, AABB Satellite Symposium transcript, U. Cincinnati and RedMedEd, October, 2015 (attachment)
This is an updated version of a previous post.

At HMC Doha, I used service level agreements SLA to specifically define responsibilities in a contract for service between various entities including:
The following is a sample of a previous SLA between the Transfusion Medicine Division and one of HMC’s hospital’s operating theatres.



