








Collection of plasma, platelets, RBCs, or combinations of them using apheresis equipment















Principle:
The Division Head, Transfusion Medicine and Blood Banks, is responsible for all aspects of transfusion medicine at HMC for the State of Qatar. The following policy documents some of those roles.
Policy:
References:
Standards for Blood Banks and Transfusion Services, Current Edition AABB, Bethesda, MD, USA
Donor physical examination, along with the donor questionnaire, are important both for donor and patient safety. In general:
Is it safe for the donor to donate?
Is it safe for the patient to receive the blood even if it is safe for the donor to donate.
Any donor who does not feel well must not donate. This may be the single most important step in ensuring a safe blood supply.
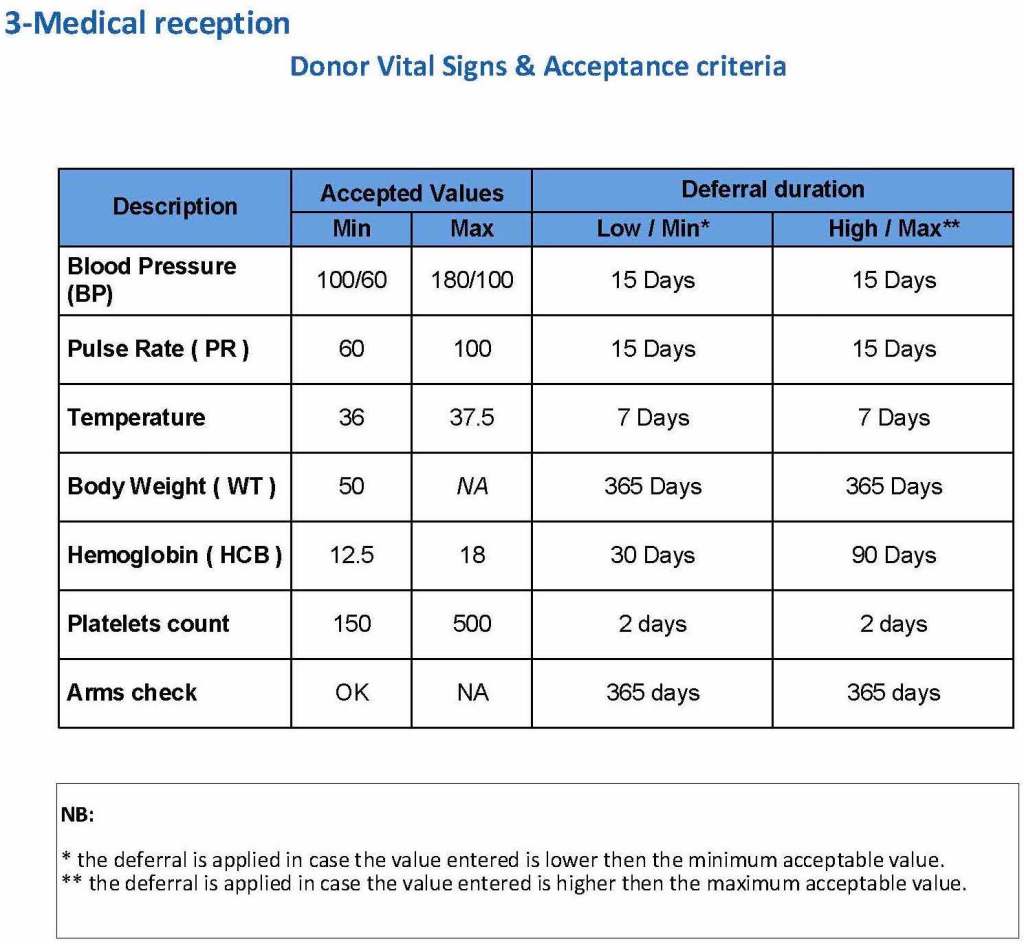
The donor physical examination includes the vital signs (blood pressure, pulse, temperature, heart rate, and temperature). I have attached a sample set of criteria for review. All are user-definable. Note how the arm examination is also included (looking for scarring, skin lesions, etc.)
