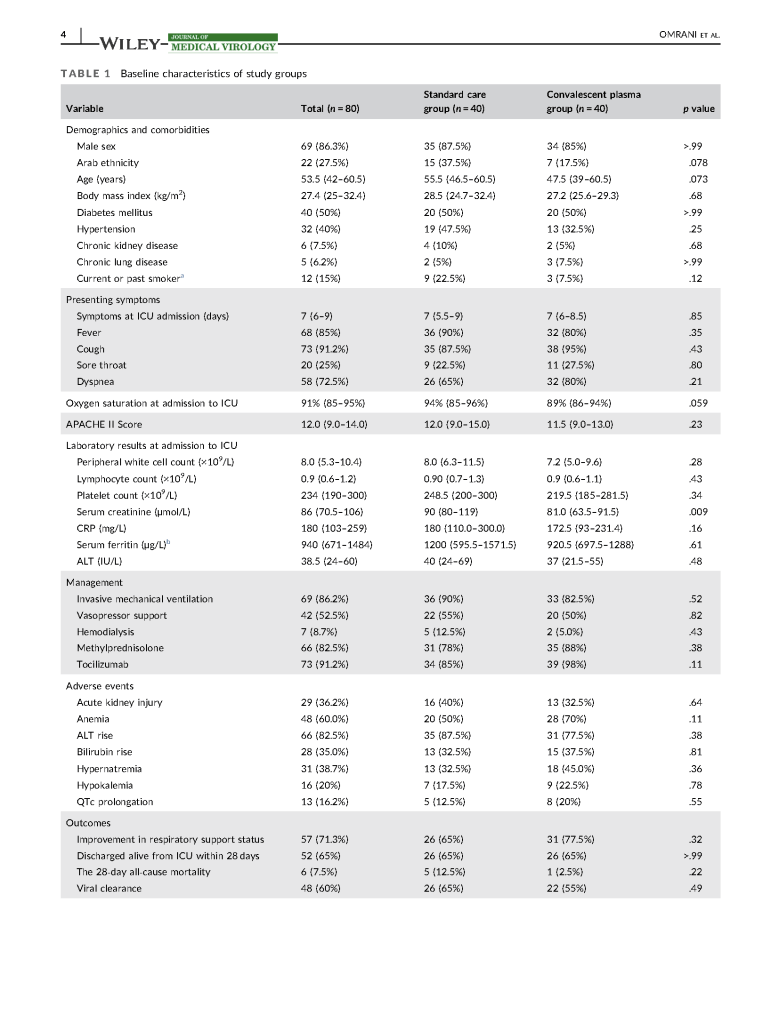
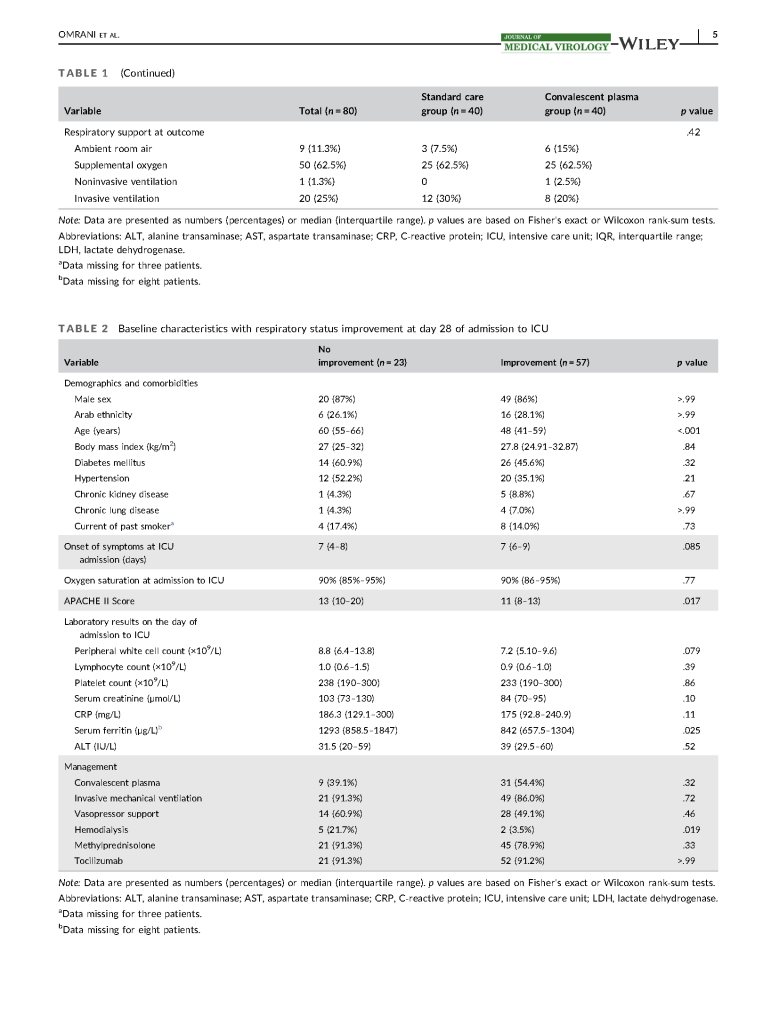
In February 2020, I established COVID-19 plasma collection and distribution for the state of Qatar. Here is a research paper prepared in conjunction with the Qatar Communicable Disease Center about using it for patients there.








