These are questions based on my medical student lecture on Community Practice of Transfusion originally given at National Guard Health Affairs in Riyadh:






Immunohematology testing and processes
These are questions based on my medical student lecture on Community Practice of Transfusion originally given at National Guard Health Affairs in Riyadh:





In the previous post, the Medinfo document for Inter-Depot transfer had many pages of rules for matching RBC antigens. Multiple actions were available:
To avoid mistakes, the blood bank computer system enforced the rules. There was no mercy. Only specific individuals could override this( in many cases, but certain allocations (e.g. group O RBCs to a Bombay Oh patient) were not permitted under any circumstances.
Prophylactic Antigen Matching:
Please also refer to my prophylactic antigen matching post made last week for the rules I selected for Qatar.
Prophylactic antigen matching is common in Europe. I have been doing this during the many years I have worked in the Middle East. Most patients were not local nationals but transient. They would return to their home countries where blood bank testing (antibody screening/identification/antigen matching) or intrauterine exchanges might not be always available.
For pregnant patients, we would prophylactically match K-negative and c-negative—regardless if there were antibodies detected—R1R1 units for R1R1 patients. At the end of my time in Qatar, we had several pregnant women with various Rh deletions, so we added routine extended Rh(D) and Kell typing to all.
For sickle cell patients, especially African type, I would prophylactically match Rh antigens (D, C, c, E, e) and Kell because of the polymorphisms in the CE gene, some of which may lead to pan-Rh antibodies.
I would consider selective prophylactic antigen matching in chronically transfused populations, again regardless if clinically significant antibodies were detected.
If a patient makes any antibody, regardless if is clinically significant or not, I would consider that patient as a candidate for prophylactic antigen matching (but NOT necessarily for a clinically insignificant antibody).
In Qatar, blood bank services (testing and components) were not charged to the patient. In many other parts of the world, blood bank is a cost center. No prophylactic antigen matching may be routinely performed. If it is done, it must be charged to the patient or the hospital must assume the cost. I have gone to conferences in such locales where not even R1R1 patients were not matched and subsequently developed anti-c, which complicated management. It would have been cheaper to do the antigen matching than to pay for the consequences of the alloimmunization.
This is a teaching case presented to the Medical and Nursing staff at one of my previous affiliations.













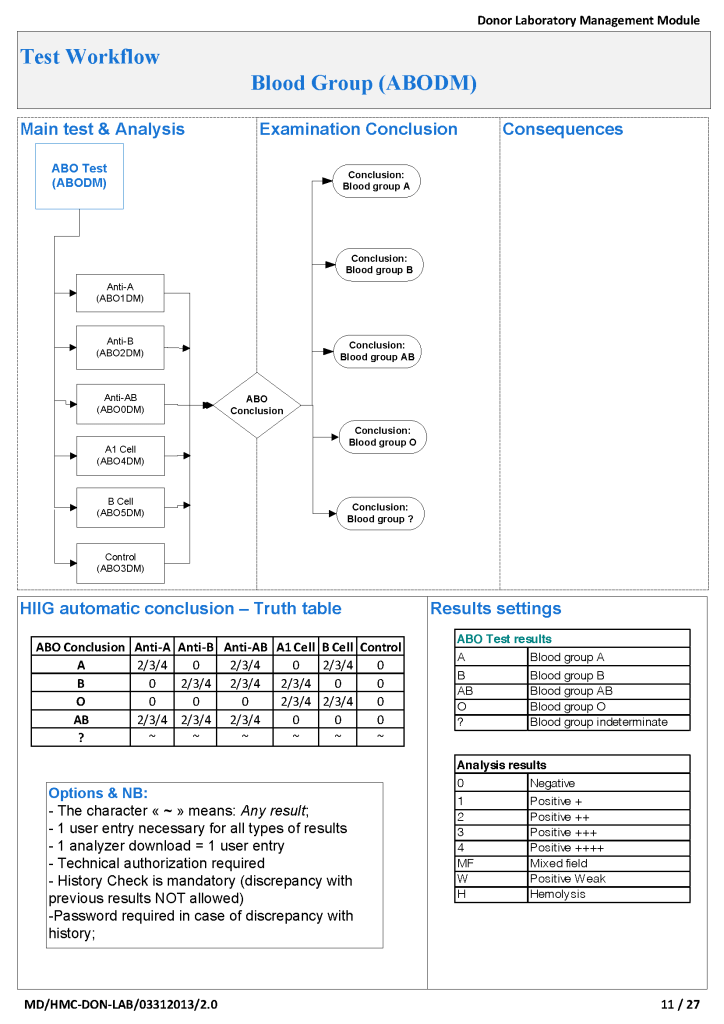
Medinfo Hematos IIG software is rules-based so the institution may set its own custom rules for all processes. One chooses a framework and then adds any additional rules it needs for optimization. Turnkey systems do not offer this flexibility.