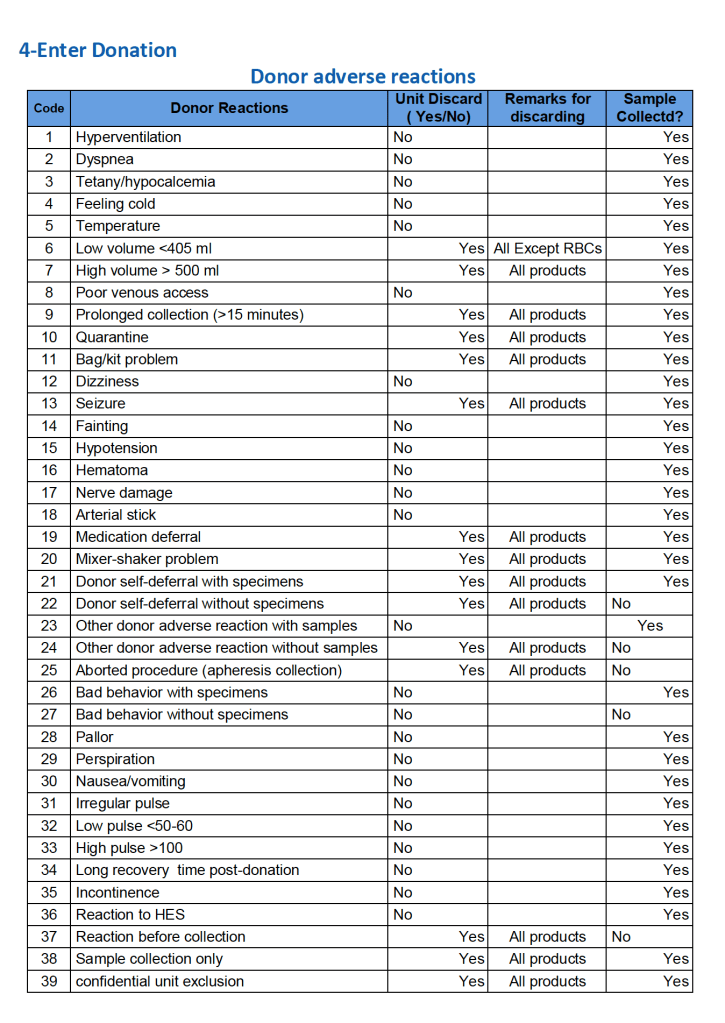
For all types of donations, there may be adverse reactions. These must be documented in the record along with the disposition of the donation. Will the donor need an extended deferral if the RBCs in the apheresis run are not returned? This can be built from the reaction documentation. Note the following sample table of reactions.
To Be Continued
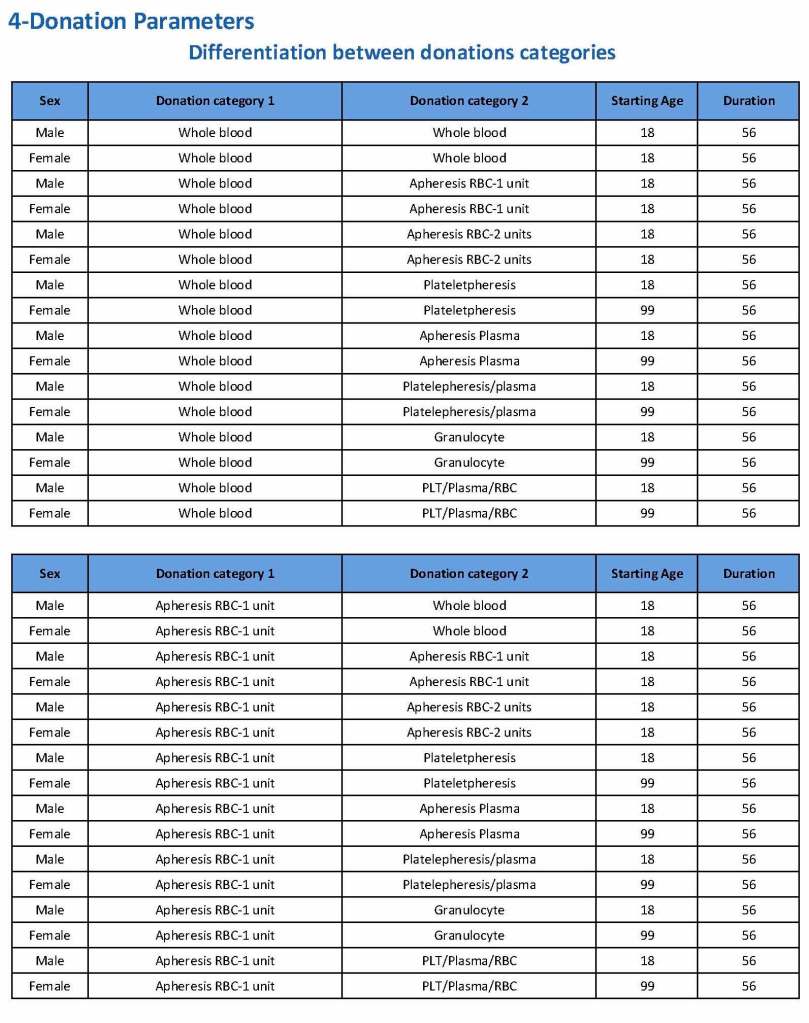
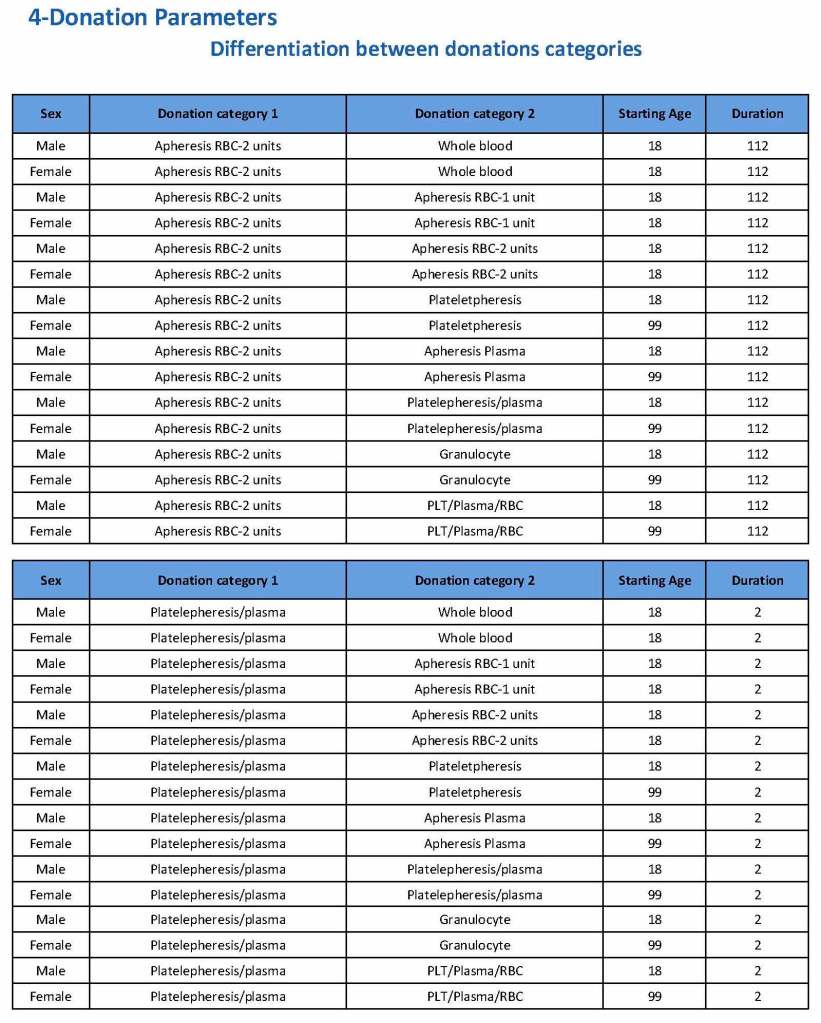
