


Donor ABO/D typing, antibody screening, antibody identification, ABO antibody titration


Principle:
There are several different types of comments in HIIG:
Physicians may enter any of these comment types. Comments may be entered before or after a test is authorized/verified. If entered after authorization, the test must be modified to accept the comment. Only result comments are visible in the patient’s medical record.
Policy:
Procedure—Patient Result Comments After Results are Authorized/Verified:
Sign into HIIG and select Patient module, Patient Consultation, and enter the patient hospital HC number.
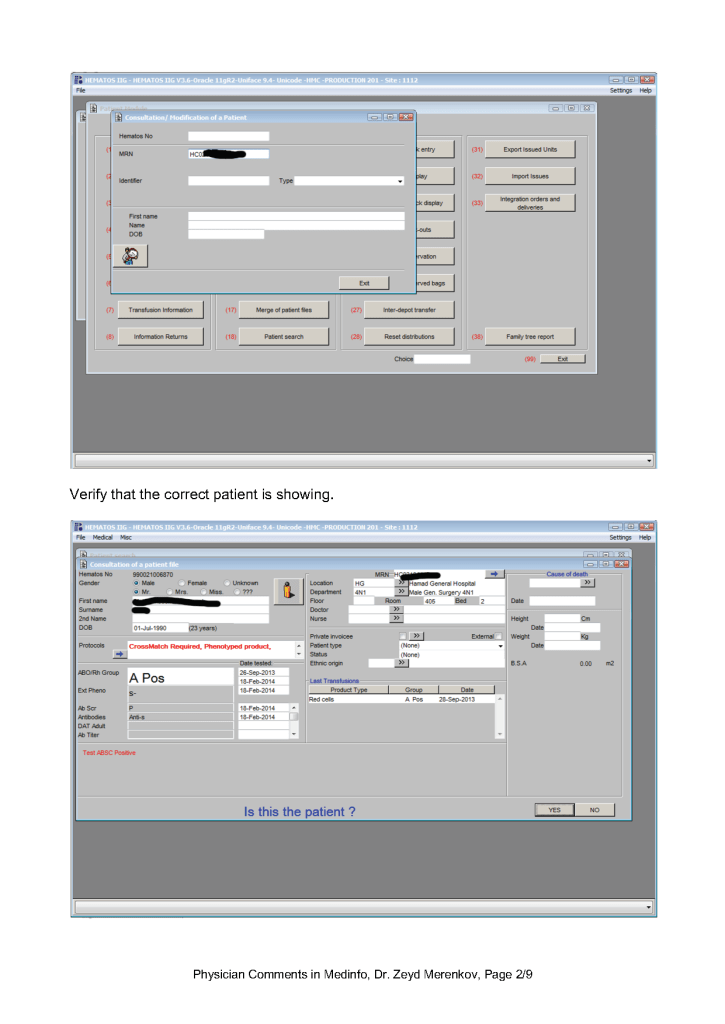