The rules for platelet and plasma components are much simpler than those for RBCs since usually we only consider ABO type. There are two modes: regular and emergency, the latter applying if not all the patient testing (including historical checking) is available. The components, on the other hand, must meet all criteria before being considered for patient use.
Please note that any donor with antibodies is automatically excluded from plasma and platelet production based on our donor testing criteria.
Example rules for plasma follow:


For platelets, note that for adults and anyone else >= 20 kg, I gave any type of platelet pool or plateletpheresis component without regard to ABO matching. With our production method, I did not give Rh immunoprophylaxis to females of child-bearing age receiving platelets from D-positive donors based on our clean (essentially RBC-free) Reveos automated production process.
For platelets, there were also different allocation rules for regular and emergency mode:

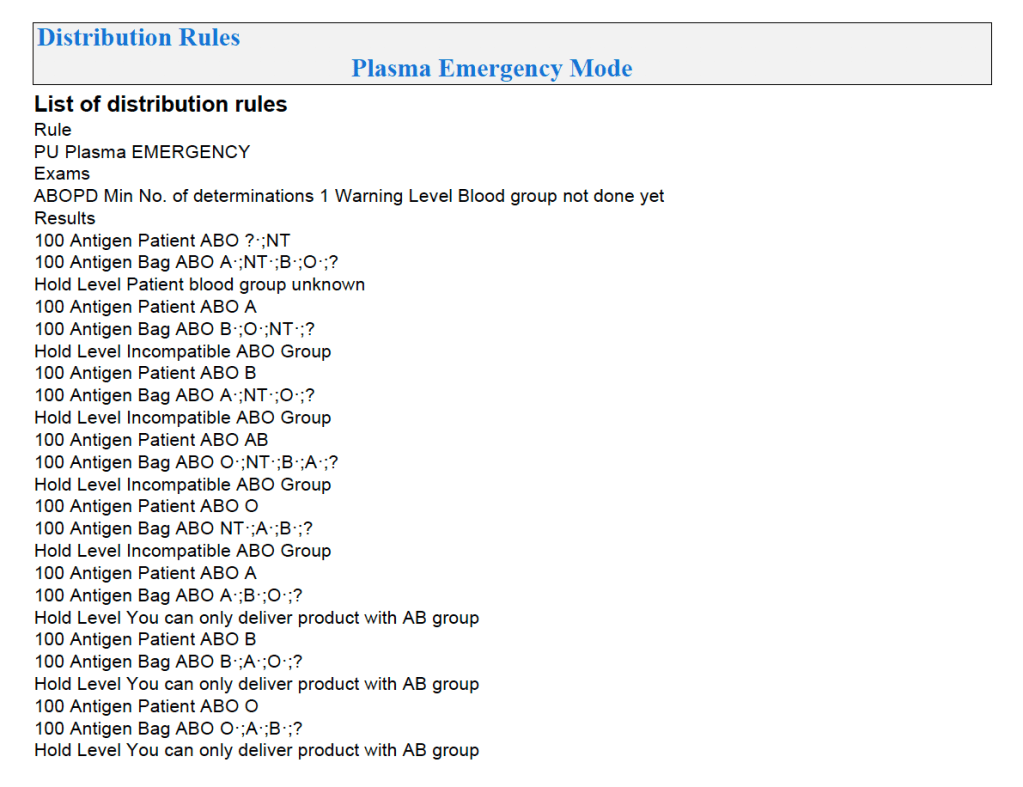
Similarly, allocation rules for granulocytes, etc. can be made and enforced by the software. Low-B-titer group A universal plasma would also be easy to implement.
