This is the third-year hematology fellow examination I prepared for NGHA Riyadh:


Includes therapeutic and donor apheresis
This is the third-year hematology fellow examination I prepared for NGHA Riyadh:

This is an update of a previous post.
I have been involved with planning for several plasma fractionation projects in the Middle East.
Many clients expressed the interest in using local plasma to make plasma derivatives (e.g. factor concentrates, intravenous gamma globulin, albumin), feeling that local plasma was safer than using imported plasma. Some of these are in short supply in the world market so the only way to ensure their uninterrupted availability is to consider to manufacture them for local consumption.
Still, the major issue today is that it is difficult for any country in the region to collect enough plasma to make such a project feasible. When I first considered such planning, we were looking for as much as 250,000 raw liters of plasma annually. Since then, there are newer technologies that allow much smaller batches to be cost-effective. Alternatively, one could charge higher prices for using smaller batches from local plasma.
Still, it is likely that plasma must be imported to sustain a plant. There are different regulations for plasma donor qualification country-to-country. Many of these jurisdictions may do less screening and testing than is done for normal blood and apheresis donors. Other countries use their blood donors with the same requirement for both commercial plasma and blood donations.
In this era of emerging infectious diseases, I personally favor using the stringent blood donor criteria—same as routine collections. It is not what we know, but the unknown pathogens that are potentially the most dangerous.
In addition to building a fractionation plant, one must train staff for this highly technical operation. This may require developing a special curriculum to prepare students for these jobs.
To export the plasma to certain regions, one may have to use plasma quarantine. In this protocol, plasma is held or quarantined until the next donation is collected and passes screening. This requires a robust blood bank production software such as Medinfo to track serial donations.
There are other processes to consider: how to develop a transport network to keep plasma frozen at minus 80C viable in a region that reaches very high ambient temperatures.
I would recommend a graded approach to develop such an industry. First I would negotiate a plasma self-sufficiency arrangement. We would collect local plasma in the country and export it to a manufacturing plant in another country and the derivatives would be returned to us. This may require inspection by the accreditation agency of the processing country to allow importation of the raw plasma for manufacture.
Since it is unlikely any one country has enough plasma for manufacture, recruiting neighboring countries to participate in a manufacturing plant is important. Technology for such a plant is complex so establishing a joint venture with one of the plasma industry companies is essential. Some manufacturers are very keen to develop extra capacity since there is a world-wide shortage of plasma fractionation and are even willing to help obtain external plasma sources for such a plant.
Such a plant is an excellent way to develop local talent to run such a plant, including training of local staff to be the industrial engineers in the plasma fractionation process. It would take approximately two years of training to prepare engineers on-site at a plasma fractionation site if they have studied the necessary science and mathematics subjects.
Such a program would take several years of planning and development. Some of the major steps needed include:
This is an update of a previous post.
Principle:
All therapeutic apheresis procedures are potentially life-threatening and must only occur by an order from a transfusion medicine physician with experience/competence in such procedures.
Definitions:
Policy:
References:
Principle:
This is the latest update on donor qualifications during the COVID-19 pandemic and addresses issues about COVID-19 vaccination, COVID convalescent plasma use and donation, return of donors into the donor pool after COVID-19 vaccination. All of this information is subject to change as new regulations are released.
Policy:
References:




The COVID-19 pandemic imposed new challenges to our system. In general, these could be divided into:
There were fewer donors in the early phase and the nurses also had to add a large number of donor plasmapheresis collections for COVID convalescent plasma CCP. Still they had to maintain all donor and therapeutic apheresis services with no increase in staff. Although elective procedures had been cancelled, there were still obstetrical, oncologic, and trauma services in full action.
Many of our staff were on leave when the borders were closed. Some had to wait months before they could return to work. Others had COVID-19 infection and were quarantined for several weeks. This further reduced staffing. We could not just hire outside staff since considerable training is involved in these processes.
I dedicated a separate donor collection space for the CCP program away from the regular donors as well as a quarantine processing area. Similarly, the CCP plasma was kept segregated from the regular plasma supply and a specially designed location was identified for release of this product. Working for this program diverted resources from blood collection to this special project, again without increasing resources.
With disruptions to shipments of supplies, including the Reveos whole blood kits and Trima donor apheresis sets, we had to rely on our large in-home inventory until the situation stabilized. We prescreened the CCP donor candidates before we would collect them to avoid wastage of kits.