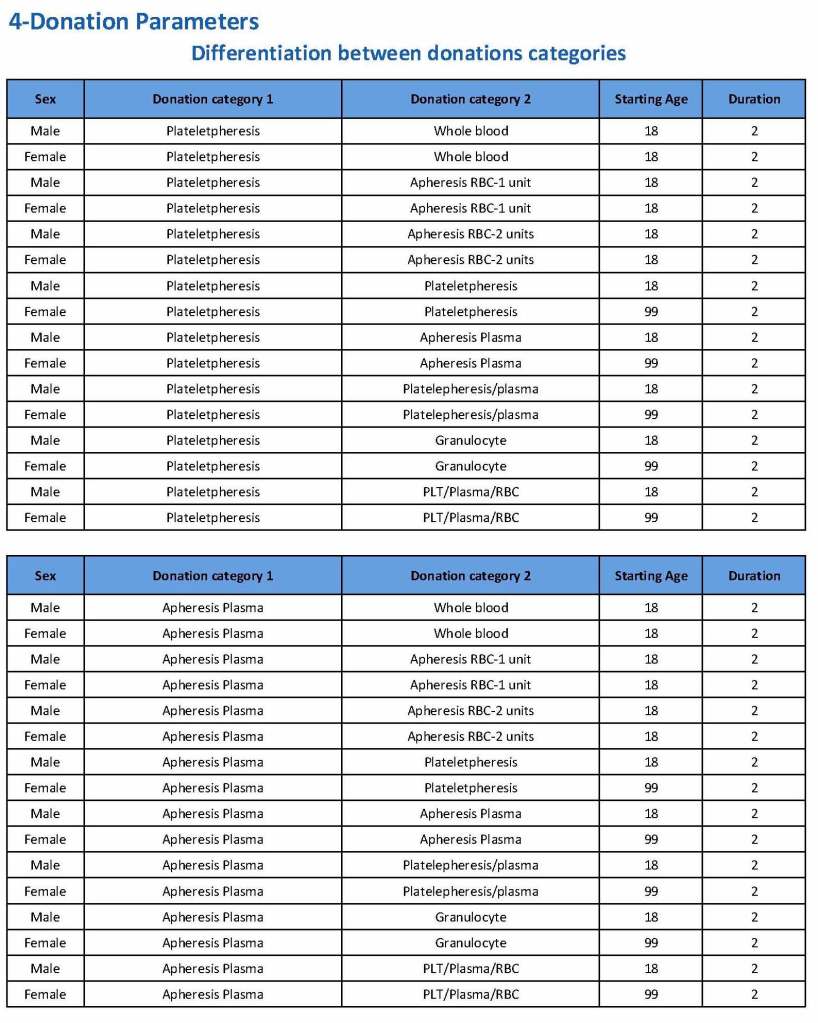
Donation can be whole blood or apheresis-based. The sex and age for each donation type is specified. At HMC, we did not accept females for platelet or plasma donations, so the starting age is listed as 99 years. Otherwise, in accordance with Qatari law, the starting age for donation is 18. All these parameters are user-definable, and a transfusion medicine physician can override the rules if necessary.
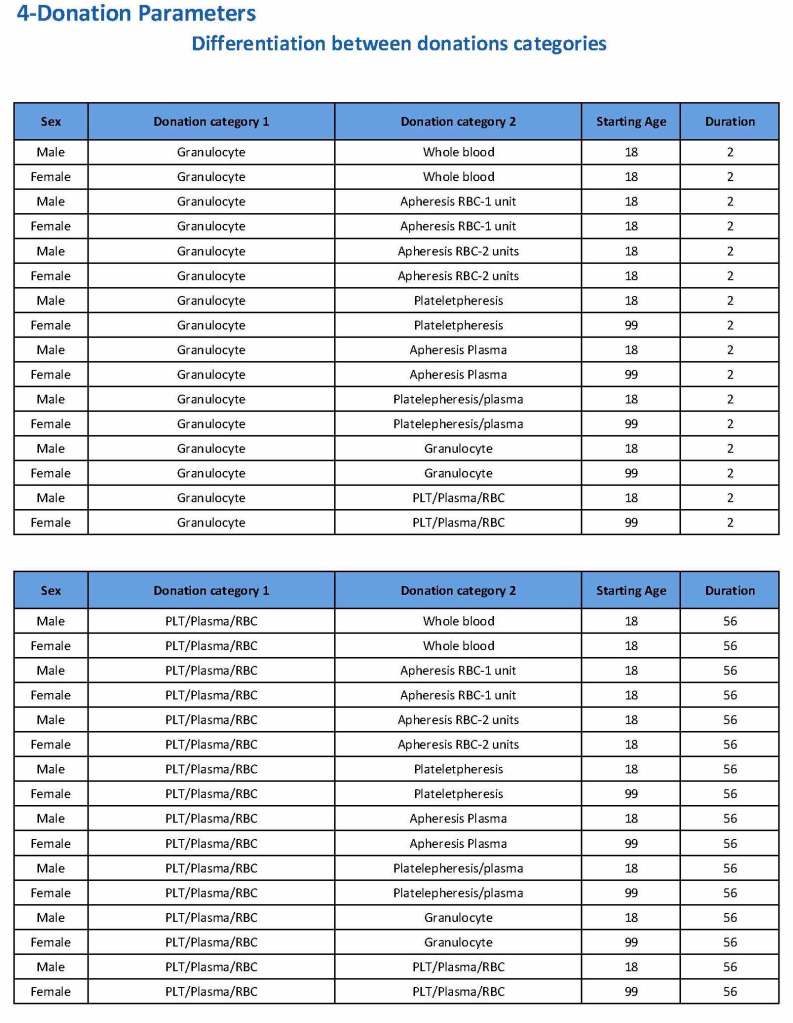
For each and every combination of donations, the deferral interval must be specified. Examples follow. The temporary deferral period is in days:
Previous donation whole blood, current donation whole blood: 56
Previous donor platelets, current donation whole blood: 2
Previous donation whole blood, current donation platelets: 56
Also note how for each possible combination there is an entry for male AND female. Females are restricted to whole blood donation and only RBCs will be made from the collection.