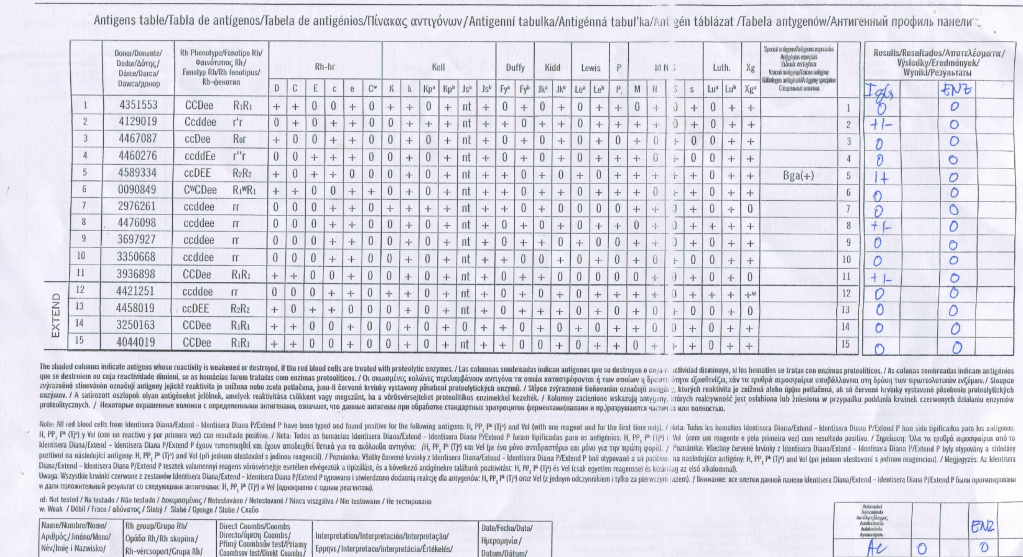
In my Middle Eastern experience, I have found antibody panels from a certain manufacturer show a lot of weak, nonspecific reactivity which disappears with another manufacturer’s panels, particularly when gamma-heavy-chain-specific reagents are used:
Normally, in my protocols, the first encounter in this situation, I would do extended RBC phenotyping beyond Rh and Kell systems as well.
In my experience, the nonspecific reactions detected are not clinically significant and they may be ignored if the conditions of the following algorithm is used:
What do you do when you encounter this is in your laboratory? Please share your thoughts.
Manufacturer 1’s Panel:
Manufacturer 2’s Panel:
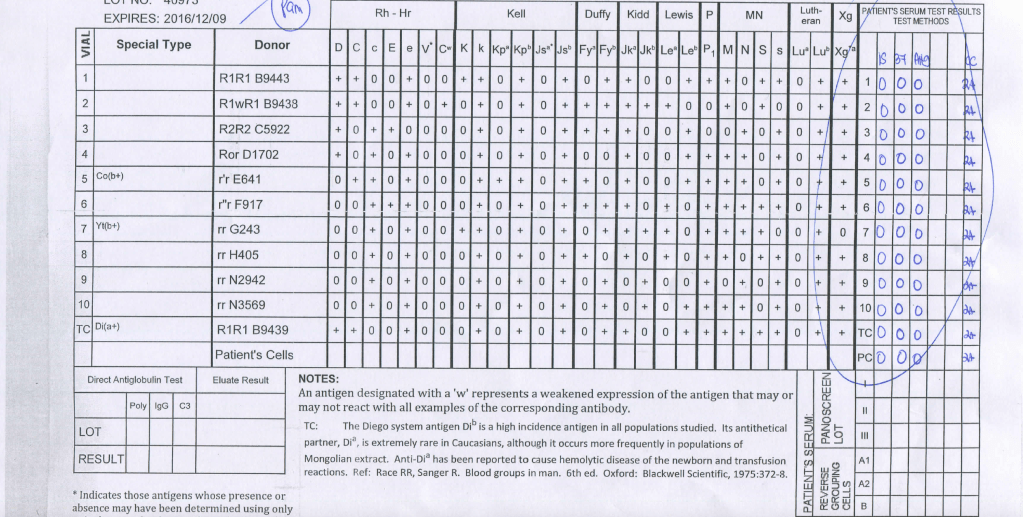
Note: This is an update of a previous post.
In the previous posts I outlined how Medinfo handled antibody screening and identification. This post reviews how antigen matching is used based on these results.