Fortunately, our throughput was minimally affected because our equipment and processes had always stressed speed. We used single-well NAT testing to minimize the need of additional runs. Also, we used Reveos automated component processing to greatly speed production (one Reveos can process four whole blood units in about 23 minutes or about 12 units in 75 minutes.) One technologist could operate all 4 of our machines simultaneously and perform other tasks while the machines were working.
In the system I developed in Qatar, we could complete processing into components (RBCs, buffy coat platelet pools, leukodepleted plasma), all marker and immunohematology testing, leukoreduction of the pools and RBCs, Mirasol pathogen inactivation, and platelet additive solution in as little as five hours.
In rapid turn-around events, it is most helpful to have a robust blood bank computer system that can scale to the challenge. Also, it must mercilessly enforce all the rules starting with donor qualification, screening, collection through testing and production. At times of emergency, it is difficult to meet Good Manufacturing Processes manually.
I had built parallel separate donor collection, donor processing, and transfusion service/hospital blood bank processes specifically for CCP and had to staff them with available personnel, limited our capability to process regular donors. The blood bank computer software restricted CCP use to designated physicians and transfusing locations. For those interested, there is a separate series of posts about the CCP project and its implementation in the dedicated blood bank Medinfo HIIG.
COVID-19 vaccinations should have minimal effect in donor qualification since mRNA or antigen-based ones do not cause donor deferral. Live attenuated COVID vaccines will defer donors for 2 weeks by current rules—the same as other live vaccines. Donors who had previously received CCP will be deferred for three (3) months after last receiving this product.
In summary, the COVID pandemic reduced staffing and affected donor recruitment. We had production mitigations to maximize throughput. The system was stressed by the reduced staffing and special demands to produce CCP. However, the extent of our automation allowed us to maintain throughput throughout the crisis.
Principle:
As part of good manufacturing process, we must trace everything in Transfusion Medicine, from registration through release of components. The adoption of the Medinfo Hematos IIG computer system allows us to document anyone and everyone who “touches” the blood components and all processes.