If there is a collection incident and the apheresis procedure is not completed, the interval will be set to 56 days. This will be covered in the post on donor adverse effect reporting.
To Be Continued:
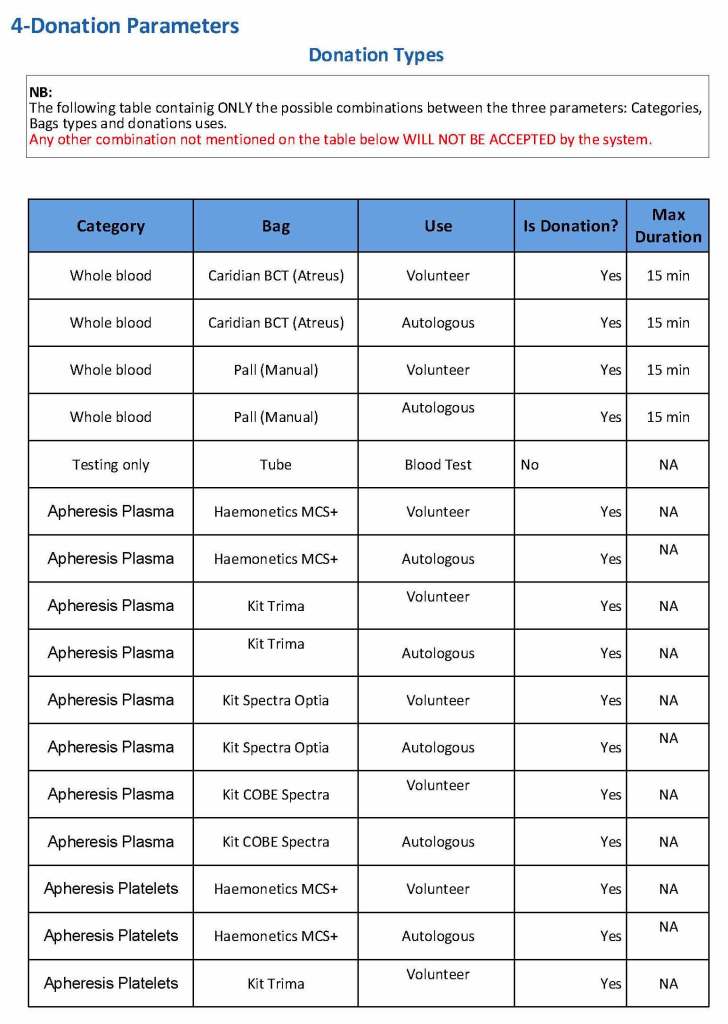
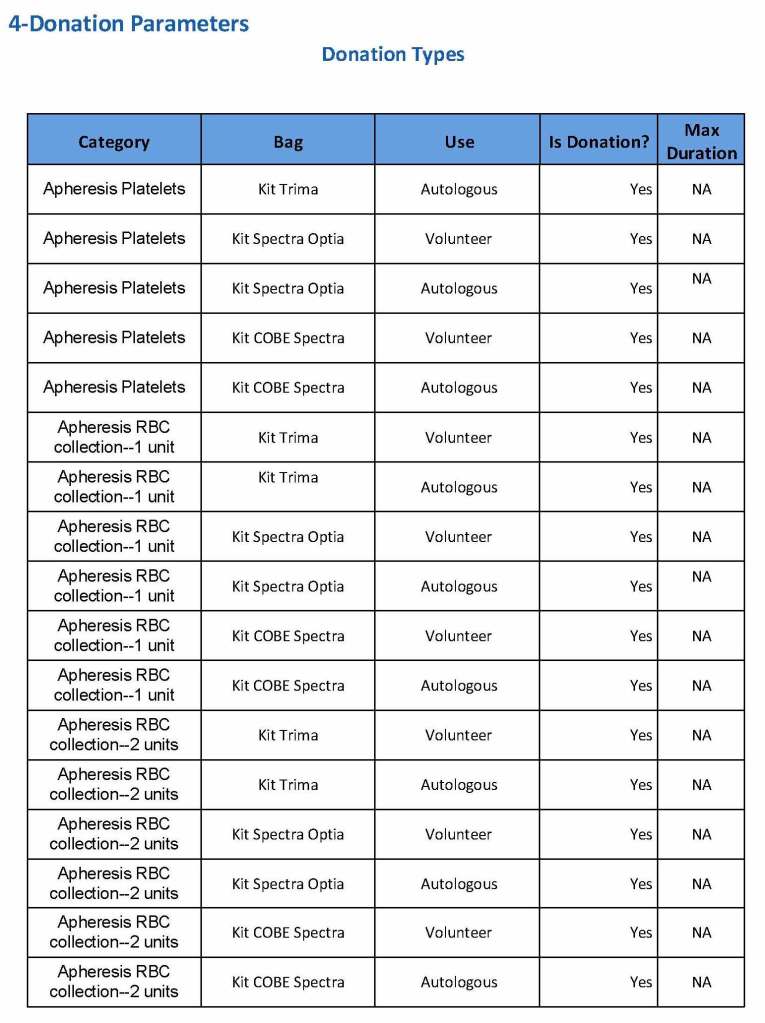
At the time of registration, the type of donation must be specified. In my last position, this could include whole blood for automated Reveos, whole blood for cryoprecipitate, plasmapheresis, COVID 19 convalescent plasmapheresis, plateletpheresis, concurrent platelet and plasmapheresis, concurrent platelet, plasma, and RBC apheresis, RBC apheresis-one unit, and RBC apheresis-2 units.
There is also a specimen-only donation without actual collection that includes database check, assignment of an ISBT specimen number, donor questionnaire, physical examination, and specimen collection only..
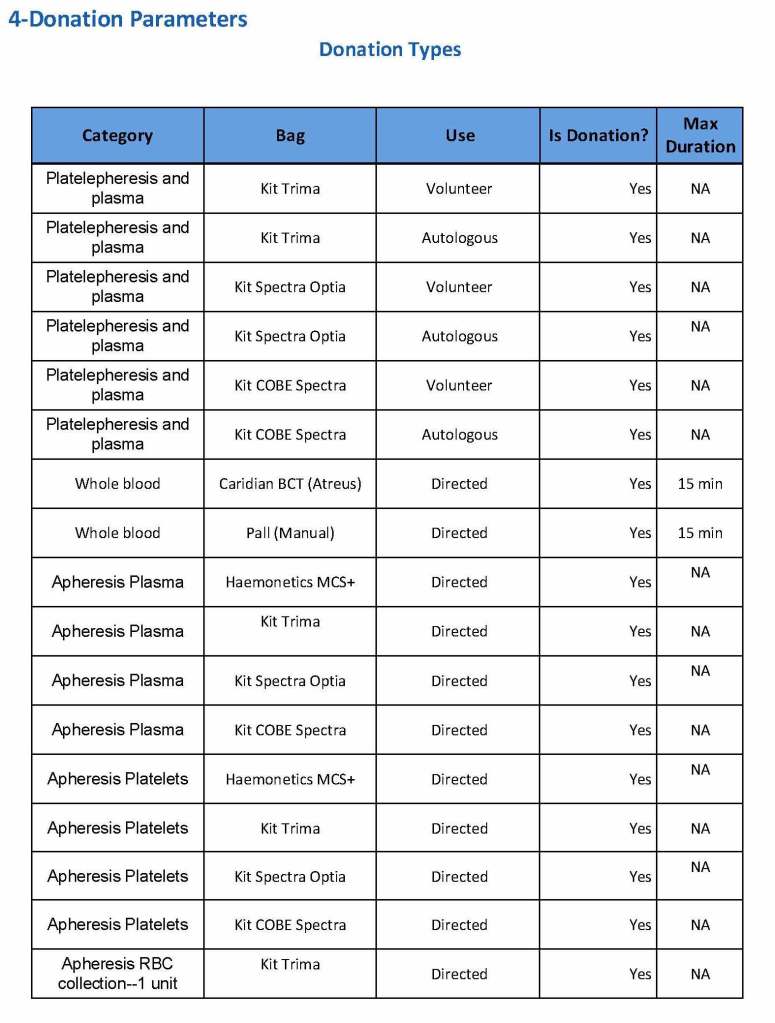
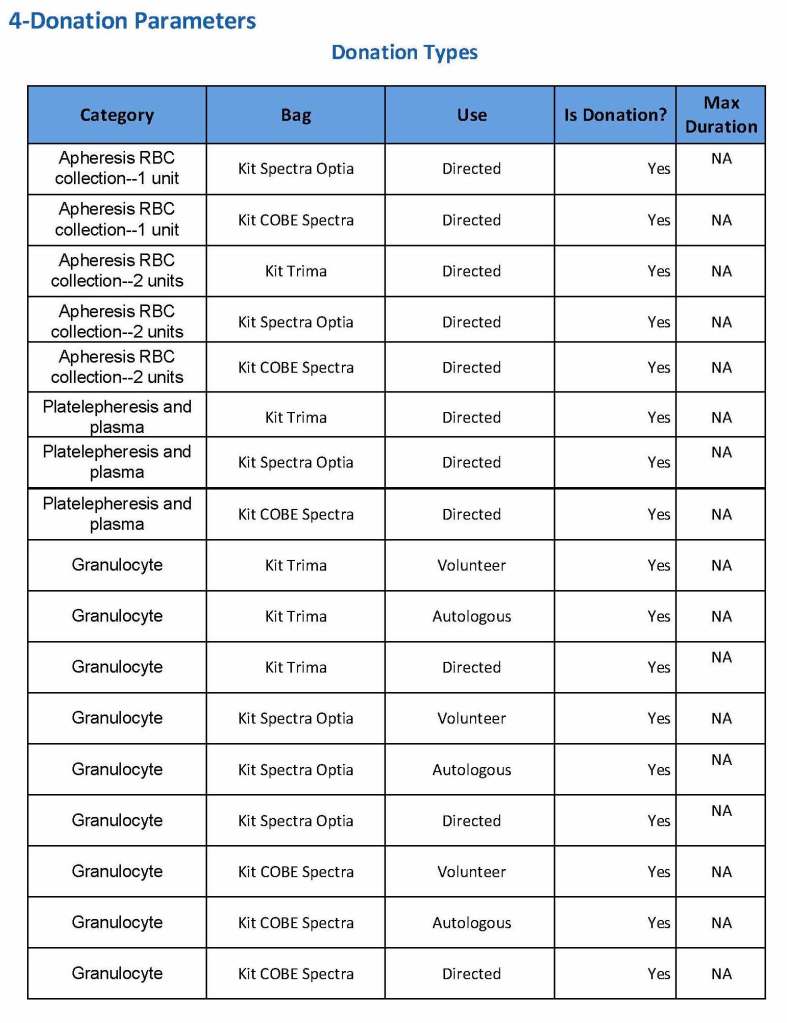
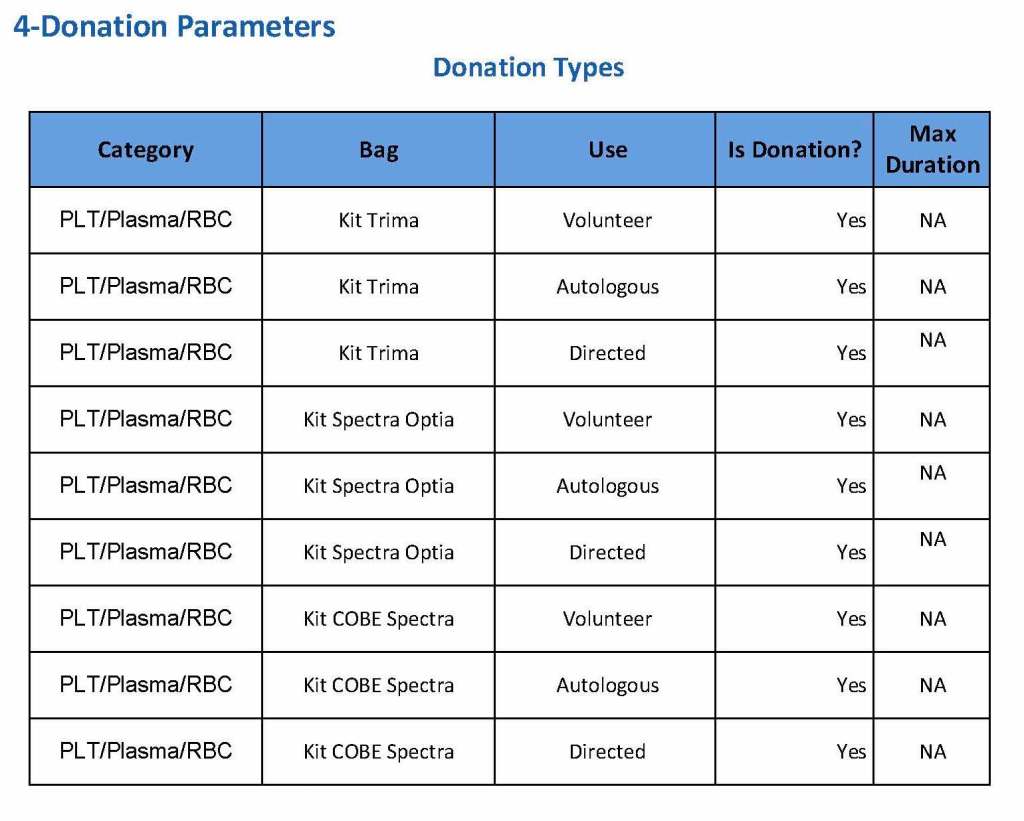
We specified which bag or kit could be used for each type of donation so when it was selected, only that bag type would be accepted by Medinfo
