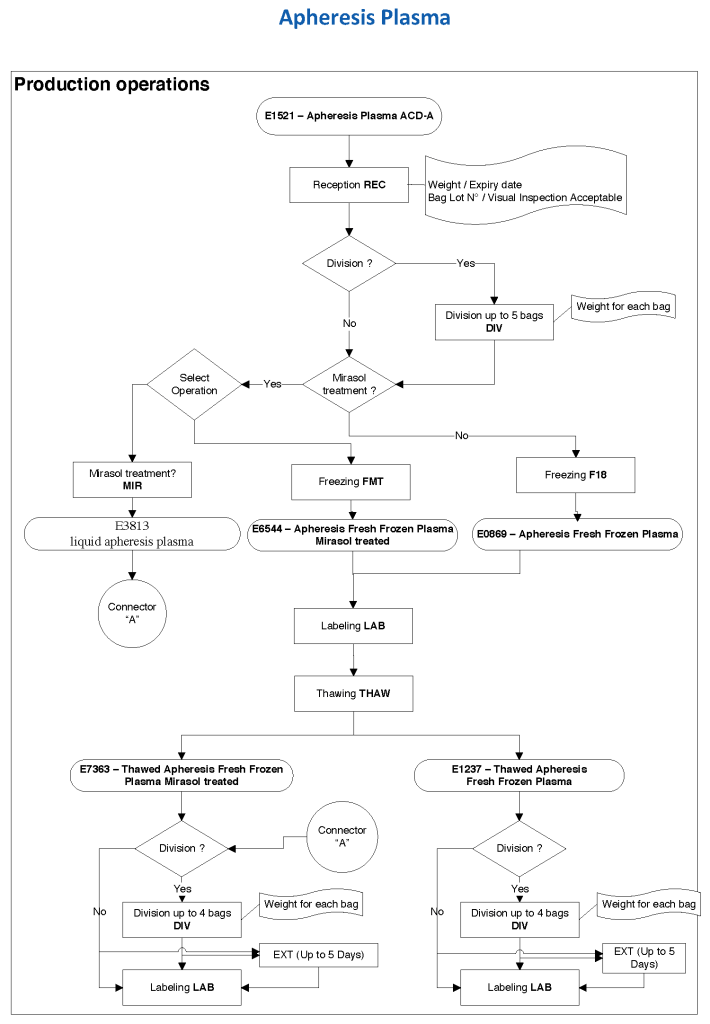
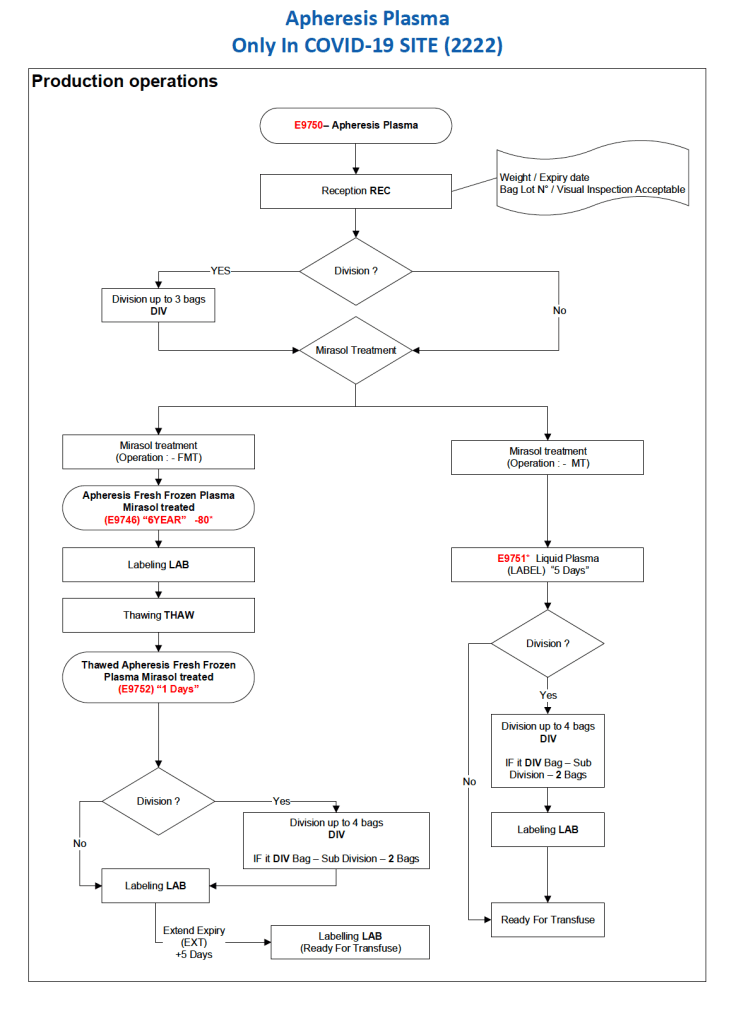
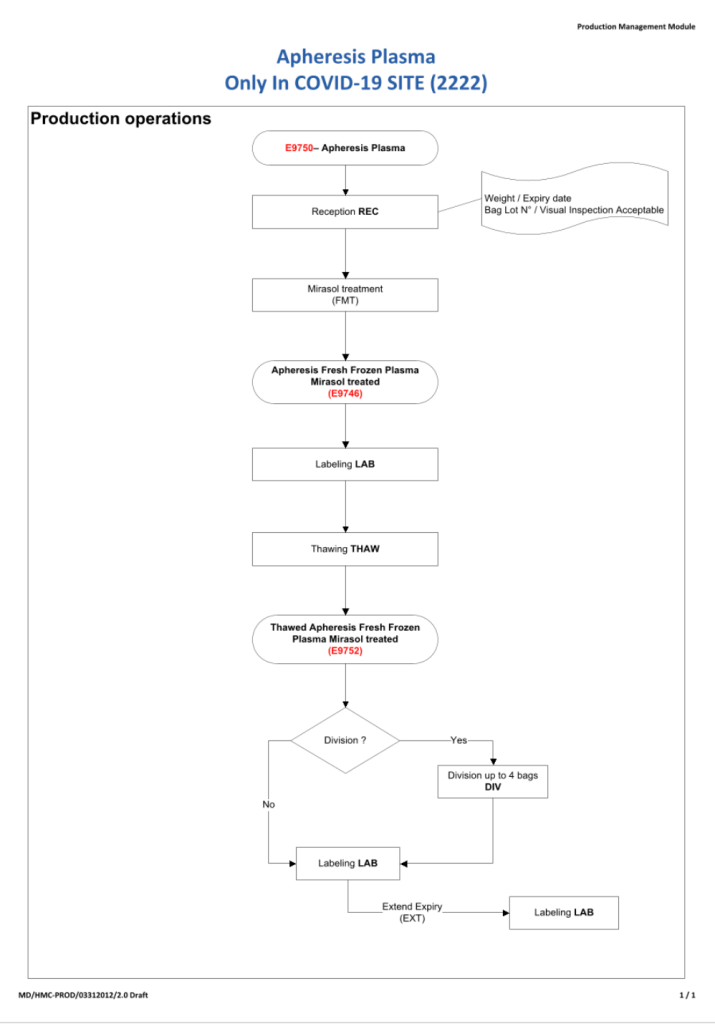
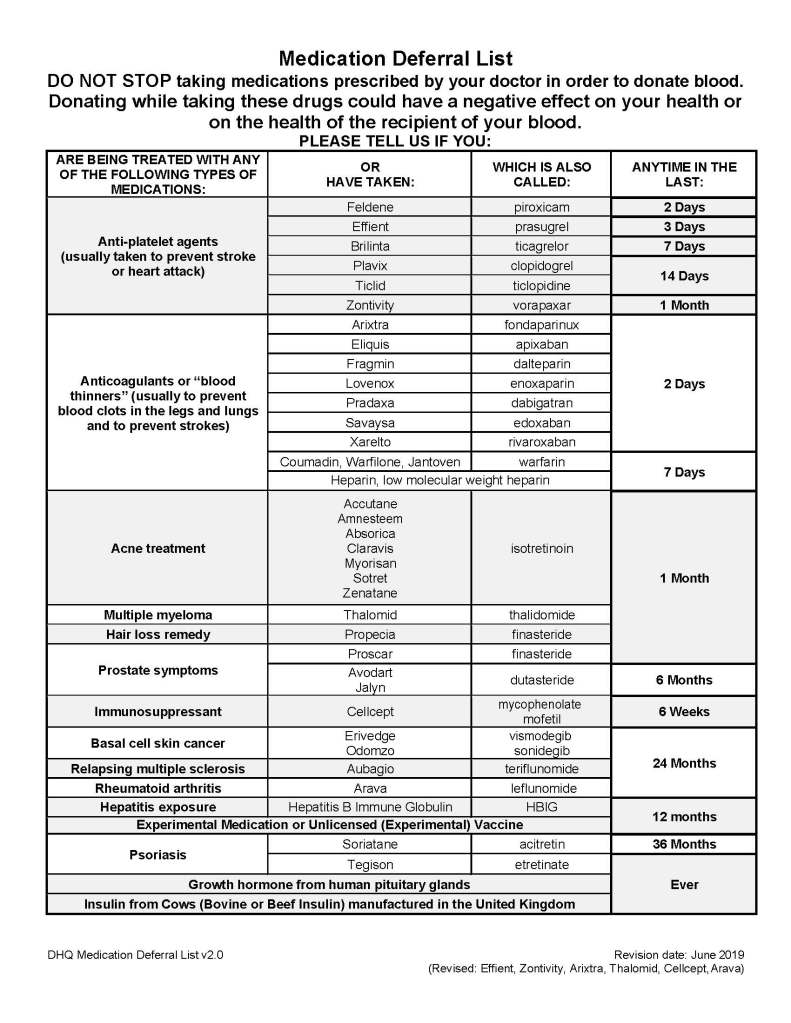
Principle:
Under AABB and FDA rules in the Uniform Donor History Questionnaire, unlicensed, investigational vaccines have a 12-month deferral or as indicated by a responsible physician. In light of the anticipated vaccination trials for COVID-19, this policy gives interim guidance until more definitive information is available.
For COVID-19 Convalescent Plasma CCP donation, investigational vaccine recipients should not donate COVID-19 convalescent plasma until further information is available about their antibody profile.