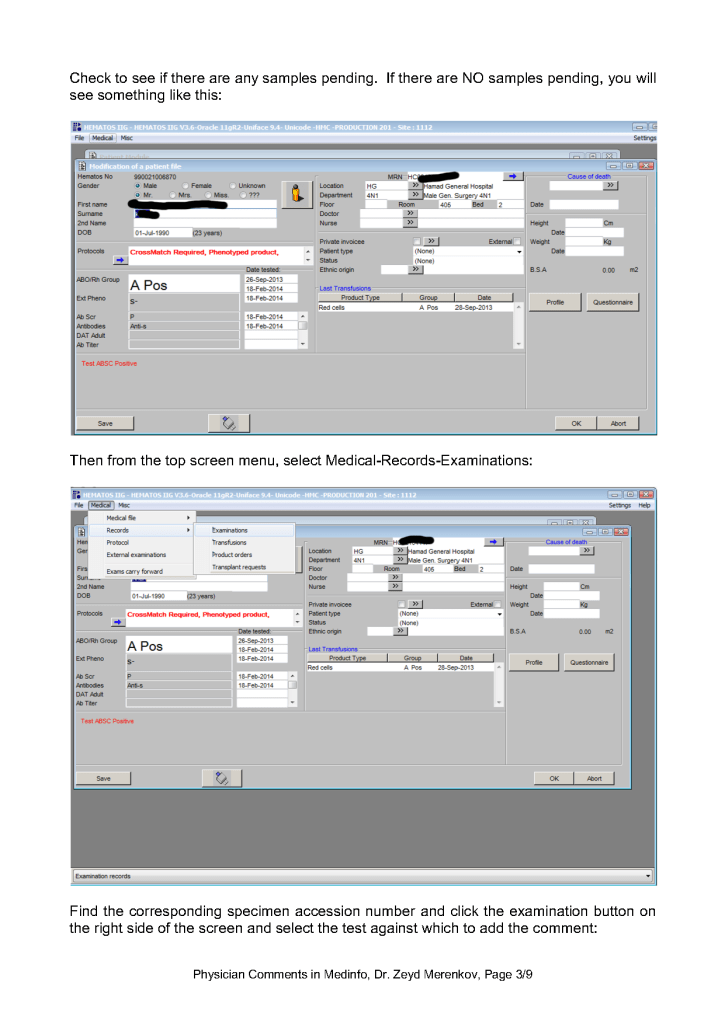
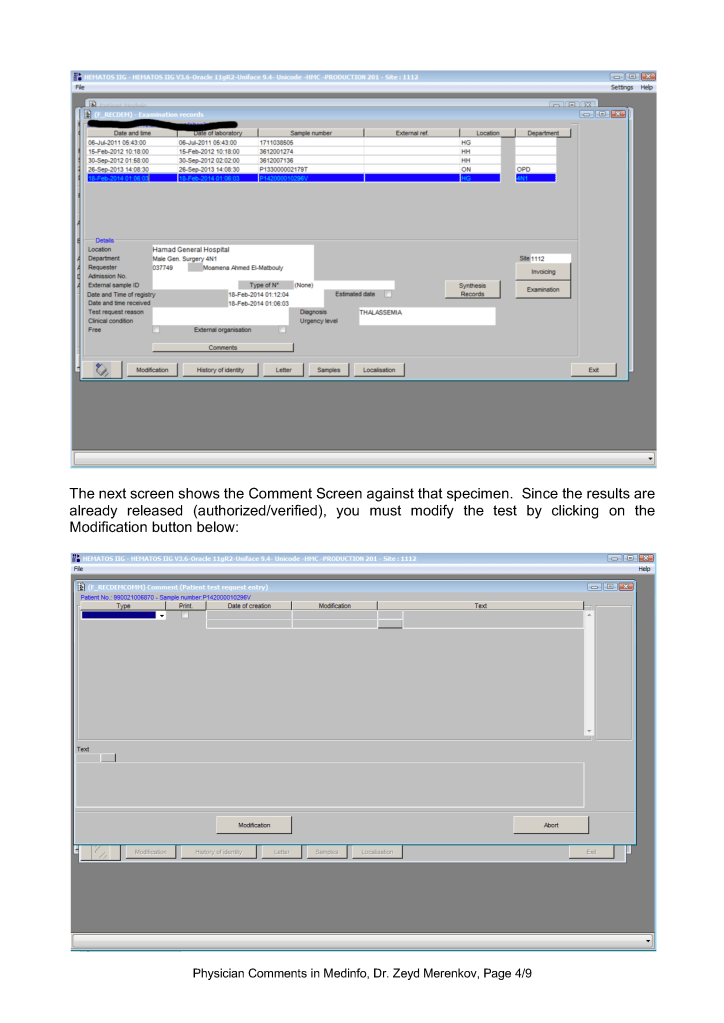
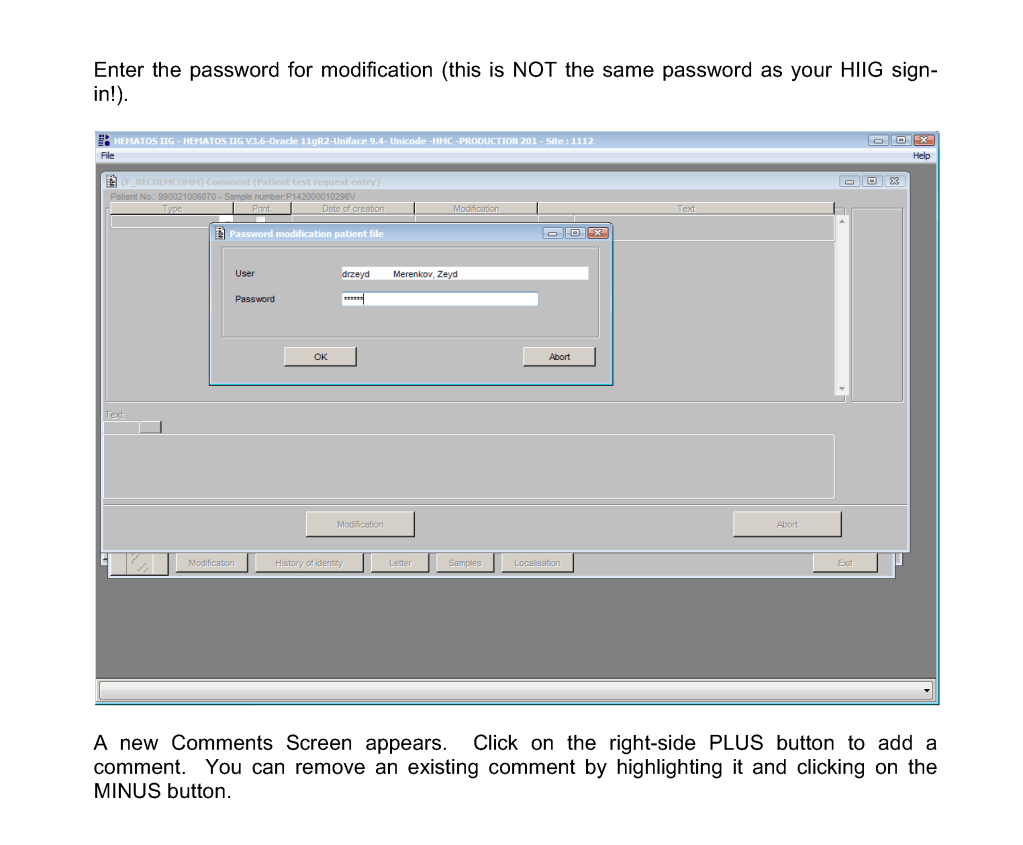
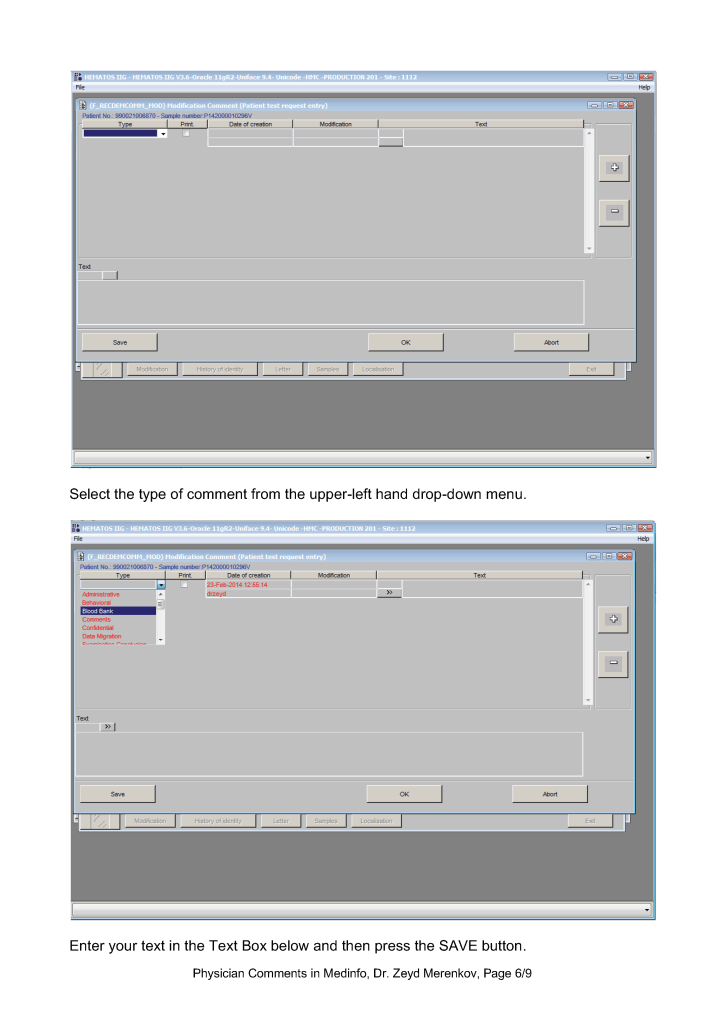

This post is the policy for using comments in Medinfo software. A subsequent post will show the process of entering comments.
Principle:
There are several different types of comments in HIIG:
