









Establishment of a local plasma fractionation to ensure a supply of plasma derivatives such as IVIG, RhIG, albumin, factor concentrates











This is an update of a previous post.
I have been involved with planning for several plasma fractionation projects in the Middle East.
Many clients expressed the interest in using local plasma to make plasma derivatives (e.g. factor concentrates, intravenous gamma globulin, albumin), feeling that local plasma was safer than using imported plasma. Some of these are in short supply in the world market so the only way to ensure their uninterrupted availability is to consider to manufacture them for local consumption.
Still, the major issue today is that it is difficult for any country in the region to collect enough plasma to make such a project feasible. When I first considered such planning, we were looking for as much as 250,000 raw liters of plasma annually. Since then, there are newer technologies that allow much smaller batches to be cost-effective. Alternatively, one could charge higher prices for using smaller batches from local plasma.
Still, it is likely that plasma must be imported to sustain a plant. There are different regulations for plasma donor qualification country-to-country. Many of these jurisdictions may do less screening and testing than is done for normal blood and apheresis donors. Other countries use their blood donors with the same requirement for both commercial plasma and blood donations.
In this era of emerging infectious diseases, I personally favor using the stringent blood donor criteria—same as routine collections. It is not what we know, but the unknown pathogens that are potentially the most dangerous.
In addition to building a fractionation plant, one must train staff for this highly technical operation. This may require developing a special curriculum to prepare students for these jobs.
To export the plasma to certain regions, one may have to use plasma quarantine. In this protocol, plasma is held or quarantined until the next donation is collected and passes screening. This requires a robust blood bank production software such as Medinfo to track serial donations.
There are other processes to consider: how to develop a transport network to keep plasma frozen at minus 80C viable in a region that reaches very high ambient temperatures.
I would recommend a graded approach to develop such an industry. First I would negotiate a plasma self-sufficiency arrangement. We would collect local plasma in the country and export it to a manufacturing plant in another country and the derivatives would be returned to us. This may require inspection by the accreditation agency of the processing country to allow importation of the raw plasma for manufacture.
Since it is unlikely any one country has enough plasma for manufacture, recruiting neighboring countries to participate in a manufacturing plant is important. Technology for such a plant is complex so establishing a joint venture with one of the plasma industry companies is essential. Some manufacturers are very keen to develop extra capacity since there is a world-wide shortage of plasma fractionation and are even willing to help obtain external plasma sources for such a plant.
Such a plant is an excellent way to develop local talent to run such a plant, including training of local staff to be the industrial engineers in the plasma fractionation process. It would take approximately two years of training to prepare engineers on-site at a plasma fractionation site if they have studied the necessary science and mathematics subjects.
Such a program would take several years of planning and development. Some of the major steps needed include:
This is a teaching document of a process to irradiate RBC components when our irradiators are not functioning. In our system, we used Mirasol pathogen-inactivation so our RBC units were affected. The number of units for advance irradiation was based on our historical usage of irradiated units across our system. The workweek is Sunday-Thursday so on Thursday special effort was made to have the minimum number of irradiated units available.
We used irradiated units for compatibility testing to avoid the possibility that the unit would be released without irradiation.
