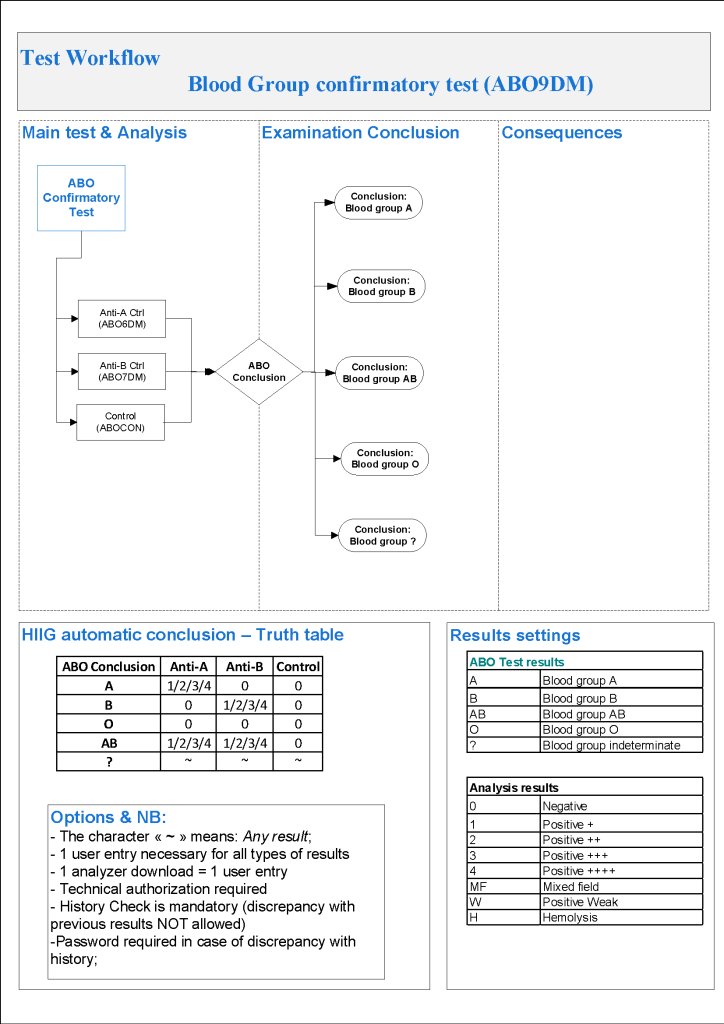
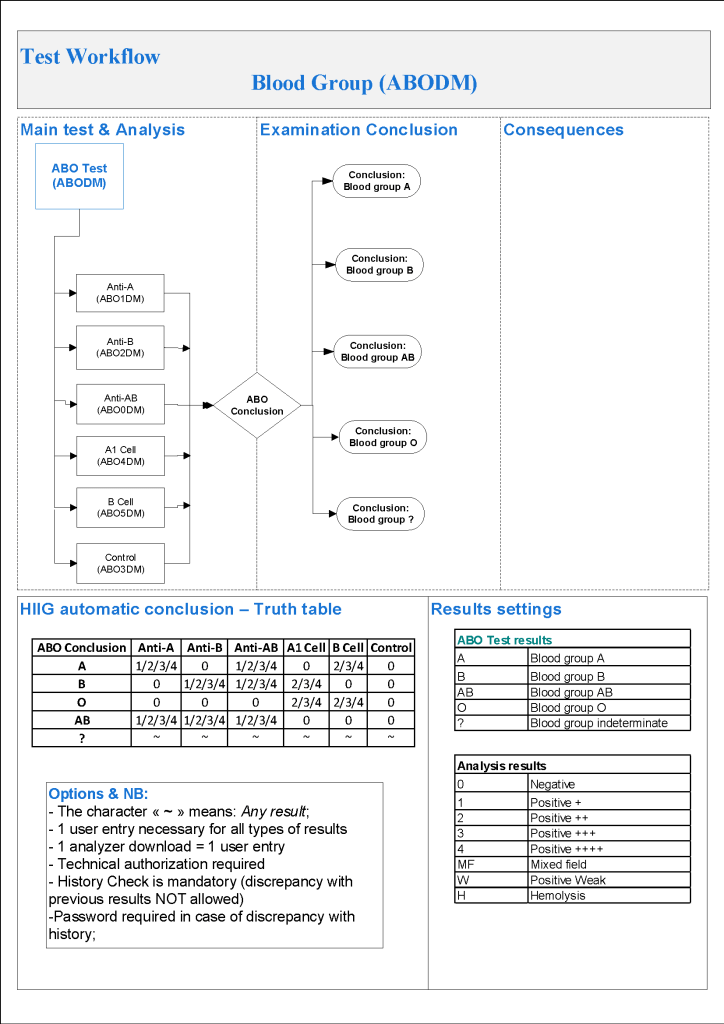
For donor ABO confirmatory typing, I always had both automated and manual methods set up on the blood bank computer system Medinfo Hematos IIG. The automated method had a bidirectional interface between Medinfo and the instrument. Medinfo did not need a separate middleware. A truth table was prepared for acceptable results for automatic interpretation. Other results had to be manually interpreted by someone with the appropriate security level.
The manual testing option is structured similarly. Within Medinfo, it is easy to change the methodology if the system is so built. Thus, if the analyzer for ABO typing is down, the staff can select the manual methodology. Likewise, if one testing center goes off-line, the world can be completed at another site—no need to repeat testing already completed from the first site. This flexibility can apply to any test in system.
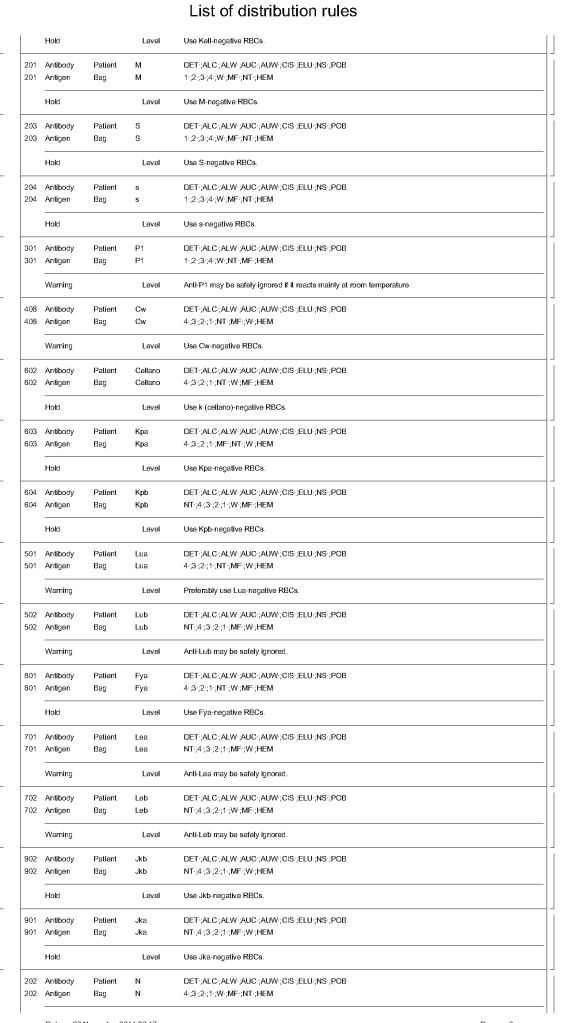
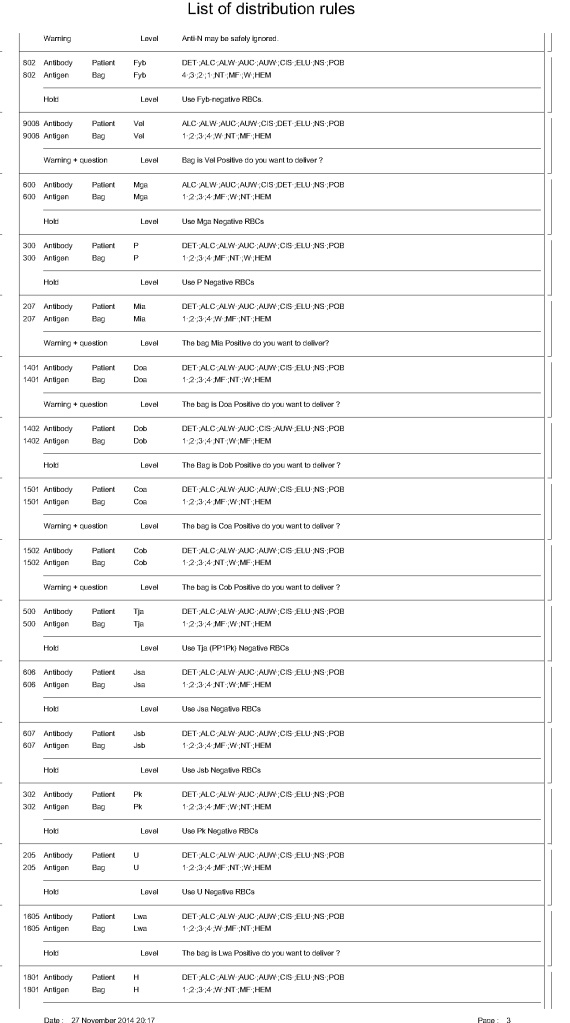
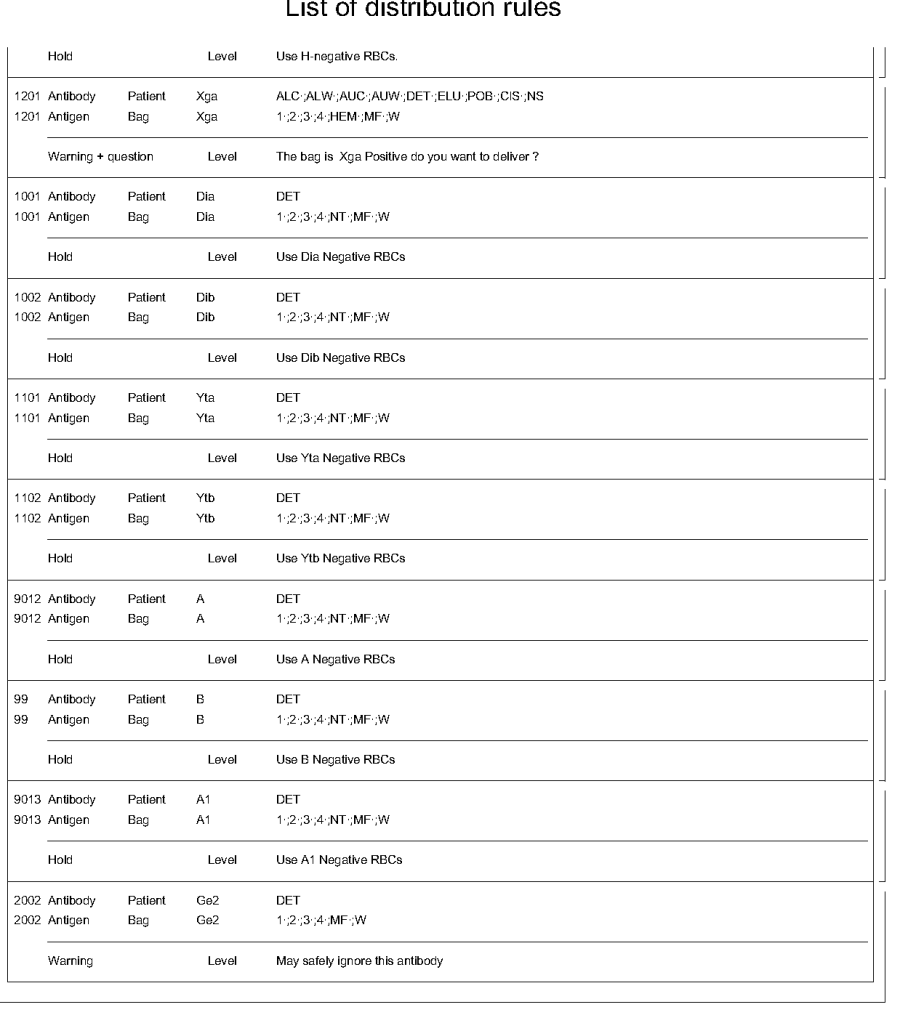