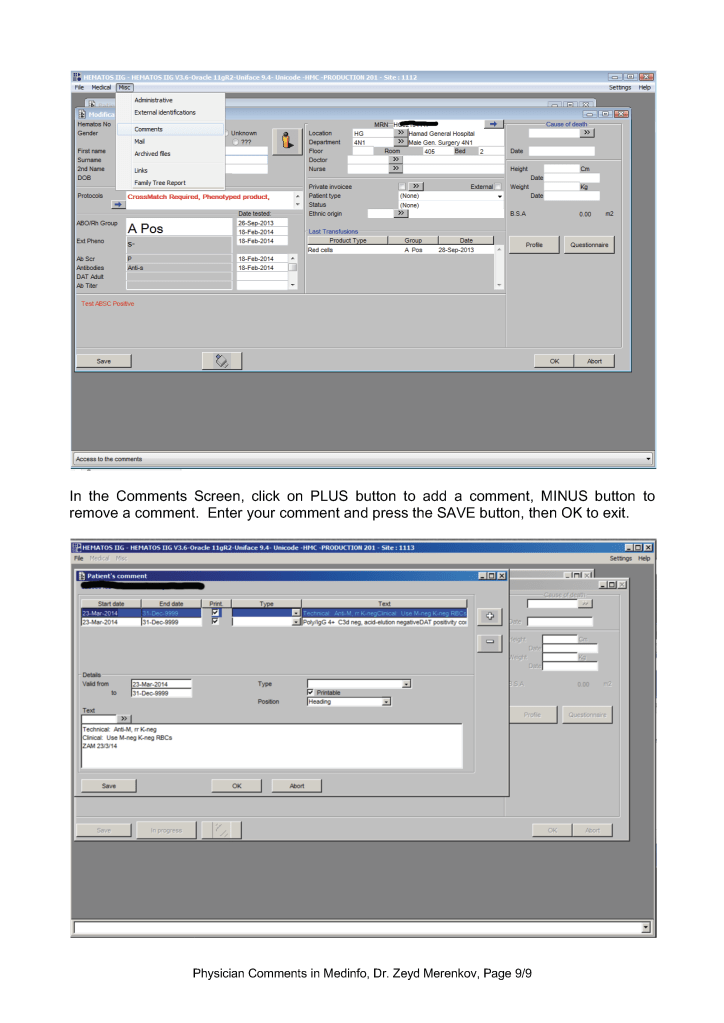
Global Comments appear on the first main screen of either the donor or patient record. The presence of comments is indicated by a bar at the bottom of the screen (in yellow or blue saying Presence of Comments. Double-clicking opens the list of entered comments.