Emergency Interim Procedure:
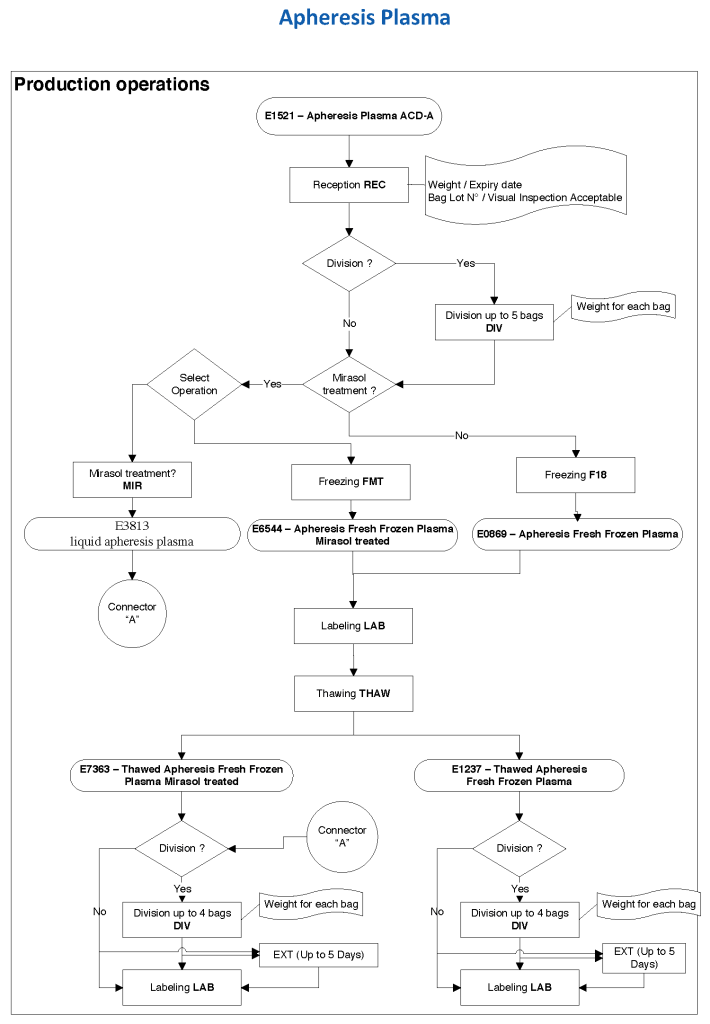
At HMC during my tenure, all plasma products—whole-blood and apheresis-derived were pathogen inactivated with riboflavin (Mirasol). In our software processes, I had options to release both Mirasol-treated and untreated (the latter in emergencies) and to aliquot either as needed. The same processes applied to COVID-19 convalescent plasma CCP except that they were performed in a quarantine production area. There were specific ISBT codes for CCP.
24/9/20
Transfusion Medicine includes laboratory and non-laboratory functions. The non-laboratory and purely clinical functions are unique and have no analogy within the general laboratory.
The transfusion service/hospital blood bank laboratory is the closest to a laboratory operation, but there is component modification and complex manual testing, especially for reference immunohematology testing. The staff must make detailed manual decisions, the errors for which could be life-threatening for the patient.
The blood donor center manufactures a pharmaceutical, i.e. blood components with collection, donor qualification, donor abnormal results review, infectious disease marker testing, component production, and donor immunohematology testing—all subject to Good Manufacturing Practices. Never forget: Blood is a drug!!
No other laboratory section is directly responsible for treatment of critically ill patients. Therapeutic apheresis is essential for organ and stem-cell transplants, nephrology, neurology, etc. No other laboratory section is directly responsible for treatment of critically ill patients. Transfusion Medicine physicians are functioning as intensivists. There is no hiding in the laboratory from clinical medicine.
There may also be an industrial manufacturing plant to extract various blood derivatives (e.g. factor concentrates, albumin, Rh immune globulin, etc.) This is pharmaceutical manufacturing on a large-scale basis. There is medical, technical, and special administrative expertise.
Many functions may operate 24/7. The transfusion medicine physician may be on-call for donor issues and review of complex immunohematology problems to acutely decide which blood component (and phenotype) should be given as well as review all adverse reactions to transfusion.
The unique blend of clinical skills is unlike anything else in the laboratory. Also, those outside the blood bank rarely have the skills or judgments for the best course of action for transfusion medicine or for its operations.
The clinical transfusion medicine physician must make acute, life-threatening decisions unlike anyone else in the laboratory. The blood bank technologist is at the cutting edge of the battle with his testing and interpretations. No other area of the laboratory is at such risk for injuring or even killing the patient. There is high stress and burn-out.
I have talked with many blood bankers and many seem to share the exasperation that the laboratory does not understand us. The latter looks at blood bank testing like that coming off a hematology or chemistry analyzer—although patients rarely would have severe morbidity or mortality like the blood bank from errors in those analyzers.