There are two modes, regular and emergency. If the patient has not had at least two ABO/D determinations and/or does not have a recent antibody screen within the past 3 days, then emergency mode must be selected with its own rules. Otherwise, the regular mode applies.
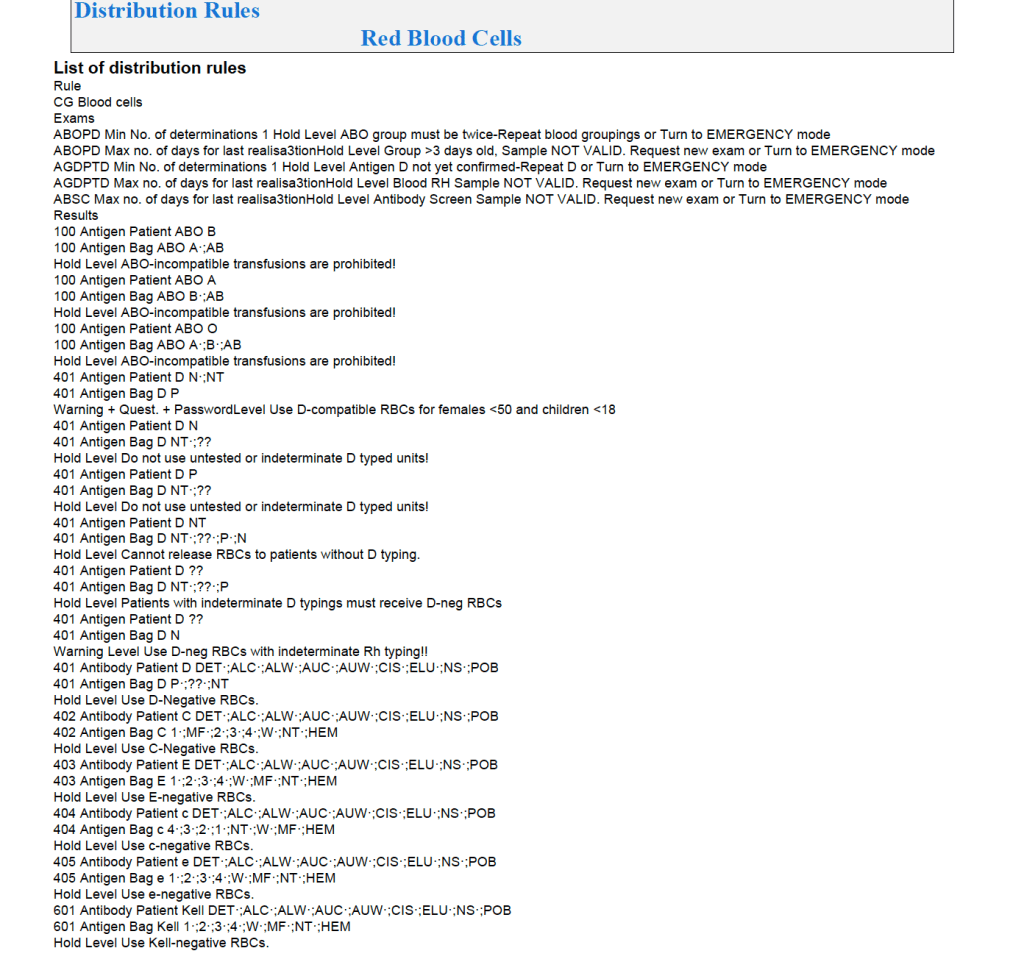
Regular Mode:
In general, if there is a clinically significant antibody, an RBC unit which has not been matched for the corresponding antigen or has the corresponding antigen cannot be routinely selected. However there is a hierarchy here also:
Examples of Regular Mode rules follow (these are not the complete lists but just provided to show the complexity of the process).