Policy:
Any donor who has received a COVID-19 (SARS-CoV-2) vaccine will be deferred as follows:
- Whole blood or apheresis donation (except COVID-19 convalescent plasma):
- Live, attenuated vaccine: 14 days post vaccination
- Non-replicating, inactivated, or RNA-based vaccine: NO DEFERRAL
- COVID-19 Convalescent Plasma CCP Donation: DO NOT ACCEPT
Reference:
Text from the AABB Weekly Report:
Novel Coronavirus Update, Regulatory Update: Investigational Vaccines and Deferral for Donor of Blood and Convalescent Plasma, AABB Weekly Report, 7 August 2020
“FDA recognizes AABB’s DHQ which includes unlicensed (experimental) vaccines on the medication deferral list as a 12-month deferral or as indicated by the responsible physician.
“For routine blood donation, the responsible physician may wish to consider the potential infectious risk associated with the vaccines, and the use of short deferral periods (e.g., 14 days) for live attenuated vaccines and no deferral for non-replicating, inactivated or RNA-based vaccines.
“We agree that no deferral is necessary for routine blood donors who might have received the mRNA-1273 Moderna vaccine.
“At this time, we suggest that individuals who have received a COVID-19 investigational vaccine should not donate COVID-19 convalescent plasma until further information is available about their antibody profile.”