Recruitment and Collection
In February 2020, I established COVID-19 plasma collection and distribution for the state of Qatar. Here is a research paper prepared in conjunction with the Qatar Communicable Disease Center about using it for patients there.









This is the policy I developed for HMC Doha Blood Donor Center:
Policy:
References:
Principle:
Until this guidance, everyone with hepatitis at age ≥ 11 has been permanently deferred, regardless of the type of hepatitis. Except for confirmed cases of HCV at any age, donors with other causes may be reassessed to determine if they may be re-entered into the donor pool. I have also included assessing levels of anti-HBs as per my previous policy at HMC Doha.
Policy:
References:
Requalification of Donors Previously Deferred for a History of Viral Hepatitis after the 11th Birthday, FDA-2017-D-5152–Requalification for History of Hepatitis Guidance-Final, US FDA/CBER, September 2017
Principle:
Under AABB and FDA rules in the Uniform Donor History Questionnaire, unlicensed, investigational vaccines have a 12-month deferral or as indicated by a responsible physician. In light of the anticipated vaccination trials for COVID-19, this policy gives interim guidance until more definitive information is available.
For COVID-19 Convalescent Plasma CCP donation, investigational vaccine recipients should not donate COVID-19 convalescent plasma until further information is available about their antibody profile.
Policy:
Any donor who has received a COVID-19 (SARS-CoV-2) vaccine will be deferred as follows:
Reference:
Text from the AABB Weekly Report:
Novel Coronavirus Update, Regulatory Update: Investigational Vaccines and Deferral for Donor of Blood and Convalescent Plasma, AABB Weekly Report, 7 August 2020
“FDA recognizes AABB’s DHQ which includes unlicensed (experimental) vaccines on the medication deferral list as a 12-month deferral or as indicated by the responsible physician.
“For routine blood donation, the responsible physician may wish to consider the potential infectious risk associated with the vaccines, and the use of short deferral periods (e.g., 14 days) for live attenuated vaccines and no deferral for non-replicating, inactivated or RNA-based vaccines.
“We agree that no deferral is necessary for routine blood donors who might have received the mRNA-1273 Moderna vaccine.
“At this time, we suggest that individuals who have received a COVID-19 investigational vaccine should not donate COVID-19 convalescent plasma until further information is available about their antibody profile.”
I developed this policy for HMC Doha where most of the local population are CMV-seropositive. Note that I used the CE definition of <1E6 instead of the American <5E6.
Principle:
Since most of the local population (>90%) are CMV-seropositive, it is impractical to rely on CMV-negative donors as our basis for CMV prophylaxis. Instead, we perform universal leukodepletion and pathogen-inactivation to greatly reduce this risk:
Policy:
References:
Principle:
New rules have been approved by US FDA CBER for body fluid exposure, tattooing, body fluid exposure, and body-piercing. By similar logic we will extend this also to HIJAMA. We will not make any changes to our sexual history/practices or history of sexually transmitted disease treatment.
Policy:
Reference:
Revised Recommendations for Reducing the Risk of Human Immunodeficiency Virus Transmission by Blood and Blood Products, Guidance for Industry, U.S. Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research, April 2020
Principle:
Maintaining an adequate blood supply and expedited compatibility testing are critical in disaster planning. This plan is assuming that the Blood Donor Center is functional and can process donors and make components.
Medinfo Hematos IIG System is critical to monitoring inventory, preparing blood components expeditiously using Good Manufacturing Processes, and distributing blood components in a timely controlled manner.
Policy:
References:
Revised 10/9/20
Principle:
HMC Blood Donor Center is implementing a policy to limit or help limit iron deficiency in its blood donors (whole blood and/or apheresis). The reasons for this are:
Definition:
Donors high-risk for iron loss include:
Policy:
References:
AABB Association Bulletin #17-02, Updated Strategies to Limit or Prevent Iron Deficiency in Blood Donors, 26/3/17
Updated 6/9/20
Principle:
Normally, outside donor campaigns still connect to the main server via wireless 4G/5G with a VPN. However, if there is a “dead” spot, Medinfo can provide a local area network using one of the PCs/laptops to serve as a server. The local server receives an uploaded image of the donor database. Upon return to the donor center, the local server’s data is uploaded and synchronized.
When you cannot establish a direct link to the live Medinfo program, you must arrange for Medinfo/VHT to create a local server that will have the current Medinfo donor database for use at outside campaigns where the internet connection cannot be used. This can also be used if for some reason the Blood Donor Center link is down in order to register donors and check the donor deferral database.
Policy:
7/9/20
Reading Assignments:
Study Questions:
Revised:
29/8/20