



Immunohematology testing and processes



Once the blood component has left the hospital blood bank, it should be directed transported to the patient’s bedside for immediate transfusion.
At the bedside, the transfusionist (usually a nurse or doctor) must verify the information on the blood component labels (both the ISBT and specific reservation one for the patient). With a bedside device, this includes:
All of these parameters can be entered into a hand-held device that transmits them to the blood bank computer system (e.g. Medinfo Hemotrace.)
The Blood bank computer system (patient module) should verify that this is the proper unit for the intended recipient and that the transfusion is starting within the reservation limit of the unit and serves to verify the information on the reservation label attached on the back of the blood unit.
With a device such as the new Umana T1 device from the GPI group, the vital signs together with an EKG lead and oxygen saturation can be automatically uploaded into the handheld device that feeds into the blood bank computer system such as Medinfo Hematos IIG and/or directly transmitted to the blood bank software. This device can continuously record this information for several days and detect post-transfusion adverse effects such as TRALI/TACO and delayed hemolytic transfusion reactions.

Note:
Some other nursing hand-held devices are available for recording patient data but many cannot read the ISBT unit number or descriptor information. They also do not check with the blood bank computer system to check if the right unit of blood is being offered to the patient during the validity of the transfusion period (four hours after leaving the blood bank).
Note: This is an updated version of a previous post.
Principle:
Washing RBCs removes plasma and reduces the leukocyte count only by 1 log. For leukodepletion, we must rely on filtration to reduce the WBCs to less than 1 x 106 per unit according to CE rules. Red cells or platelets in additive solution contain only minimal plasma (about 35 ml). There are few definite indications for washing RBCs and it should be rarely necessary.
Policy:
Washing RBCs should only be done in the following circumstances:
Note:
Reference:
Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
In my opinion, the direct antiglobulin test is the most important concept that a transfusion medicine physician or technologist must understand in interpreting complex serology patterns.
Like all other testing, the DAT must not be interpreted alone but rather in the context of other laboratory and clinical results. Still, it is very important to understand the significance of the DAT in hemolysis.
The mere presence of immunoglobulin on the RBC surface does not necessarily mean severe hemolysis. The DAT strength increases with increased immunoglobulin coating of the RBCs but does not necessarily indicate how quickly the RBCs will be cleared. That depends on the class and subclass of the antibody, whether and if so, how avidly it fixes complement, etc.
One trick question I give in my lessons to staff is, “What is the clinical significance of a negative DAT?”
In my career, I have seen severe hemolysis with either negative or weak DAT, the latter especially if there is weak C3 staining. The DAT can be negative because there is no significant antibody OR there is a highly destructive antibody causing massive hemolysis, leaving only the antigen-negative cells (and in that case, there is still the possibility of innocent bystander hemolysis).
I show them the following case of an ABO-incompatible acute hemolytic transfusion reaction:
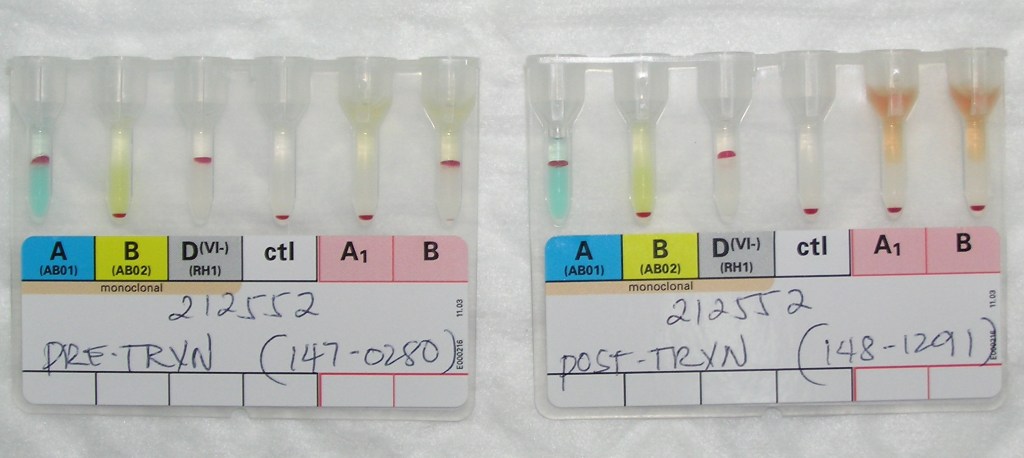
In this case, a group A patient received a group B RBC unit intended for a patient with a similar name who was group B and was in a bed next to him.