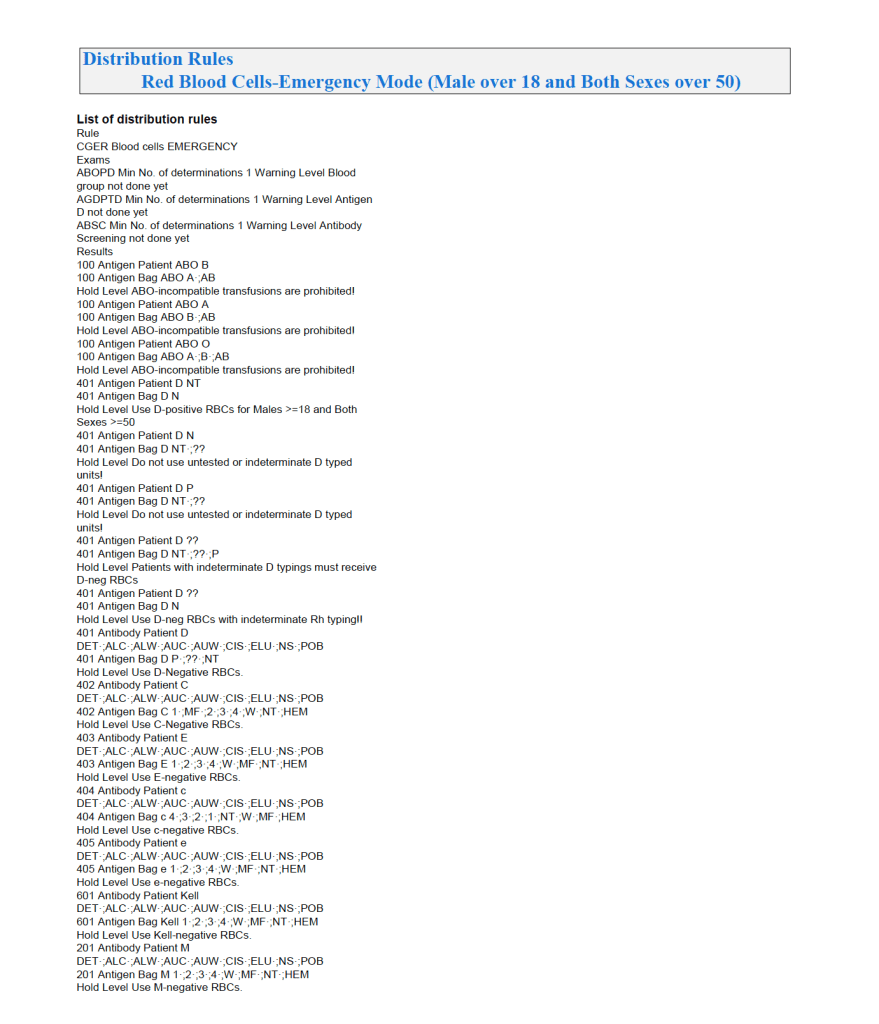
Emergency Mode:
This is much more restricted for selection of ABO/D and other antigen typings. An example follows:
If low-titer group O whole blood is available, then a specific rule must be added to both regular and emergency mode to allow this to be given to any ABO type except Bombay.
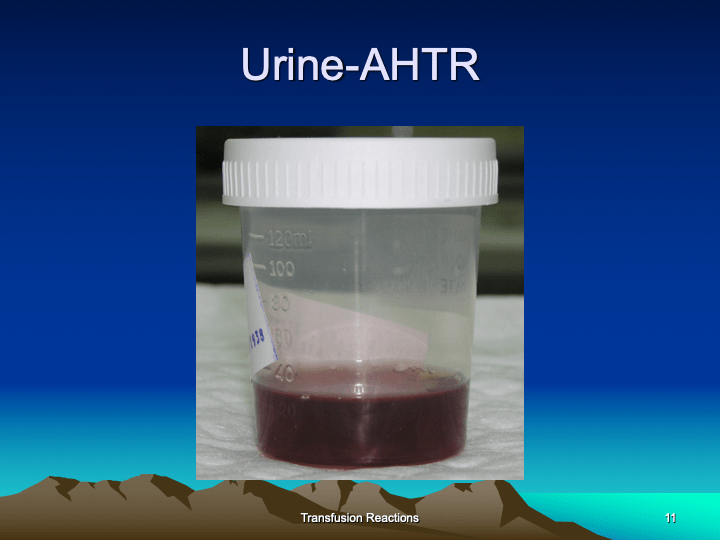
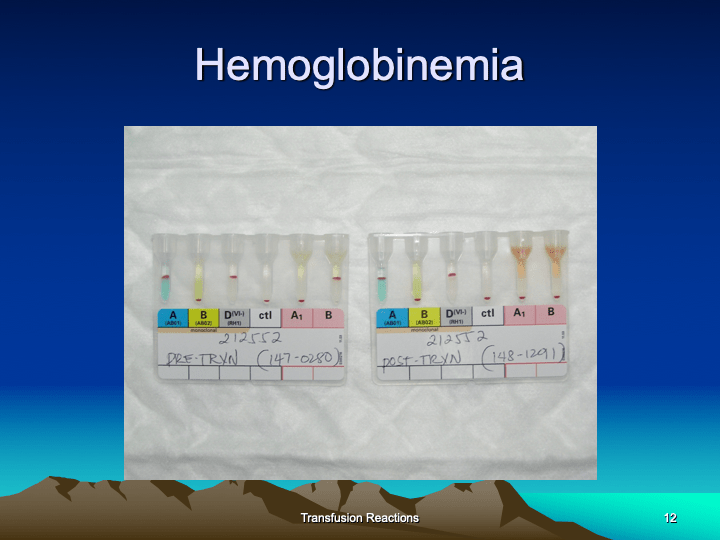
This is the third part of a lecture given to the general medical staff to help them appreciate the critical nature and stress of working in a tertiary hospital blood bank. It was originally given at National Guard Health Affairs in Riyadh.