Policy:
References:
This is the early Qatar experience of treating severe COVID-19 using locally produced Covid convalescent plasma CCP. At that time, the plasma was not tested for SARS-CoV-2 antibody levels.
https://drzeydbloodbank.com/wp-content/uploads/2021/01/jmv.26537.pdf
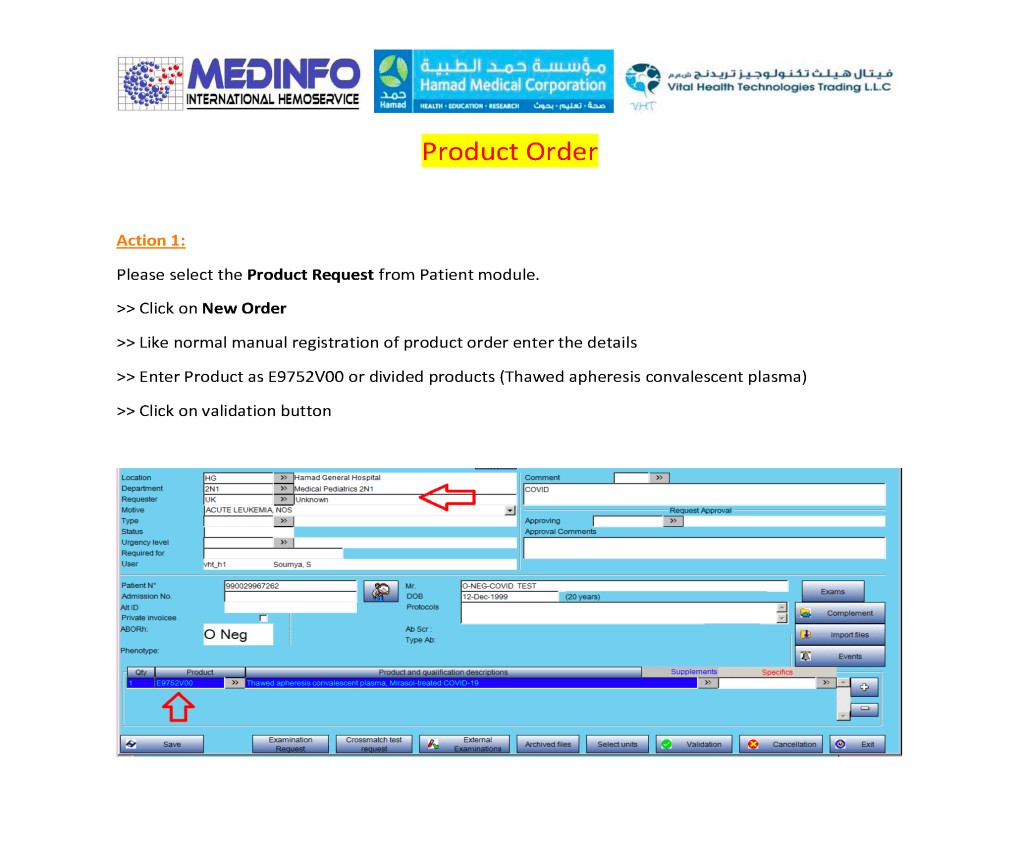
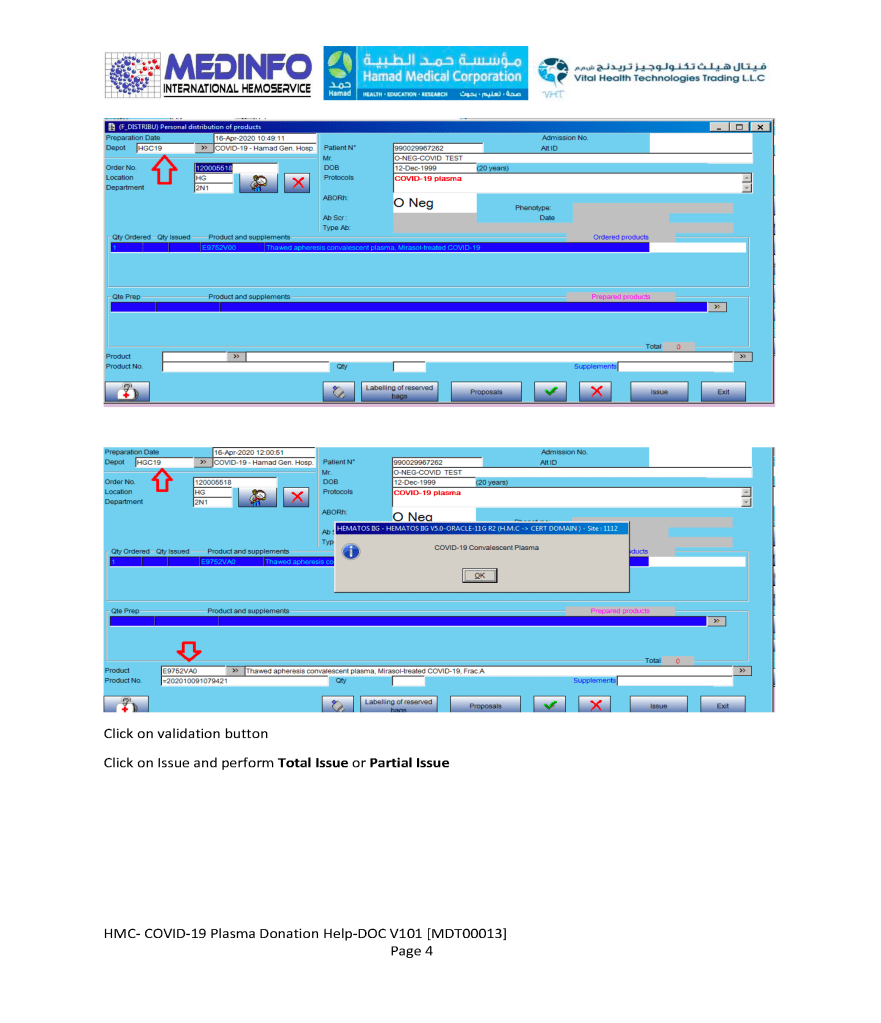
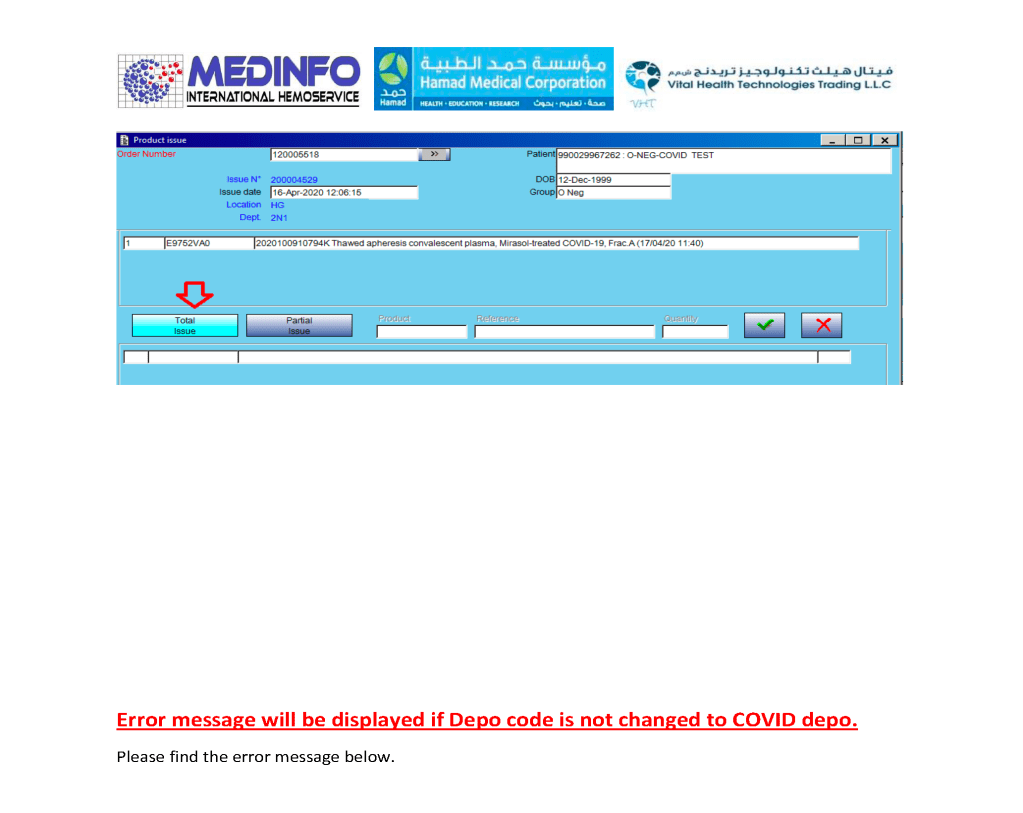
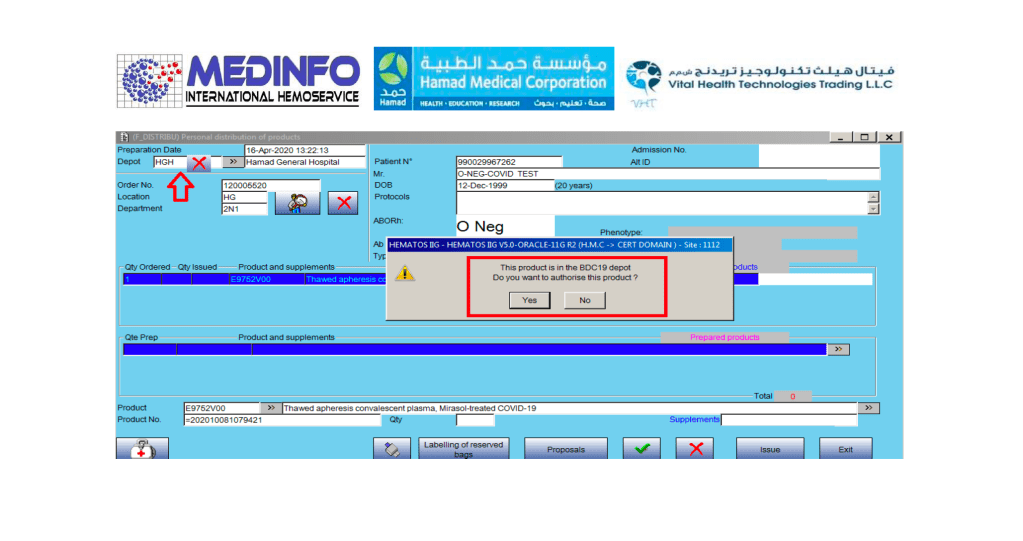
This is the conclusion of a continuing series of posts on the actual Medinfo design of the CCP donation and release processes and covers the transfer of completed units to the hospital blood banks. It highlights specific changes made for the parallel CCP system I developed at HMC Doha.
A blood component is either located at a production site, a destination hospital blood bank site, or in transit. Here a quarantine production site is specified. The actual release process is documented in this post.
In summary, with the exception of the donor marker testing and immunohematology testing, all other CCP processes are handled by special quarantine processes. There are abbreviated marker testing specific for plasma and a special Predonation screening to minimize wastage of the expensive apheresis kits.