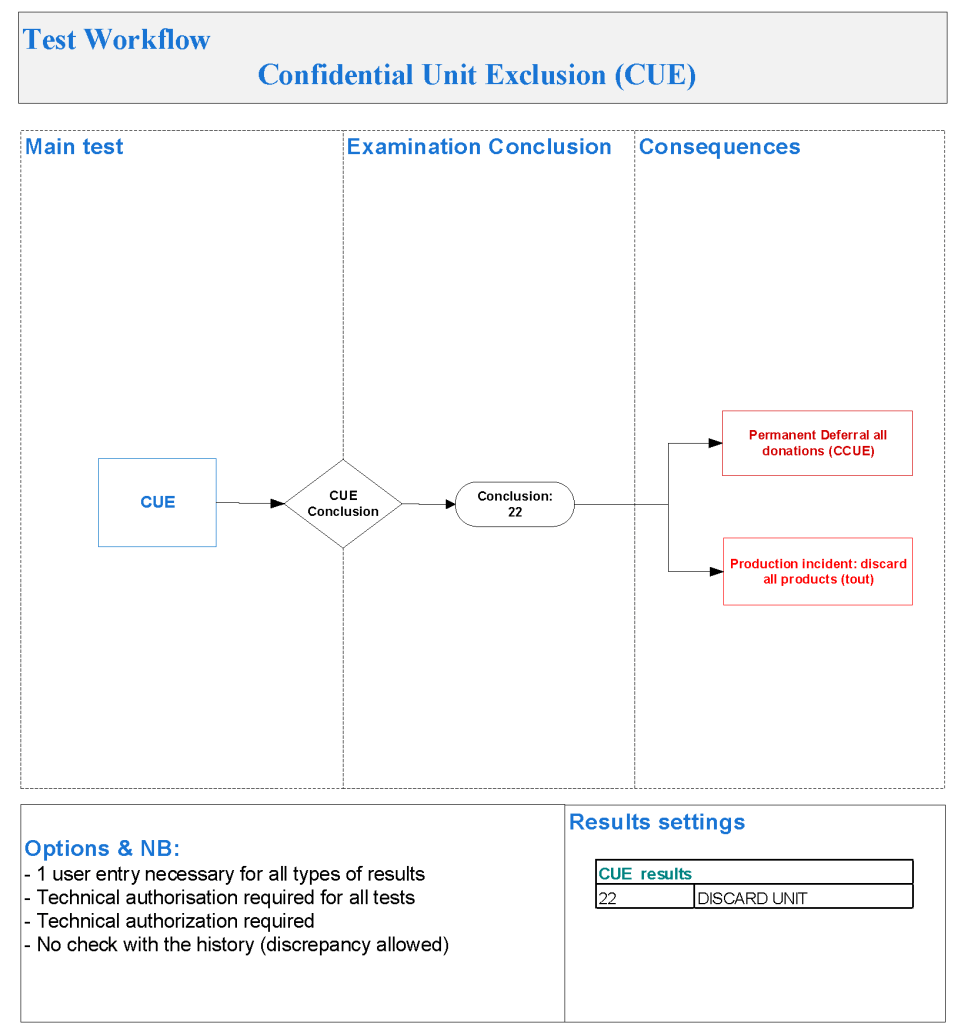
Principle:
All donor unit mislabeling is potentially life-threatening and must be stringently investigated as soon as possible after the discrepancy is detected. Most importantly, if there is one error, there may be possibly ADDITIONAL donor unit errors (e.g. switch of donor tubes or units, etc.). All donor units processed in the same batch must be also quarantined until the discrepancies are resolved.
The blood bank computer system will detect many errors; however, if the donor unit or its samples are mislabeled in the beginning, these may not be detected. Medinfo enforces checks on the final ISBT label and will compare current results to the historical record and will alert to any errors. Additionally, the use of ISBT specimen labels will obviate the risk of barcode reading errors.
Definitions:
Responsible blood bank physician: specialist or consultant physician on-call at the time the discrepancy is detected
Policy Details:
The following steps MUST be performed as soon as possible:
- The Component Processing Supervisor or Senior Technologist must be IMMEDIATELY notified of any discrepancy.
- The Blood Bank Supervisor will inform the Division Head, Transfusion Medicine. If the Head is not available, notify the Transfusion Medicine on-call.
- Quarantine ALL donor units collected and processed in the same batch.
- Obtain copies of all testing including photos of the gel/glass bead cards documenting the discrepancy.
- Obtain copies of all worksheets used in donor processing for the affected batch.
- Perform repeat ABO/D typing of ALL DONOR UNITS in the affected batch. Any further discrepancies must be investigated and resolved.
- Identify all staff who were involved in handling the donor unit (phlebotomist, blood bank technicians processing and labelling the unit). Identify those associated directly with the error.
- Submit all documents and photos to the Blood Bank Supervisor or designate.
- Prepare an occurrence/variance OVA report documenting all the data, findings, and interpretations.
- All investigations must be reviewed by the Supervisor, responsible blood bank physician, and one of the senior consultants.
- All such investigations must then be finally reviewed and approved by the Division Head, Transfusion Medicine or his designate. Only when the issue(s) are completely resolved and investigation is approved may the donor unit be properly relabeled and released into available stock. Also, only at that time may the other units in the affected batch be released into available stock!!
- Photograph the correctly relabeled unit and attach it to the other documentation of the incident.
- If the discrepancy cannot be resolved, ALL units in the affected batch must be discarded.
- The implicated staff’s personnel record should be reviewed for previous errors. Appropriate disciplinary action should be taken and documented in the personnel record. If a verbal warning is given, it should still be documented in the written record.
- If there is a systemic cause for the error, appropriate measures should be taken to minimize reoccurrence.
- All actions must be in accordance with the institution’s policies and regulations.
2/11/20