No laboratory pathologist has the pressure of the blood bank physician on-call. It really is 24/7 and requires a broad, clinical background to make the right decisions. It is very stressful and does not permit a good night’s sleep.
Thus, I make my case to separate us from the laboratory. We can form our own more effective administrative organization and optimize our own planning. Regretfully, I have never worked in such an administrative structure. I also am a realist that cost-containment nowadays makes it much less likely high administration would permit this change for a mere cost center. This will probably never happen during my career.
Finally, Transfusion Medicine is an essential service. Blood components are essential drugs. The operations and staff must be free of political influences. This is a service for the entire region or country like the fire department, civil defense, etc.
8/8/20
I have been involved with planning for several plasma fractionation projects in the Middle East.
Many clients expressed the interest in using local plasma to make plasma derivatives (e.g. factor concentrates, intravenous gamma globulin, albumin), feeling that local plasma was safer than using imported plasma. Some of these are in short supply in the world market so the only way to ensure their uninterrupted availability is to consider manufacture them for local consumption.
Still, the major issue today is that it is difficult for any country in the region to collect enough plasma to make such a project feasible. When I first considered such planning, we were looking for as much as 250,000 liters annually. Since then, there are newer technologies that allow much smaller batches to be cost-effective. Alternatively, one could charge higher prices for using smaller batches from local plasma.
Still, it is likely that plasma must be imported to sustain a plant. There are different regulations for plasma donor qualification country-to-country. Many of these jurisdictions may do less screening and testing than is done for normal blood and apheresis donors. Other countries use their blood donors with the same requirement for both commercial plasma and blood donations.
In this era of emerging infectious diseases, I personally favor using the stringent blood donor criteria—same as routine collections. It is not what we know, but the unknown pathogens that are potentially the most dangerous.
In addition to building a fractionation plant, one must train staff for this highly technical operation. This may require developing a special curriculum to prepare students for these jobs.
To export the plasma to certain regions, one may have to use plasma quarantine. This requires a robust blood bank production software such as Medinfo to track serial donations
There are other processes to consider: how to develop a transport network to keep plasma frozen at minus 80C viable in a region that reaches very high ambient temperatures.
I would recommend a graded approach to develop such an industry. First I would negotiate a plasma self-sufficiency arrangement followed by recruiting neighboring countries to participate in a manufacturing plant. Technology for such a plant is complex so establishing a joint venture with one of the plasma industry companies is essential. Some manufacturers are very keen to develop extra capacity since there is a world-wide shortage of plasma fractionation and are even willing to help obtain external plasma sources for such a plant.
Such a plant is an excellent way to develop local talent to run such a plant, including training of local staff to be the industrial engineers in the plasma fractionation process. It would take approximately two years of training to prepare engineers on-site at a plasma fractionation site if they have studied the necessary science and mathematics subjects.
Such a program would take several years of planning and development. Some of the major steps needed include:
I would be glad to discuss the matter in more detail if there is further interest.
I am an independent consultant in Transfusion Medicine. Effective 16 April 2020, I am no longer associated with Hamad Medical Corporation or the State of Qatar.
I am willing to consider other opportunities in Transfusion Medicine (donor, patient, apheresis) and blood bank informatics.
Just before leaving HMC, I established the COVID19 convalescent plasma program with full good manufacturing practices using Medinfo Hematos IIG blood bank software.
I have 10 year’s experience in pathogen inactivation and blood component automated production. I established the first site using Terumo Atreus (later Reveos) with Mirasol pathogen inactivation AND platelet additive solution. I established Medinfo interfaces with all production equipment to achieve GMP.
I have worked with laboratory information systems, especially but not limited to blood bank systems (donor, component processing, donor marker testing, pathogen inactivation, platelet additive solutions) and serve as the Head of the Medinfo IIG (Nice, France) Software Users Group.
I was involved with planning for the national plasma fractionation project in Saudi Arabia. I have worked with this industry while I was practicing in the United States.
It is my philosophy to start with an international framework (e.g.FDA, CE) and localize it for the country’s particular needs. My operation sites have served as international reference sites for combined IT and medical/technical processes.