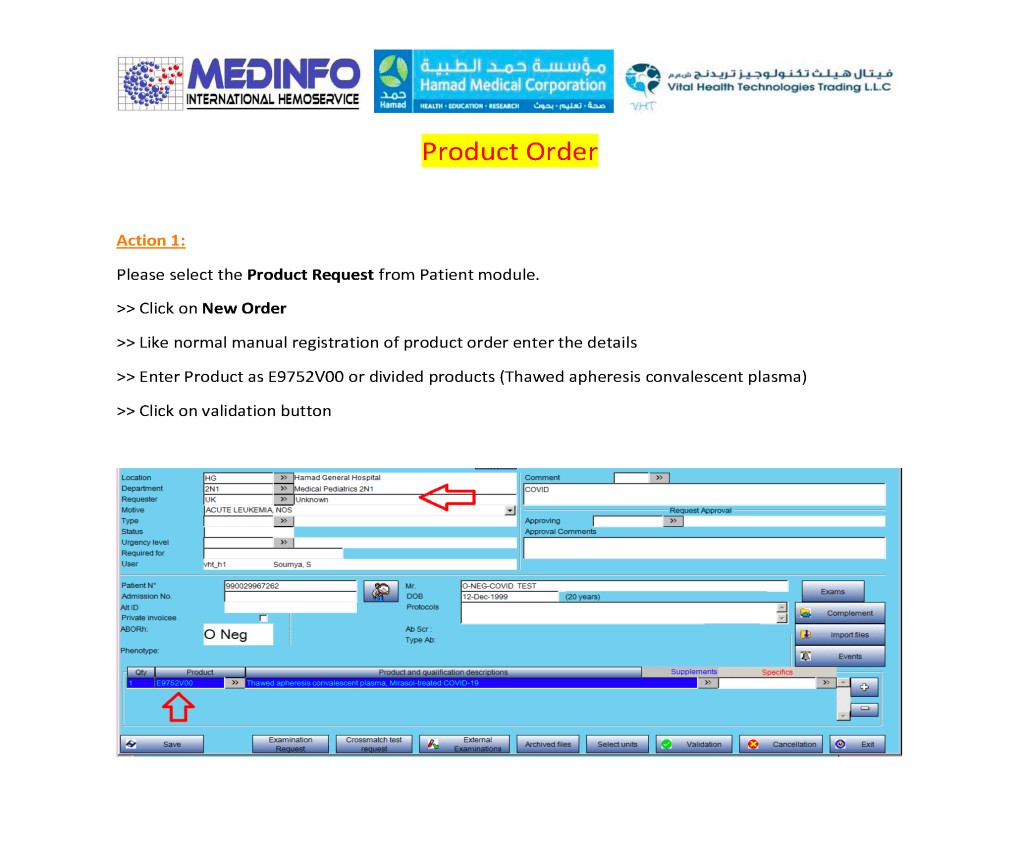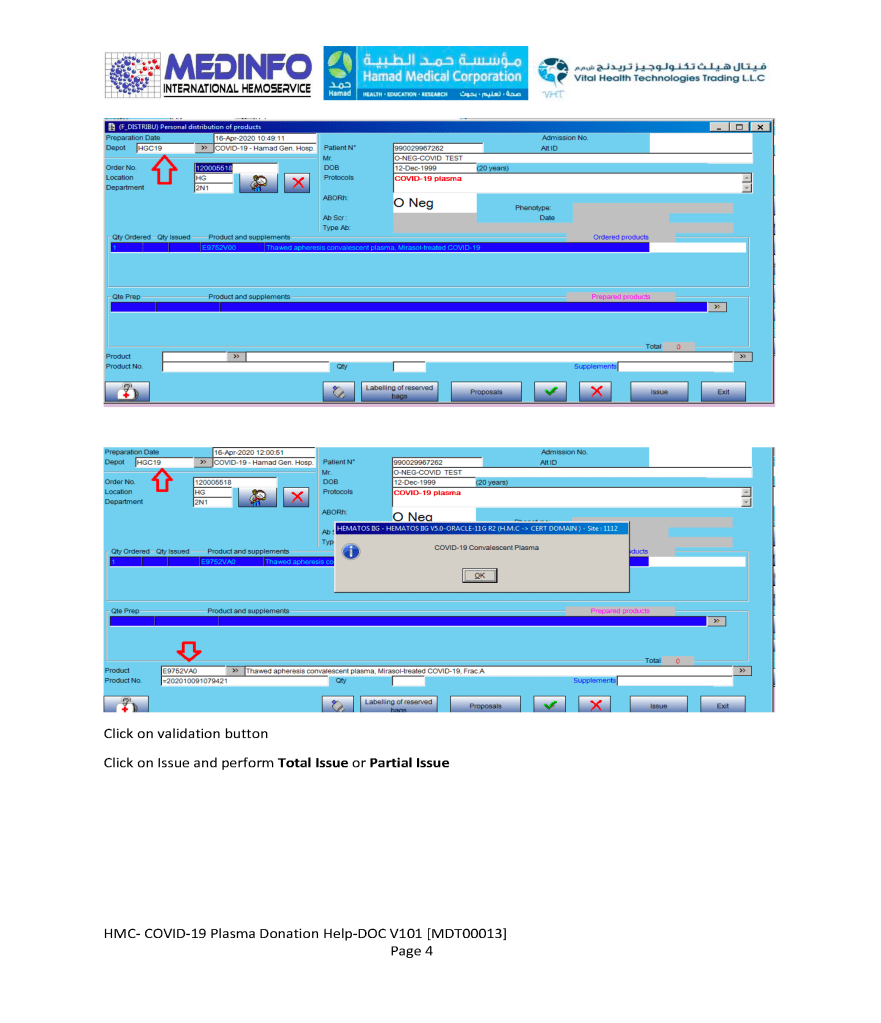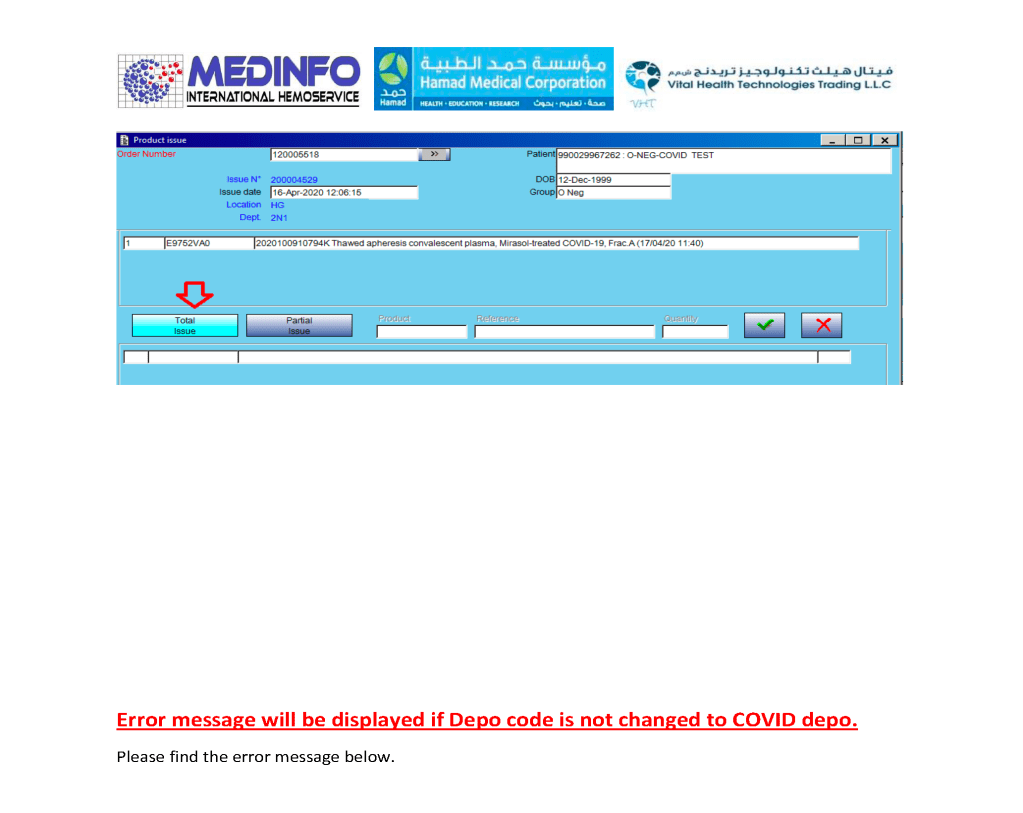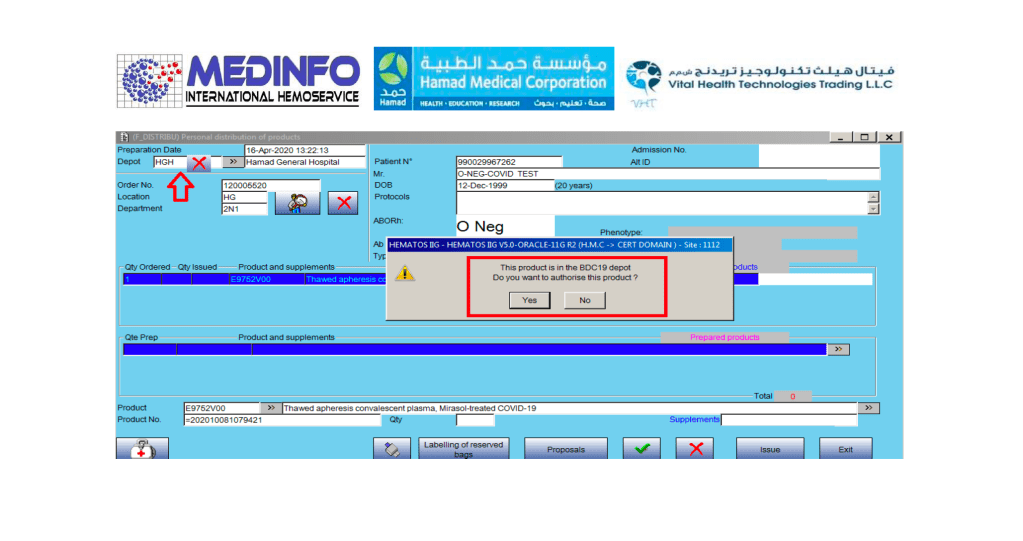