Examination/Results Comments appear only when you open the result to which it is attached. You must know in advance to which result they are linked to find them.
Contraindication Comments appear when entering a donor deferral code
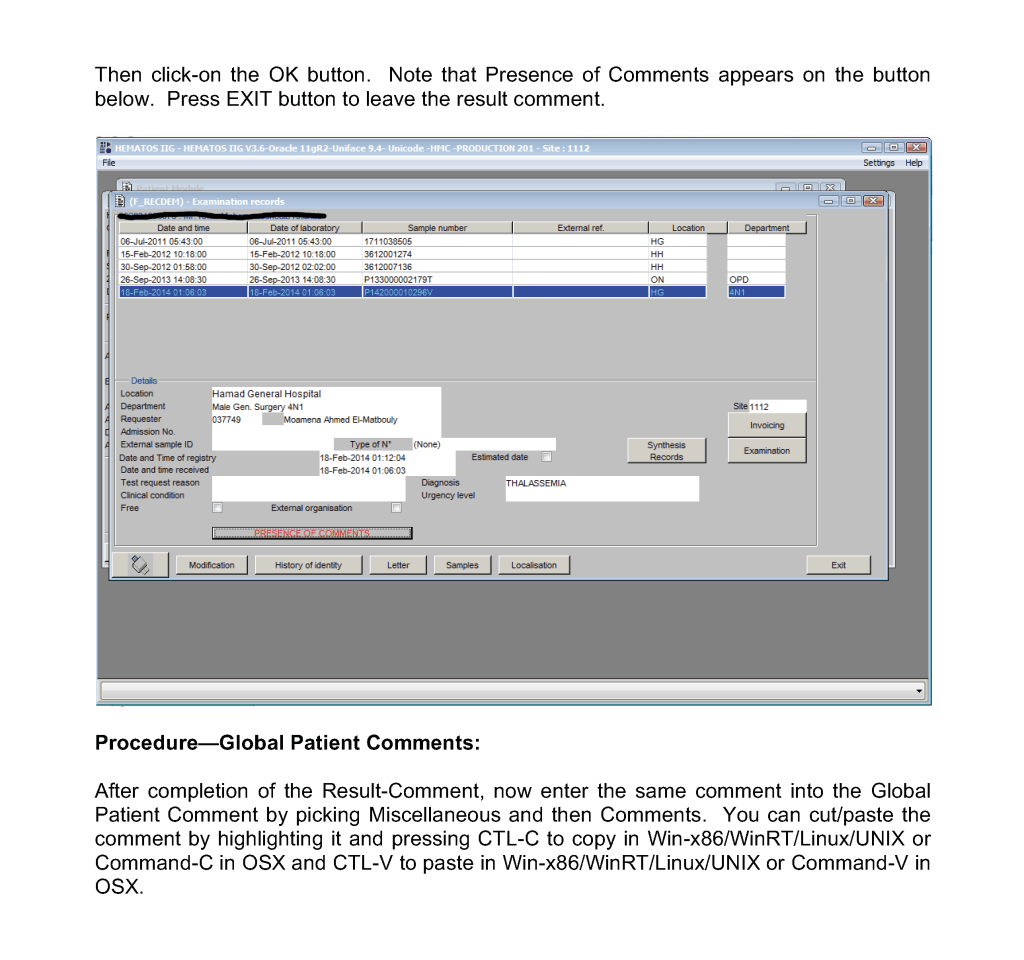
At HMC, we will enter examination/results comments again as global comments (donor or patient) so it is easy for staff to retrieve them and see them with all other comments. You can do this by cut and paste.
Physicians may enter any of these comment types. Comments may be entered before or after a test is authorized/verified. If entered after authorization, the test must be modified to accept the comment and require a special password (not the user sign-into HIIG). Only results/examination comments are visible in the patient’s medical record. Global, analysis, and contraindication comments are visible only in HIIG! Donor comments are only visible in HIIG.
The presence of comments documents physician review of abnormal results as required by the various accreditation standards.