Notice the patient’s complete loss of the reaction to group B cells in the reverse type and the supernatant hemolysis. The DAT was negative. The transfused B cells were not even present in the post-transfusion gel.
Here is the urine specimen from that case showing gross hematuria:

So, in severe, life-threatening hemolysis, even antibody-mediated, you may have a negative DAT. DAT negativity may also be seen in non-immune hemolysis. I will discuss causes of DAT-negative hemolytic anemia is a future post.
In summary when doing a hemolysis investigation, a negative DAT does not mean everything is all right. Everything must be interpreted in the context of the clinical and other laboratory findings.
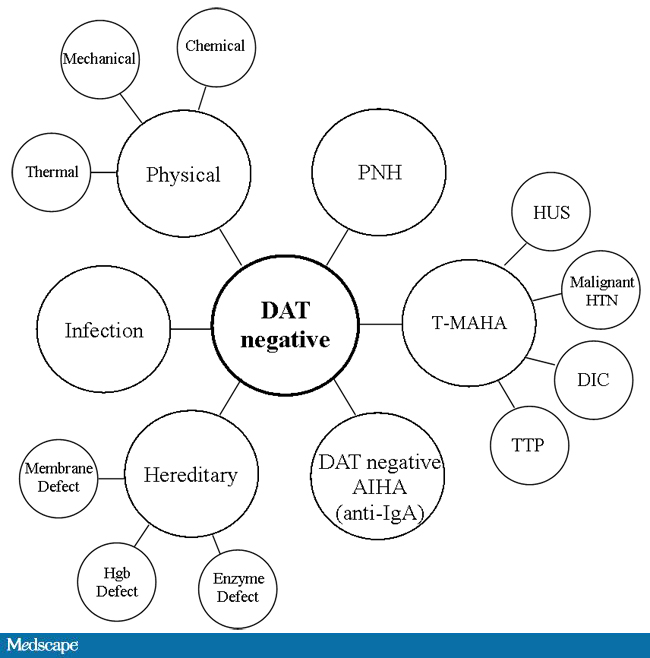
This is a summary diagram for the causes of DAT-negative hemolytic anemia, both immune and non-immune. For immune cases, a negative DAT may indicate “not-detected” with the amount of immunoglobulin on the RBC being below the threshold of the test methodology being used. Also rare IgA associated cases will not be detected unless a specific alpha-heavy chain monospecific reagent is used.
In my recent post, I provided sample flows and parameter mapping for delivery of blood components. The final components from the component preparation center may be sent to various depots (freestanding location and/or hospital blood banks. There should be complete traceability for every step (from donor reception, collection, testing, and processing) transport between locations, and finally the exact storage site, which might include which refrigerator/freezer/incubator and even shelf/position number for each component is stored. The end of that document showed rules for type/antigen matching.
For disaster planning, rapid inventory enumeration by type is very important. This can be very time-consuming manually. With our Medinfo Hematos blood bank system, we could quickly get total inventory across the Qatar or by hospital in less than one minute. We could also quickly find antigen-matched units across the system and reserve it at any one site for another if necessary.
Smart blood bank dispensing refrigerators, as offered by Haemonetics and Angelatoni, may also serve as depots and take the place of a hospital blood bank for some dispensing. These solutions can also capture vital information about the storage conditions of the components and prevent release if the storage criteria are not met. They can also interface with blood bank computer systems and use the main system’s logic for the dispensation rules. In Medinfo, they can be added as a hospital blood bank site.
Upon receipt at the hospitals from the blood processing center, the forward ABO and D typing must be confirmed. We used D reagents which detected partial D so we would call such donor units as D-positive. However, if a patient type reagent insensitive to partial D types is used, it is possible for a unit to be typed as D-negative whereas in the donor center it might be D-positive. Sometimes, nothing types consistently as D-positive: all you can say is that with a particular reagent and lot number, there is or isn’t reactivity.
The greatest complexity is for RBCs since potentially so many antigens exist. Criteria for matching/ignoring certain antigens must be made. Critically significant antibodies such as the Kell, Duffy, Kidd, and certain Rh (D and c) must be antigen matched. A robust blood bank computer system can enforce these rules.
For other components, antigen/typing may be less important. In fact, in most situations, any type of platelets can be given to anyone (except neonates). Despite the potentially incompatible plasma, there is rarely significant hemolysis. In fact, if pooling platelets without regard to blood types is done, a platelet transfusion is a common cause of a positive direct antiglobulin test DAT—something that is not clinically significant. No one died of a positive DAT by itself for this reason.