For each of these types we must specify what type of donation is permitted: volunteer, autologous, or directed.
Finally, we must indicate the maximum length of the procedure permitted. This applied to whole blood only and we set this at 15 minutes—this is user definable.
The following are a sample set of parameter settings for the above:
Note how we included contingencies for old bag sets and equipment (that we later discontinued) and for granulocyte collection (which we did not actually perform).
To Be Continued:
2011
Established automated component production using Atreus technology, plasma and platelet pathogen inactivation (Mirasol)—made HMC component production Good Manufacturing System GMP compliant
2011
Qatar is the first to adopt non-PCR-based NAT technology (Grifols/Novartis Tigress) and becomes world reference site for this
2011
Based on the above, Qatar can now completely process all whole blood into blood components (red cells, platelets, and plasma) in as little as 5 hours from collection!
2011-2020:
I established policies and procedures for the hospital blood banks/transfusion services, blood donor center, therapeutic apheresis, and laboratory information systems to bring HMC in compliance with the Council of Europe, international AABB, and other standards. I customized our own standards for our local needs based on them.
2012-2013
Implemented custom build of the multilingual blood bank computer system (Medinfo) for both patient and donor services, including development of interfaces to all production equipment including Atreus and Mirasol (world’s first) and a direct link to Ministry of the Interior to obtain patient demographics in English and Arabic—Qatar became the world’s first site to combine fully-interfaced, automated component production with pathogen inactivation: Qatar becomes world reference site for this.
2013-2014
Built, validated, and implemented laboratory build of hospital information system, Cerner Millennium
2015
Replaced and updated Atreus with Reveos automated component production to allow faster throughput and capacity with a full bidirectional interface (world’s first), introduced platelet
additive solution PAS with pathogen inactivation (Mirasol)—Medinfo interfaces updated to Reveos for all equipment: this doubles the capacity to process whole blood into components using the same physical space
2015-2019
Updated dedicated blood bank software Medinfo Hematos IIG by several versions using Division Head, LIS, and internally trained Super Users—at great cost savings to HMC by not using outside consultants (e.g. Dell Consulting)
2019
Established column absorption technology using Terumo Optia therapeutic apheresis machine for treatment of ABO-incompatible renal transplants: I validated using the Ortho Vision MAX to perform ABO antibody titers for this system and correlated it with the reference method at Karolinska Institutet in Stockholm (manual gel) to bring rapid throughput and labor savings—Qatar being the first-site in the world to do this. We saved money by using the same apheresis machine to use this column absorption technology (no need for second machine to use the columns)
2020
Expedited setup (two weeks total) of COVID-19 convalescent plasma production, initially manual and then fully integrated into the Medinfo computer system as a customized module with separate quarantine collection, production, and transfusion service functions
Other:
I was awarded two HMC Star of Excellence Awards:
2013—Liver Transplantation Transfusion Support
2019—ABO-Incompatible Renal Transplantation Support