Policy:
References:
This is a sample document for use of expired reagents I wrote for HMC Qatar.
Principle:
Due to logistics issues including the long distance between suppliers in Europe and North America and Qatar and the importation/customs clearance of critical materials, Transfusion Medicine has developed a contingency variance policy to minimize disruption of the essential transfusion medicine testing and component preparation. Approval for use of outdated reagents in special circumstances is not meant to be an excuse for untimely monitoring and improper ordering of supplies.
Definition:
Rare Reagent: Any reagent that is either used uncommonly or is in short supply and difficult to obtain in a timely matter.
Policy:
References:
Sections 1.3.2 and 7.0, Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
When I started my COVID-19 convalescent plasma CCP collection in early March, 2020, there were few antibody tests available. However, I anticipated that eventually we would want to include antibody results with the donor record. Antibody results were not used originally at all in the criteria for CCP acceptability for release.
There are many assays by type of antibody (total, IgG, IgA, IgM) and quantitation by titer and/or signal-cutoff ration S/CO. Any of these parameters may be used to define rules for acceptability to complete production and/or allocate to patients. Instrumentation used for titering/quantitation may be interfaced to the blood bank software.
Here is my generic approach to including these results with the donation record. In Medinfo HIIG, it is possible enter test results retrospectively and these can be used set rules for acceptability. Please consult with my detailed post on using rules against parameters.
All of this is easily implemented since all test information will be stored as parameters. From these parameters we can construct rules for:
Also, one can override the rules if the clinician and the transfusion medicine physician agree. For example, there is a severe shortage of group B CCP so use of low-COVID-antibody titer group B CCP could be allowed.
The key is to build whatever test methodology you use and include the manufacturer’s cutoff for low versus high titer interpretation. These results can be printed on the ISBT label as well. One can easily build multiple methodologies and acceptability criteria if different tests are used at different testing sites in your system—just as can be done for other tests (ABO/D, antibody screen, etc.) If one changes methodologies in the future, Medinfo will still use the same rules that applied for the day of production.
Here are some sample test rules:
Example 1: Total COVID antibody > 160 is high titer:
Example 2: IgG antibody with S/CO ratio > 12 is high-titer:
Example 3: IgG and IgM antibodies must have S/CO > 12:
Another option would be just to record the quantitation for each antibody type and list this on the ISBT label and permit its release regardless of the value. One could also permit low-anti-B titer group A plasma with whatever rules you set up.
Principle:
Fresh group O whole blood has viable platelets, plasma, and RBCs. Fresh whole blood may provide better resuscitation than individual components. It can replace MTP component therapy of separate RBCs, plasma, and platelets. We will use low ABO-titer whole blood units (here called O universal OU) in selected trauma cases, based on availability.
Testing for low-titer (both low-titer anti-A and anti-B) units is time-consuming and monopolizes the automated immunohematology analyzers. This is the rate-limiting step.
Policy:
References:
This post outlines a framework for establishing the use of universal group O whole blood. Manual titering large number of donor specimens in my organization is not precise. Using an automated system will also increase the precision of the results. The rate-limiting step is the ability to do the anti-A and anti-B titers.
Process:
Special notes:
References:
This is a teaching document for medical technology and transfusion fellows to explain the general structure of a validation.
Principle:
All validations must be planned. A validation protocol must be prepared with specific criteria for acceptance. All validations with attached evidence must approved by the Head, Transfusion Medicine.
Policy:
Reference:
Standards for Blood Banks and Transfusion Services, Current Edition, Bethesda, MD, USA



Note: This is an updated version of a previous post.
Principle:
Washing RBCs removes plasma and reduces the leukocyte count only by 1 log. For leukodepletion, we must rely on filtration to reduce the WBCs to less than 1 x 106 per unit according to CE rules. Red cells or platelets in additive solution contain only minimal plasma (about 35 ml). There are few definite indications for washing RBCs and it should be rarely necessary.
Policy:
Washing RBCs should only be done in the following circumstances:
Note:
Reference:
Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA