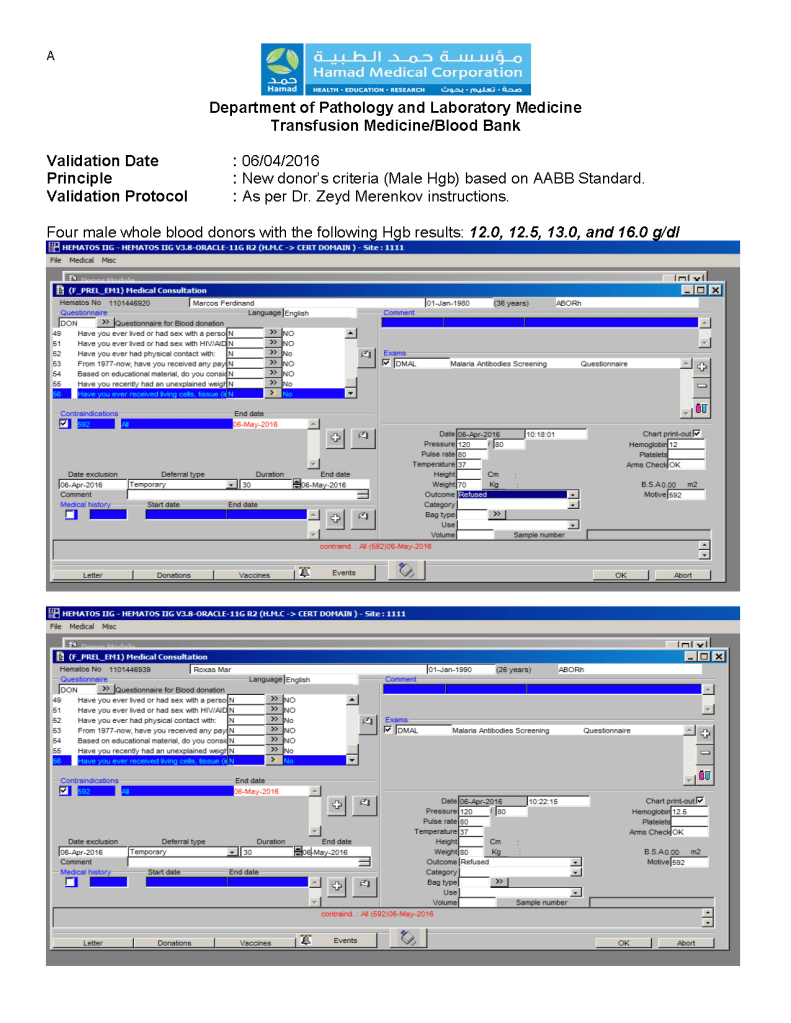
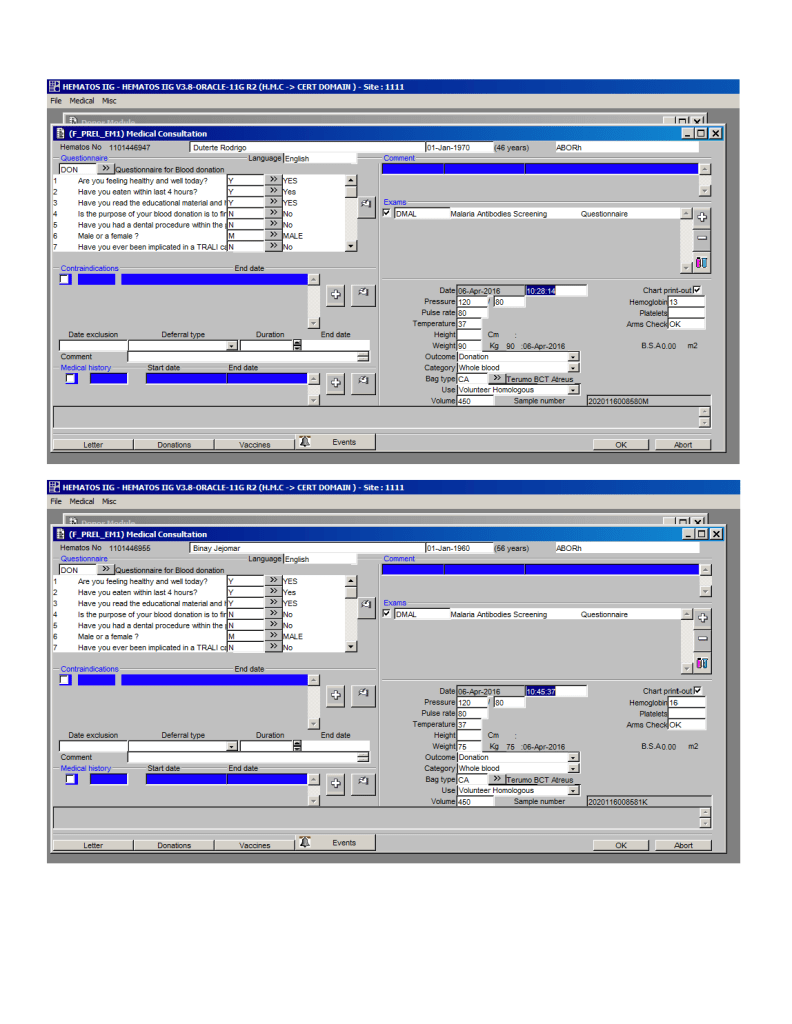
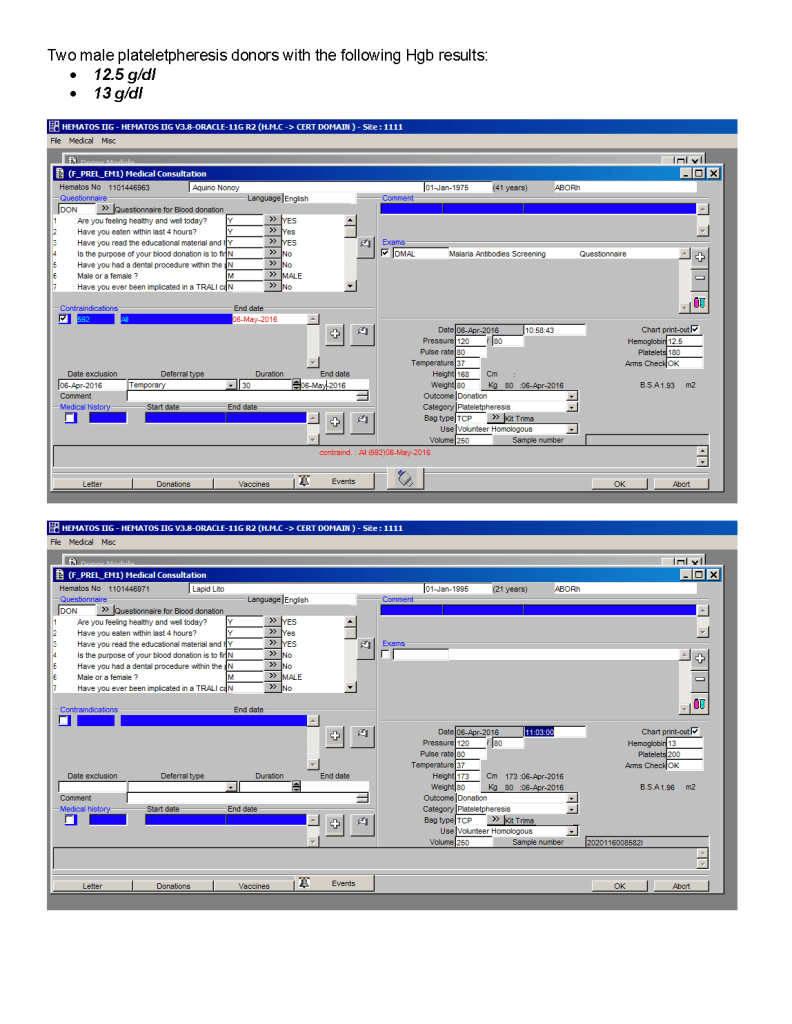
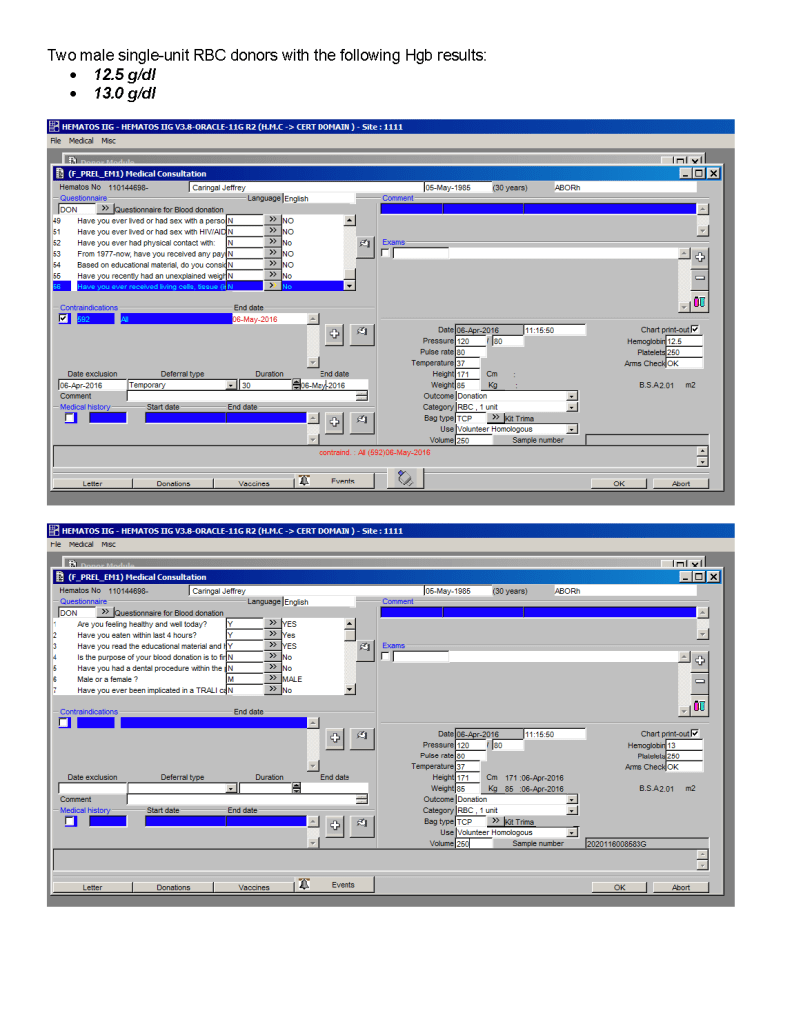
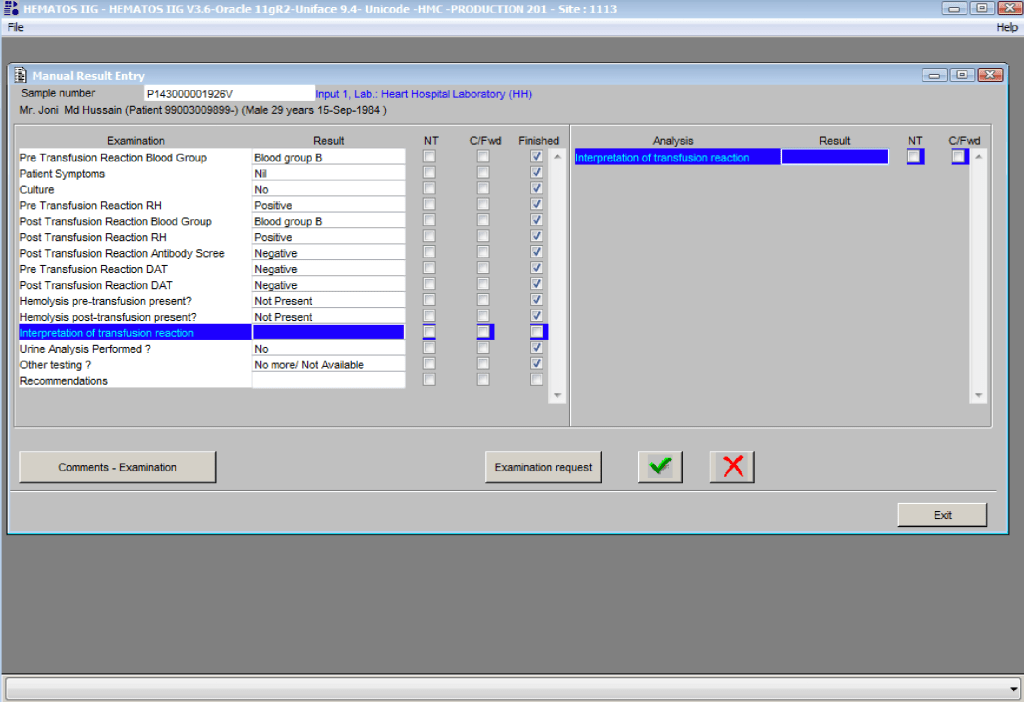
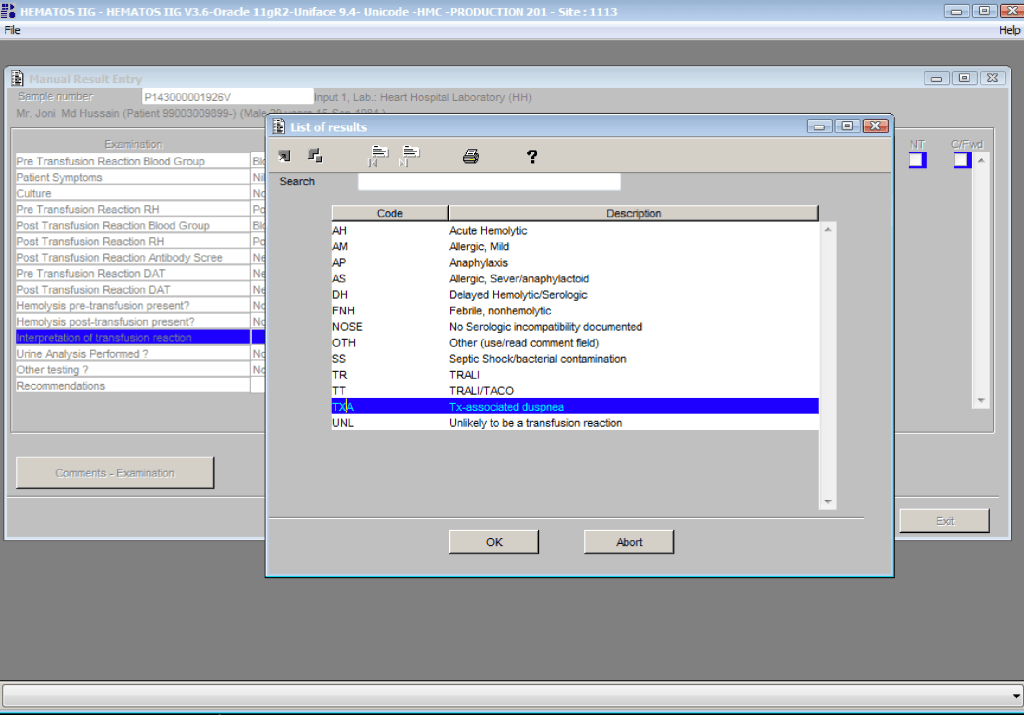
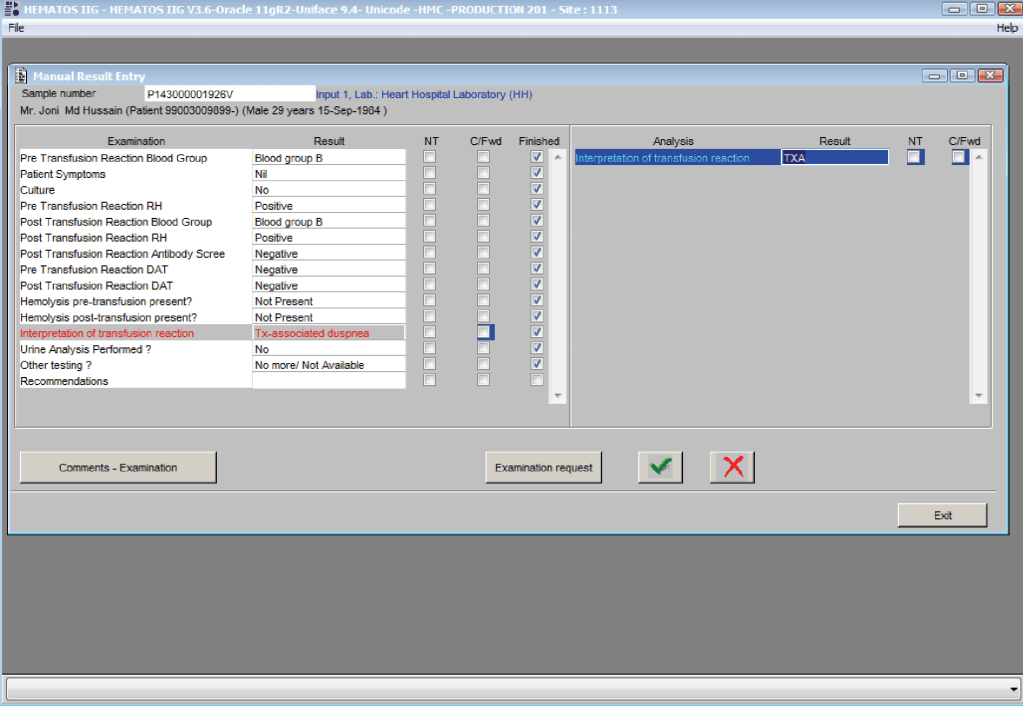
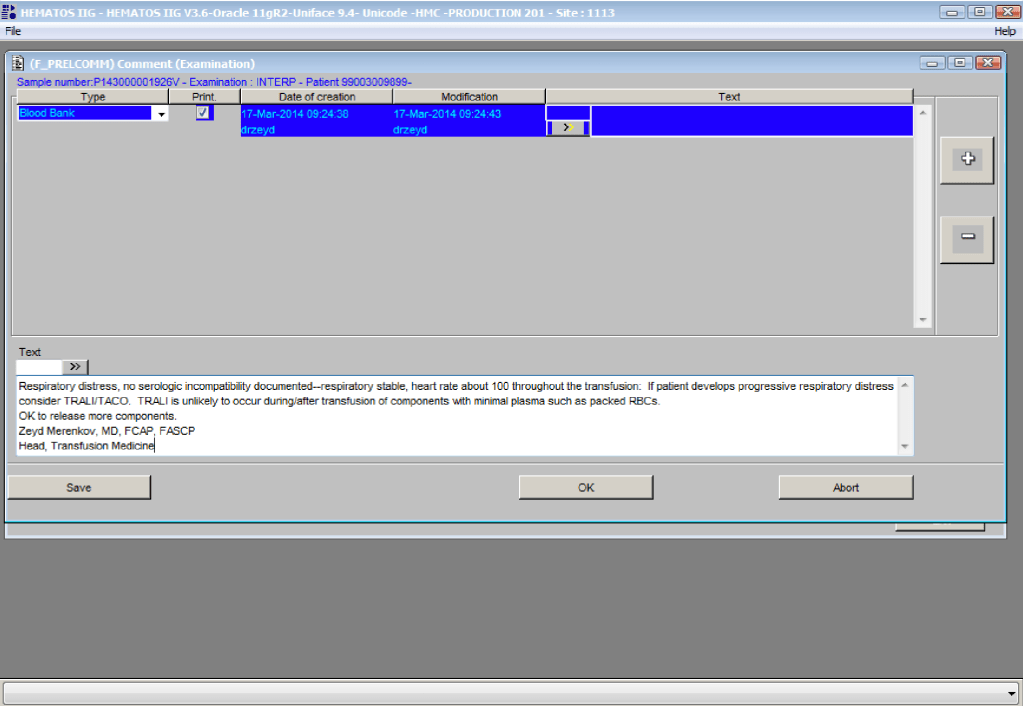
You can get the values off the therapeutic apheresis machine, but in the middle of the night when you have to write orders, it is convenient to estimate the volumes (whole blood, plasma, RBCs). These are the values from my lectures to hematology fellows while I was at HMC Doha:
Whole Blood:
Weight in kg X 70 ml/kg = whole (whole) blood volume adult
Weight in kg X 85 ml/kg =whole blood volume for child (prepubertal)
Weight in kg X 100 ml/kg = whole blood volume for neonates/premature
Example: 70 kg adult has 4900 ml blood volume (I round up to 5 liters)
Plasmacrit + hematocrit = 1.00 in fractions (100%), ignore buffy coat volume
Plasmacrit = 1- hematocrit
Plasma volume:
Plasma volume = whole blood volume x plasmacrit = whole blood volume X (1-hematocrit)
RBC volume:
RBC volume = whole blood volume x hematocrit
Estimates for blood components:
The volumes will depend on the original amount collected (e.g. 450 vs 500 ml), original preservative solution used (e.g. CPD), use of automated component production such as Terumo Atreus or Reveos, use of RBC additive solution (e.g. SAGM), leukodepletion, platelet additive solution, pathogen inactivation.
At HMC Doha, the average values were:
300 ml for leukodepleted RBCs in SAGM prepared by Reveos
300 ml for platelet pools in Mirasol and platelet additive solution (residual WBC < 1E6)
300 ml for plateletpheresis concentrate (2.4E11) in Mirasol and platelet additive solution
250 ml for leukodepleted, pathogen inactivated plasma