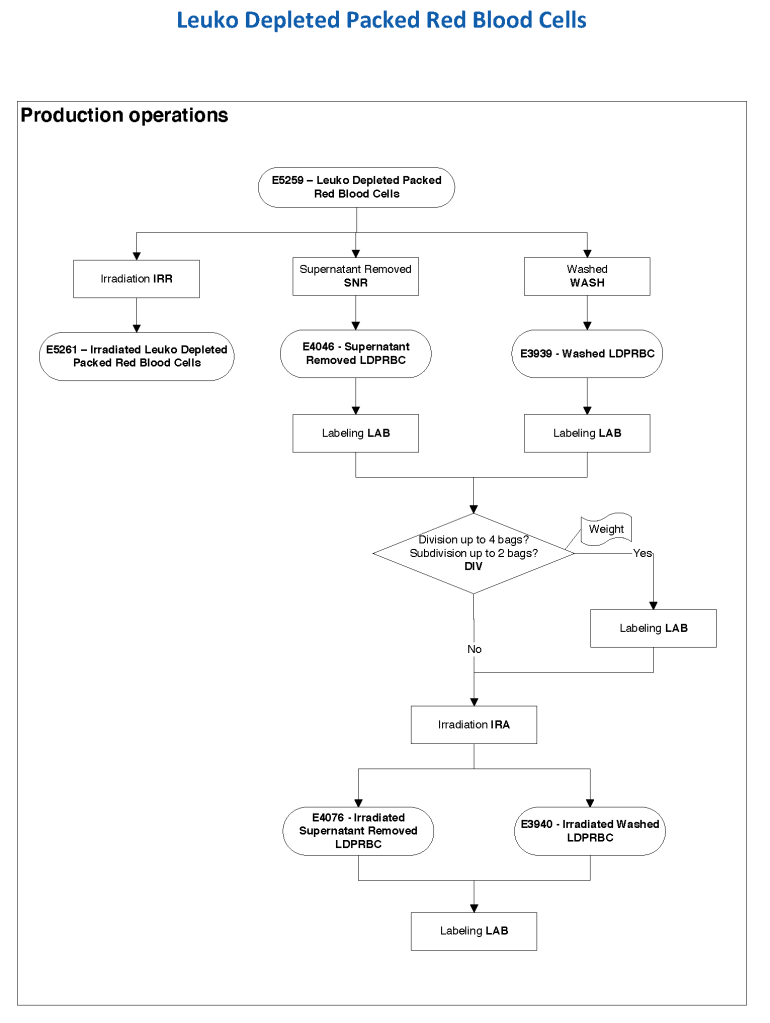
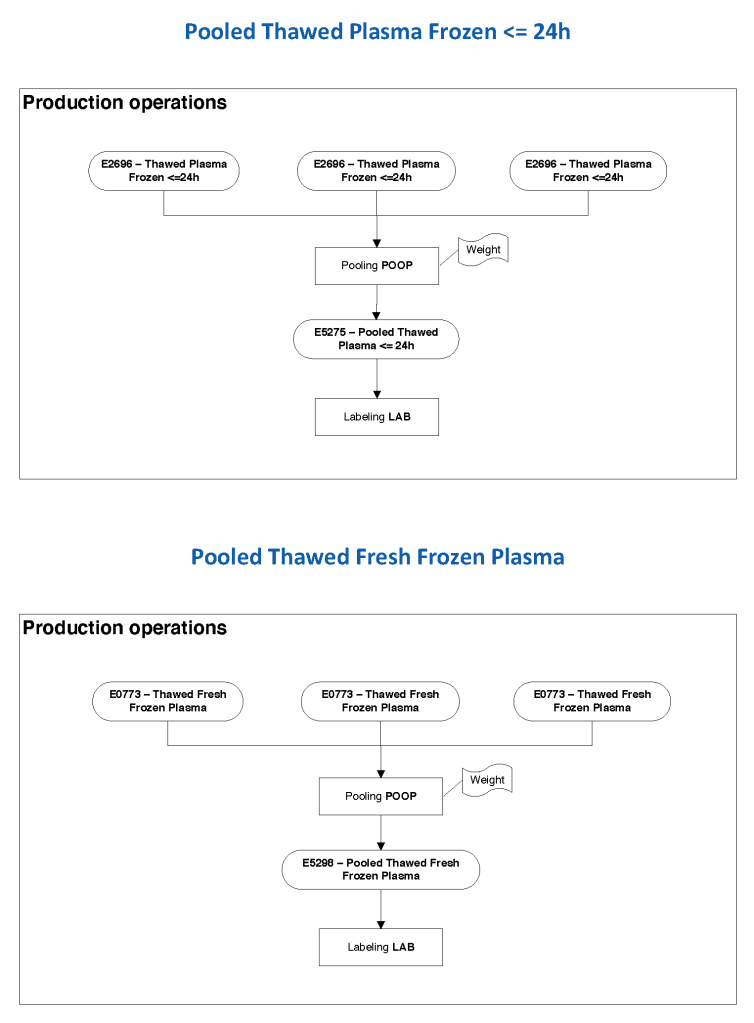
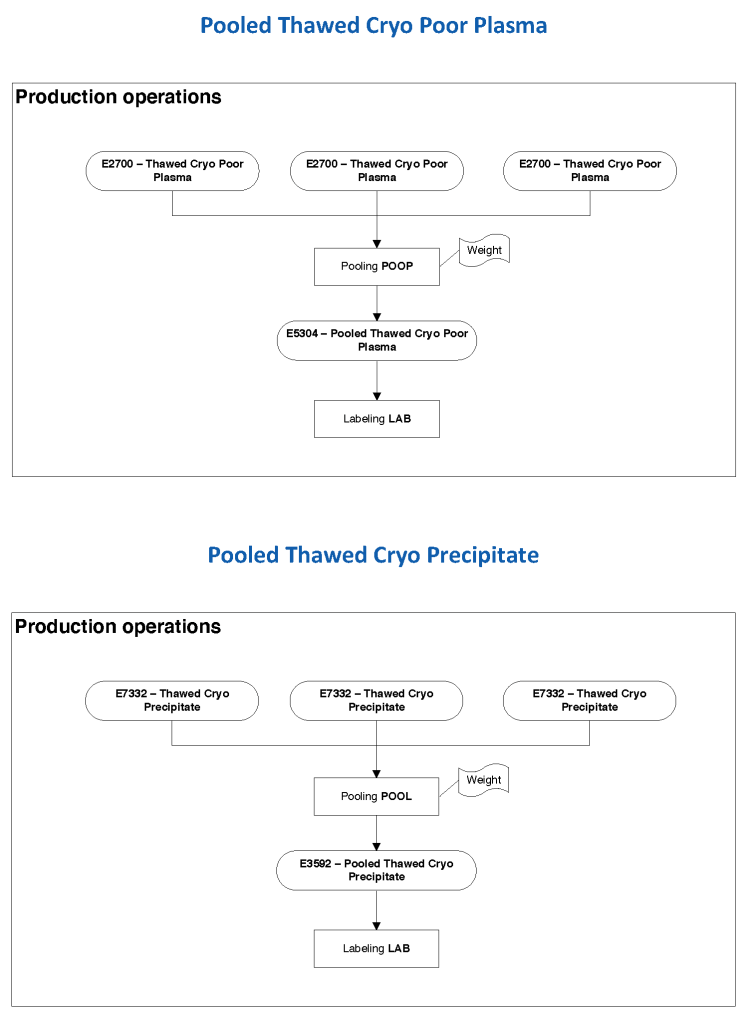
The manufacturer’s recommendations for the particular reagents in use were strictly followed. One used the range {0, 1, 2, 3, 4} as acceptable. Another used {0, 2, 3, 4}. Controls were included. Most importantly, Medinfo can be configured for any set of reagent values. Refer to the following flow diagram.
Confirmatory testing also includes D typing. That will be considered in a future post.
6/10/20