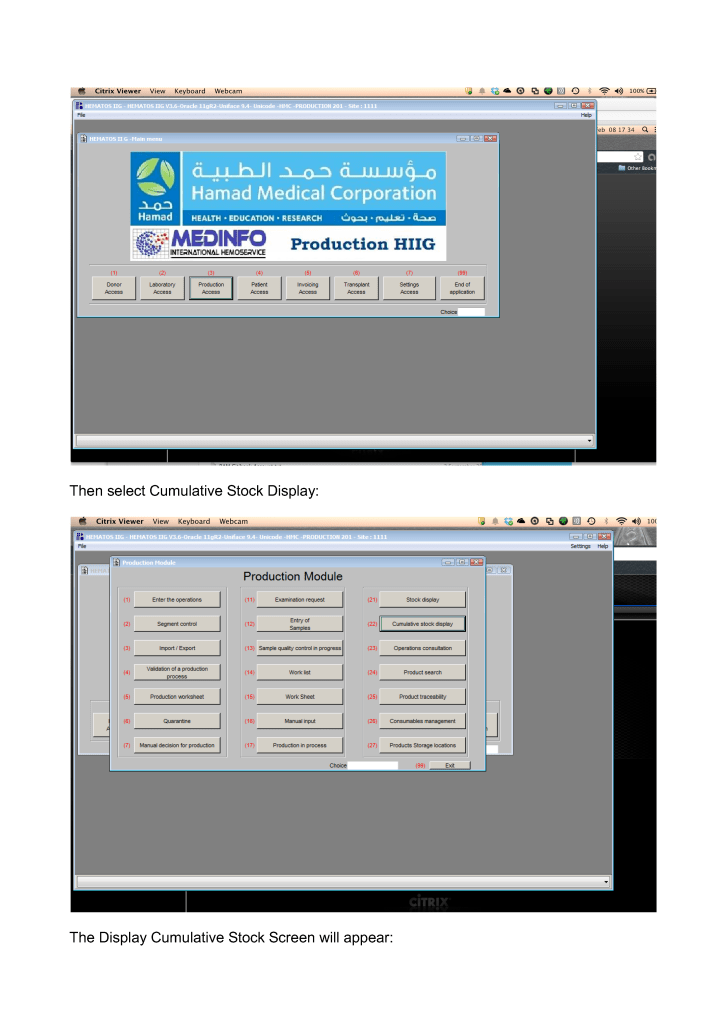
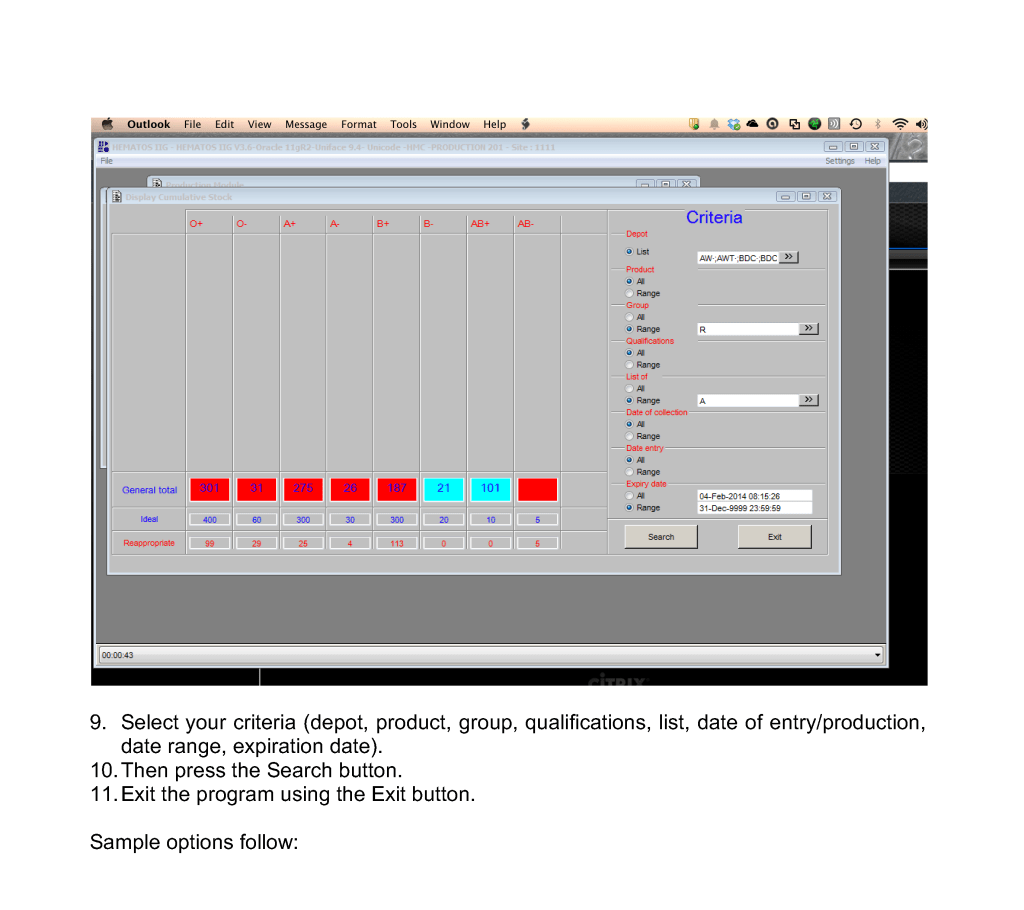
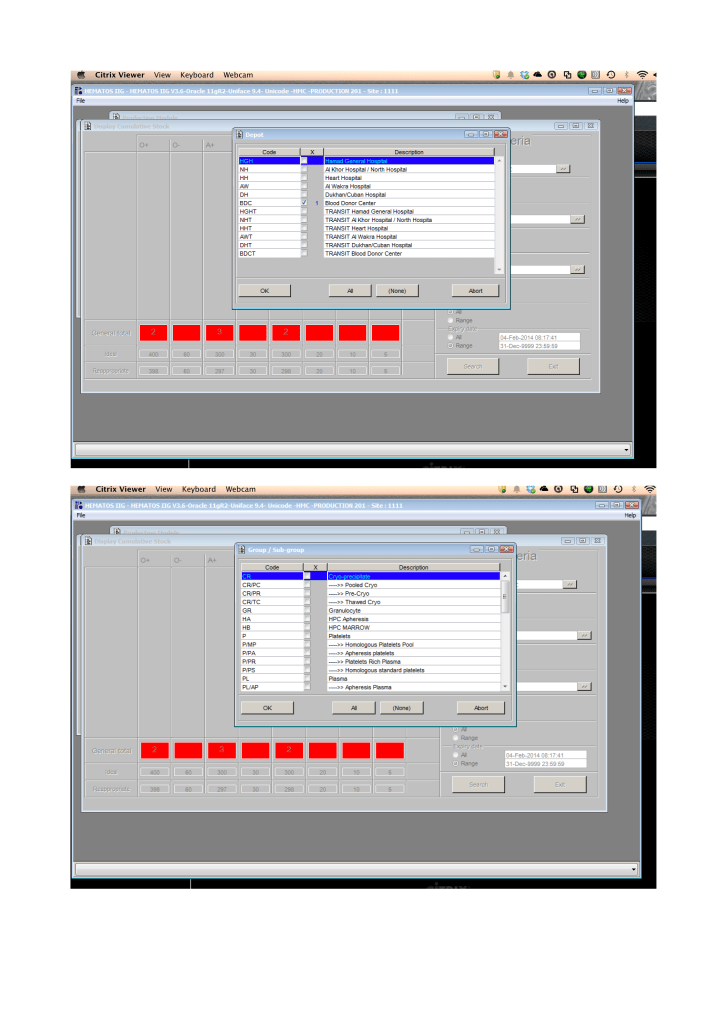
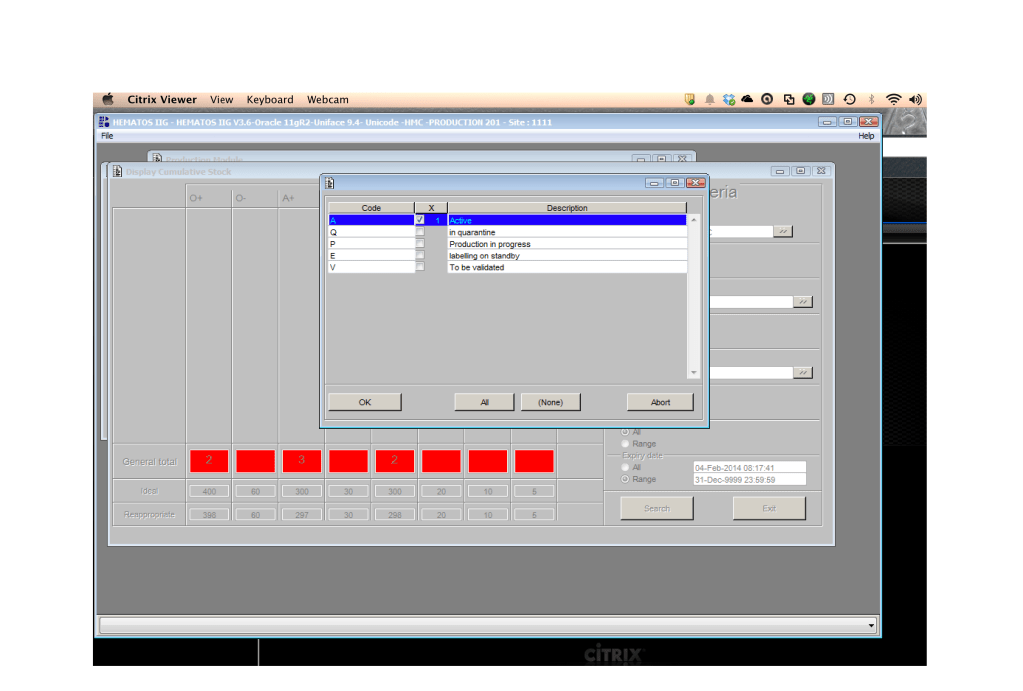

Includes registration, questionnaire, physical exam and arm check, collection, marker testing, component separation, donor immunohematology testing





Principle:
As part of good manufacturing process, we must trace everything in Transfusion Medicine, from registration through release of components. The adoption of the Medinfo Hematos IIG computer system allows us to document anyone and everyone who “touches” the blood components and all processes.