Specific rules for compatible plasma types are important, but nowadays, low-titer group A plasma may be used like universal AB plasma. The challenge is to be able to perform the ABO titration (specifically anti-B) quickly—titration can be a slow process, even with automated equipment. A similar situation for low-titer, universal group O whole blood requires both anti-A and anti-B titration (I will return to this topic in a future post). With Medinfo, I can define rules (e.g. IgM titer < 1:64) to accept these units as a universal type for all ABO groups.
Special rules can be built into the software so that production, transfer, storage, and release of COVID convalescent plasma CCP are only performed at special quarantine sites by designated personnel. This means there can be dedicated transport pathways built into the inter-depot transfer process to keep this inventory separate at all times.
I prepared the following plan for a CCP program for HMC Qatar in March, 2020. The workflow is divided into four (4) modules:
Module 1:
Module 2:
Module 3:
Module 4:
Workflow Considerations:
Logistics:
Information Technology:
When I first moved overseas from the United States, I brought the perspective of my American training and experience. I saw everything in my new blood bank through those eyes.
Yet, most of my staff were not American or even North American. Few were even native in English, and most of those were not American. They had different qualifications, many of which would not have been accepted by the American schemes. Still, they functioned well.
I also worked with the US military technologist staff during Gulf War One. Some did not even have a Bachelor’s degree; yet, they performed the work well.
I used many technologies that were not yet (or never) US FDA approved such as gel or glass bead typings and pooled buffy coat platelet production. There were rare reagents I could buy off the shelf (e.g. anti-Tja/PP1Pk).
Later, I adopted pathogen-reduction technology (Mirasol), automated component production (Atreus then Reveos), and platelet additive solution. I achieve a level of good manufacturing practice that would have been difficult to achieve by the FDA-approved methods.
My perspective had changed. In the Middle East, I studied many frameworks and came to the conclusion that the best approach was to customize them to our local needs. My particular experience was to start with one framework, i.e. Council of Europe CE, and then localize it.
To do this, I could not use an American turnkey blood bank software for either the donor or patient operations. I needed a flexible system that could be customized to my needs. Again, I chose a CE-marked system, Medinfo Hematos IIG that had already been adapted to many frameworks.
It is much easier to work solely within one system such as FDA. However, if I had done that, I would have lost so much flexibility and not had a system optimized for local conditions. I would not have used Mirasol, Reveos, Diamed, and many other reagents.
One big disappointment at such international meetings is the perspective by one country’s regulatory agency that they feel its regulations and framework will work well overseas. I would wager that those people were not well acquainted with international conditions.
Another frustration was attending another international meeting in which the presenters apologized for the source of information since it came from a foreign country (France) and not their own (United States).
No country has a monopoly on what is best for everyone. To share our experiences and compare is so valuable. No one assume his way is the best. In my career, I have had the richest experiences studying other perspectives and my organizations have benefited greatly from the exchange. We can all learn from each other. We are citizens of the world.
Principle:
Kidneys have strong expression of ABO type and must be matched the same way as RBC components. In the case of ABO-incompatible renal transplants, we must not give significant amounts of plasma incompatible to the ABO type of the donor kidney. Plasma must also be compatible with the patient’s ABO type for RBC transfusions. The amount of residual plasma in PRBCs is limited since we use an additive solution SAGM. Likewise, platelet components are suspended in platelet additive solution with only minimal residual plasma. Cryoprecipitate has only minimal plasma and is given without regard to the patient’s ABO type.
All of the following rules can be built into the blood bank computer system Medinfo without hard coding.
Policy:
References:
Setting up ABO-incompatible renal transplants is a major undertaking and requires close coordination between Transfusion Medicine and the clinical team. This post addresses my experience in setting up this program in 2019 at HMC in Qatar.
Like any process involving titration, it is best to automate it to minimize inter-technologist variability. Unfortunately, doing both IgG and IgM titers takes up to 1 hour per machine and totally monopolizes the machine during that interval. I did not have sufficient staff to even consider doing the titrations manually. Performing automated titers disrupted my workflow so I encouraged the clinicians to send the specimens for off-peak processing.
Titration:
Columns:
Apheresis Equipment:
Staffing:
Specimen Collection:
Table of Permissible ABO Types:
Software:
Selection of blood component for ABO-incompatible renal transplantation is discussed in a separate post that will follow.
25/12/20