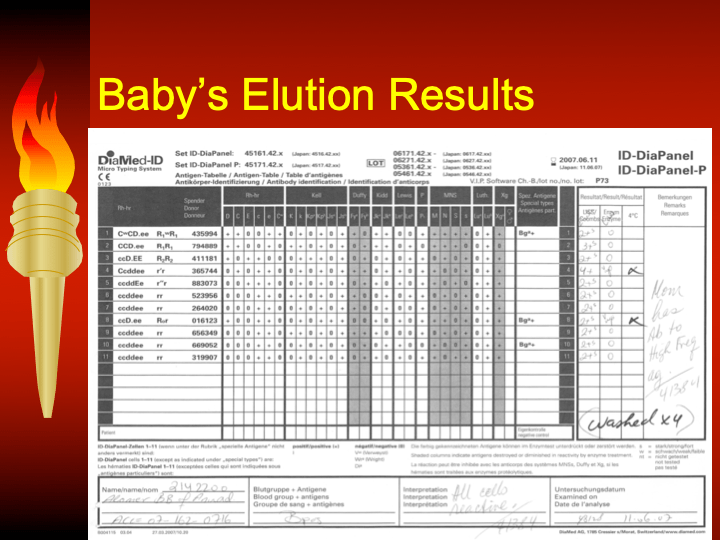











This is the second part of a lecture given to the general medical staff to help them appreciate the critical nature and stress of working in a tertiary hospital blood bank. It was originally given at National Guard Health Affairs in Riyadh.




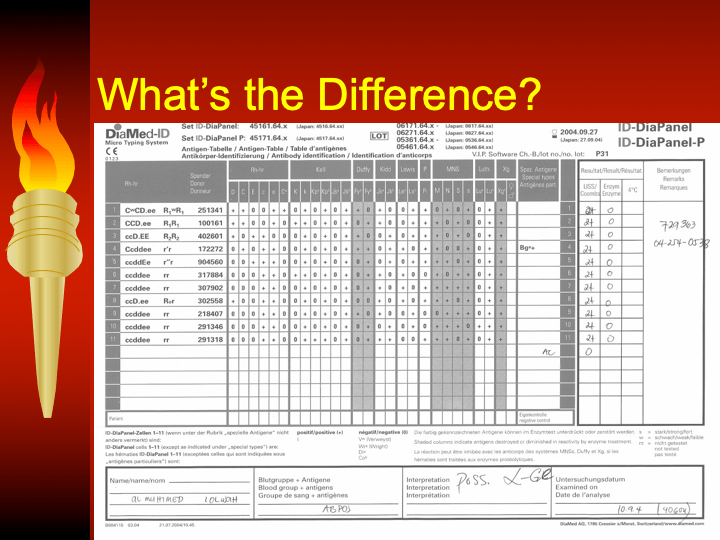






This is the first part of a lecture given to the general medical staff to help them appreciate the critical nature and stress of working in a tertiary hospital blood bank. It was originally given at National Guard Health Affairs in Riyadh.