Policy:
References:
Principle:
Blood components must be maintained at specified temperatures to avoid hemolysis, bacterial contamination, and maintain full efficacy (e.g. coagulation factor activity.)
Policy:
Note the different temperature ranges for refrigerated components stored in the blood bank (1-6C) versus transport (1-10C).
References:
Section 5.6.5, Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
Principle:
Therapeutic phlebotomy is a medical procedure that requires a written physician’s order and review/approval by a transfusion medicine physician. Transfusion Medicine is responsible for the procedure and makes the final decision of the conditions of the procedure (volume of whole blood and venue).
Policy:
References:
Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA.
Annual Review: Therapeutic Phlebotomy Criteria and Treatment Goals
This a revised version of a previous post for the processes of autologous transfusion that I developed at HMC Doha. It can serve as a template for other sites and was also a teaching document for the Transfusion Committee members.
Background:
There are four basic types of autologous transfusion: preoperative, perioperative hemodilution, intraoperative, and postoperative drainage/collection. The use of all of the above techniques can significantly decrease the need for homologous blood and as an added benefit reduce the risk of the disease transmission and immunosuppressive effects of such homologous transfusions.
Preoperative collection can make available packed red blood cells, whole blood, platelets, FFP, and/or cryoprecipitate. However, at most two units of blood per week can be collected. RBC’s can be stored for up to 42 days in the liquid state, frozen RBC’s up to ten years, platelets up to five days, and fresh frozen plasma and cryoprecipitate up to one year. The last collection cannot be less than 72 hours prior to the surgery time. Units can be collected as long as the patient’s hematocrit remains above 33%. Supplemental iron and erythropoietin can increase the number of units harvested. The biggest obstacle to using this service is the coordination of the patient scheduling for this procedure. The blood bank does not have the resources to prospectively analyze the surgical scheduling and make the various appointments, contact the attending physician, etc. Thus, this service is vastly underutilized.
PHD or Perioperative hemodilution (also called acute normovolemic hemodilution) is useful in cases when the anticipated blood loss is at least one liter and the initial hematocrit is at least 34%. This includes essentially all types of surgery, but in particular cardiac, vascular, orthopedic, and urologic cases. The patient’s hematocrit Hct. is lowered to the range of 20-25% and the blood is replaced by crystalloid in a ratio of 3:1–i.e. three times as much fluid as blood, or in the case of colloid replacement, a 1:1 ratio of colloid plus 0.5 to 1.0 ml. of crystalloid. Crystalloid has the advantage of being readily removed by diuretic use. However, this technique should not be undertaken when vascular access is inadequate or appropriate monitoring devices are lacking. The physician performing PHD must be familiar with the compensatory mechanisms normally invoked when the hemoglobin is acutely lowered.
Another new twist to PHD is the perioperative collection of platelets by a special attachment to a cell-saving machine. This could allow collection of a typical apheresis load, about 6 to 10 units of fresh platelets for potential use. There are currently studies underway to determine if this has particular clinical advantages to warrant the additional cost.
Intraoperative salvage may be performed with a number of canister or automated devices. The latter is usually used when there are large volumes (usually 3 or more units) of blood to be salvaged. Depending on the body site, the recovered material is at least filtered and may or may not be washed. Care must be taken to collect the blood at a low suction rate and with minimal turbulence to minimize hemolysis.
Postoperative drainage collection of certain sites such as post-knee replacement surgery or chest wounds involves a canister collection device. This blood may or may not be filtered before reinfusion.
Note that perioperative and intraoperative material can only be transfused up to six or eight hours at room temperature or 24 hours if refrigerated at 1-6 degrees (depending on the method used) post collection to minimize the risk of infection. Intraoperative collection may be contraindicated in cases of cancer and if the bowel has been violated.
Other Issues:
The transfusion criteria for autologous blood is the same as for allogeneic units.
The same compatibility testing algorithm applies both the autologous and allogeneic units.
Policy:
References:
I designed my blood bank software Medinfo for use by my staff at all levels and positions to adhere to and facilitate compliance to the workflow processes. Blood bank staff were restricted to access only those functions needed for their job duties.
