This is an update of a previous post.
Principle:
In 1984 effective with the 13th Edition AABB Standards, the requirements for performing a direct antiglobulin test and autocontrol for compatibility testing were eliminated. The DAT is very important to detect delayed hemolytic transfusion reactions, certain autoimmune conditions, and drug-related hemolysis.
Since that time, the immediate-spin crossmatch and now the electronic computer paperless crossmatch may be used for most compatibility testing in place of the classic, antiglobulin-phase (indirect antiglobulin test) crossmatch.
If an antiglobulin phase (IAT) crossmatch is performed, an RBC unit with a positive DAT will cause a false-positive reaction. Since most crossmatching does not include the IAT, it will not be affected by the DAT status of a donor unit.
Policy:
- Donor RBC units will NOT be routinely tested for DAT as part of component processing.
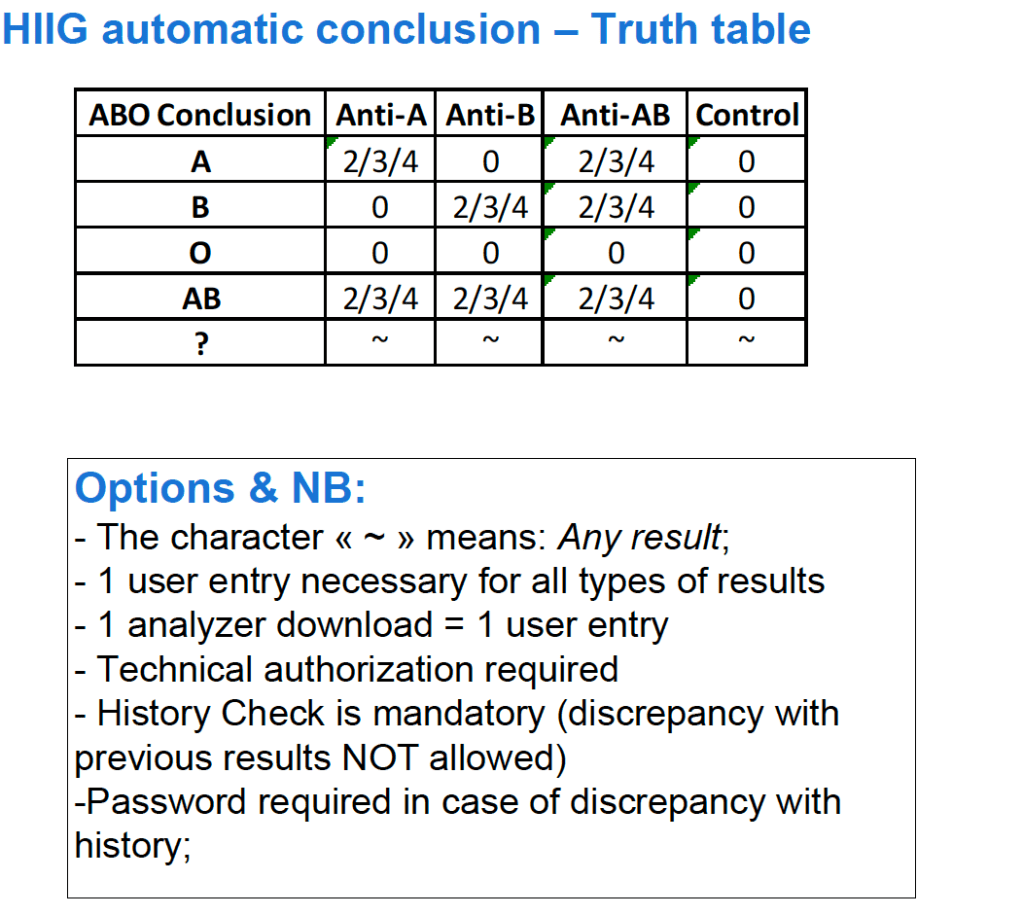
- The type of compatibility testing selected for a particular patient should be the technically simplest one (no need to do extra work unless so instructed by the transfusion medicine consultant/designate):
- Do a full antiglobulin-phase IAT crossmatch if ANY of the following applies:
- There are no two independent ABO/D typings on the patient during the current admission.
- The ABO/D type of the current admission does not match the historical information.
- The patient has a detectable antibody at 37C
- The patient has a history of a clinically significant antibody but no current antibody
- Whenever the consultant, transfusion medicine/designate requests it.
- Whenever the Medinfo HIIG record so indicates (in comment section)
- Do the immediate-spin crossmatch if ALL of the following apply:
- Only one determination of the ABO/D type
- The historical ABO/D type agrees with the current type.
- There are no antibodies reacting at 37C AND there is no history of antibodies at 37C.
- Use the computer/electronic crossmatch if ALL of the following apply:
- There are two determinations of the ABO/D type and they both agree with each other.
- The historical ABO/D type agrees with the current type.
- There are no antibodies reacting at 37C AND there is no history of antibodies at 37C.
- When to do a DAT on a donor unit:
- Patient antibody screen is negative but the full AHG crossmatch is incompatible.
- Part of a transfusion reaction workup where the AHG crossmatch of donor cells and patient serum is incompatible.
- Whenever the consultant, transfusion medicine/designate requests it.
- If a donor unit is found with a positive DAT:
- Required testing and review:
- Test with polyspecific and monospecific IgG and C3d antisera
- Perform an acid-elution.
- Send the results to the transfusion medicine consultant/designate for review.
- The reviewer will enter his review in HIIG in the Donor Consultation Section both as global donor comment and a result-specific comment against the antibody screen result.
- Use of the DAT-positive donor unit:
- Most of the time you will not know if the donor RBC unit is DAT-positive since we mainly use the electronic crossmatch. It will be used if all criteria are met.
- Otherwise, select another RBC unit for the AHG crossmatch.
- The final decision to use the DAT-positive unit will be made by the Transfusion Medicine consultant/designate.
Important: Don’t do a classic AHG/IAT phase crossmatch unless you have to do it (see conditions above.) A donor unit with a DAT is unlikely to be clinically significant and may be transfused safely to the patient in most situations. Patients receiving electronic-crossmatch and immediate-spin crossmatch are receiving units with positive DAT without incident.
References:
- Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
- Guidelines to the Preparation, Use, and Quality Assurance of Blood Components, European Committee (Partial Agreement) on Blood Transfusion (CD-P-TS), Current Edition
- Technical Manual, Current Edition, AABB, Bethesda, MD, USA, 2012