Blood Donation records could not be viewed by outside staff for confidentiality reasons. Blood donor records were not linked to patient records at all.
We did not allow access for Medinfo to non-Transfusion Medicine staff since the screens were designed to maximize efficiency of the work processes, not the viewing by outsiders of results. Outsider access was made through the hospital information system HIS.
The separate hospital information system HIS interfaced to Medinfo for the following functions:
Even within the HIS ordering capabilities, there were additional restrictions:
Results viewing in the HIS were subject to additional conditions as well:
Only certain results, not all results were viewable directed in the patient’s chart since showing all results may be confusing to the outside physicians and nurses. The selected results were sent back into the HIS for viewing. All these non-viewable results were retrievable for blood bank staff in Medinfo.
Another option, one I did not use at either HMC Doha or NGHA in Saudi Arabia, was to order tests and components by physicians directly into Medinfo. Likewise, they could view test results directly in the system. Special screens could be constructed to offer ordering and results retrieval.
This is a sample document for use of expired reagents I wrote for HMC Qatar.
Principle:
Due to logistics issues including the long distance between suppliers in Europe and North America and Qatar and the importation/customs clearance of critical materials, Transfusion Medicine has developed a contingency variance policy to minimize disruption of the essential transfusion medicine testing and component preparation. Approval for use of outdated reagents in special circumstances is not meant to be an excuse for untimely monitoring and improper ordering of supplies.
Definition:
Rare Reagent: Any reagent that is either used uncommonly or is in short supply and difficult to obtain in a timely matter.
Policy:
References:
Sections 1.3.2 and 7.0, Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
When I started my COVID-19 convalescent plasma CCP collection in early March, 2020, there were few antibody tests available. However, I anticipated that eventually we would want to include antibody results with the donor record. Antibody results were not used originally at all in the criteria for CCP acceptability for release.
There are many assays by type of antibody (total, IgG, IgA, IgM) and quantitation by titer and/or signal-cutoff ration S/CO. Any of these parameters may be used to define rules for acceptability to complete production and/or allocate to patients. Instrumentation used for titering/quantitation may be interfaced to the blood bank software.
Here is my generic approach to including these results with the donation record. In Medinfo HIIG, it is possible enter test results retrospectively and these can be used set rules for acceptability. Please consult with my detailed post on using rules against parameters.
All of this is easily implemented since all test information will be stored as parameters. From these parameters we can construct rules for:
Also, one can override the rules if the clinician and the transfusion medicine physician agree. For example, there is a severe shortage of group B CCP so use of low-COVID-antibody titer group B CCP could be allowed.
The key is to build whatever test methodology you use and include the manufacturer’s cutoff for low versus high titer interpretation. These results can be printed on the ISBT label as well. One can easily build multiple methodologies and acceptability criteria if different tests are used at different testing sites in your system—just as can be done for other tests (ABO/D, antibody screen, etc.) If one changes methodologies in the future, Medinfo will still use the same rules that applied for the day of production.
Here are some sample test rules:
Example 1: Total COVID antibody > 160 is high titer:
Example 2: IgG antibody with S/CO ratio > 12 is high-titer:
Example 3: IgG and IgM antibodies must have S/CO > 12:
Another option would be just to record the quantitation for each antibody type and list this on the ISBT label and permit its release regardless of the value. One could also permit low-anti-B titer group A plasma with whatever rules you set up.
This is the early Qatar experience of treating severe COVID-19 using locally produced Covid convalescent plasma CCP. At that time, the plasma was not tested for SARS-CoV-2 antibody levels.
https://drzeydbloodbank.com/wp-content/uploads/2021/01/jmv.26537.pdf
This is the conclusion of a continuing series of posts on the actual Medinfo design of the CCP donation and release processes and covers the transfer of completed units to the hospital blood banks. It highlights specific changes made for the parallel CCP system I developed at HMC Doha.
A blood component is either located at a production site, a destination hospital blood bank site, or in transit. Here a quarantine production site is specified. The actual release process is documented in this post.
In summary, with the exception of the donor marker testing and immunohematology testing, all other CCP processes are handled by special quarantine processes. There are abbreviated marker testing specific for plasma and a special Predonation screening to minimize wastage of the expensive apheresis kits.