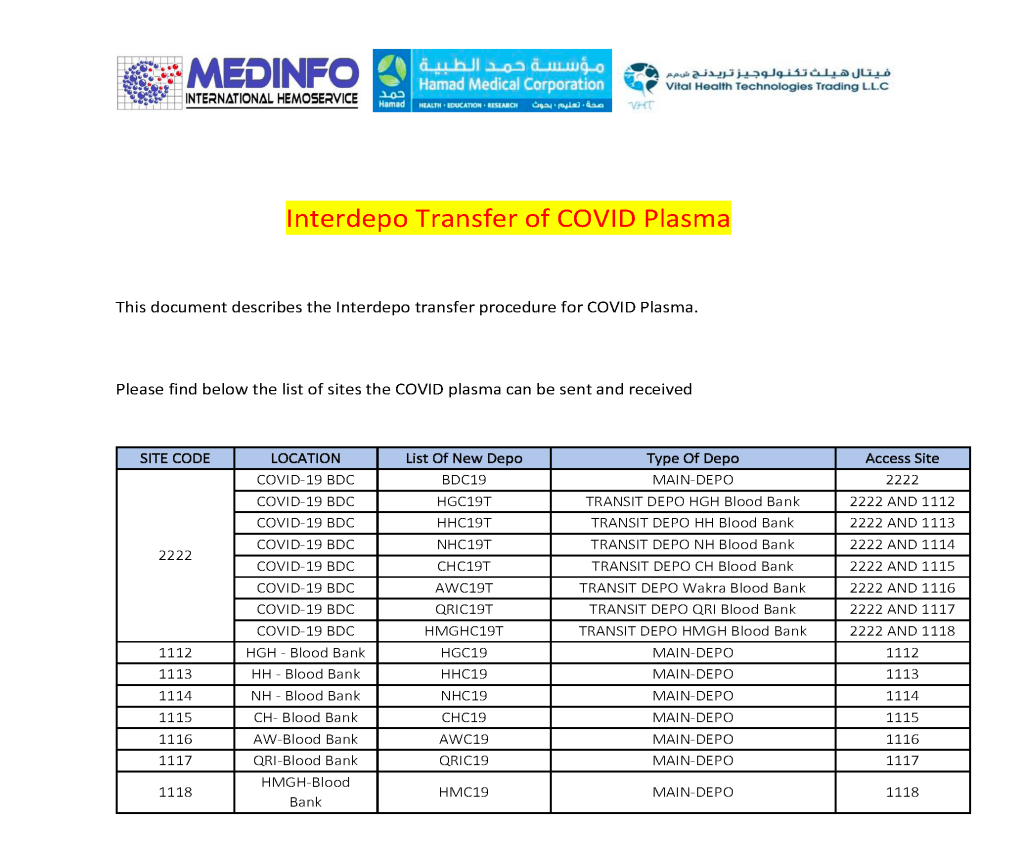
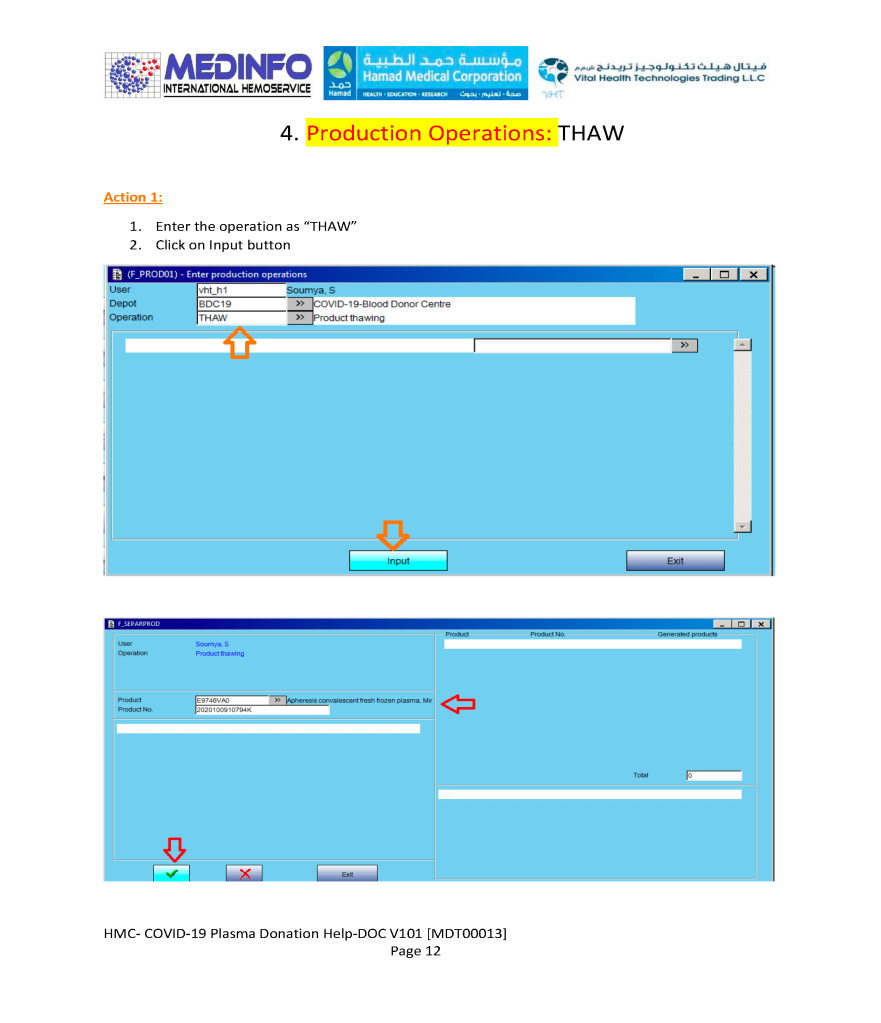
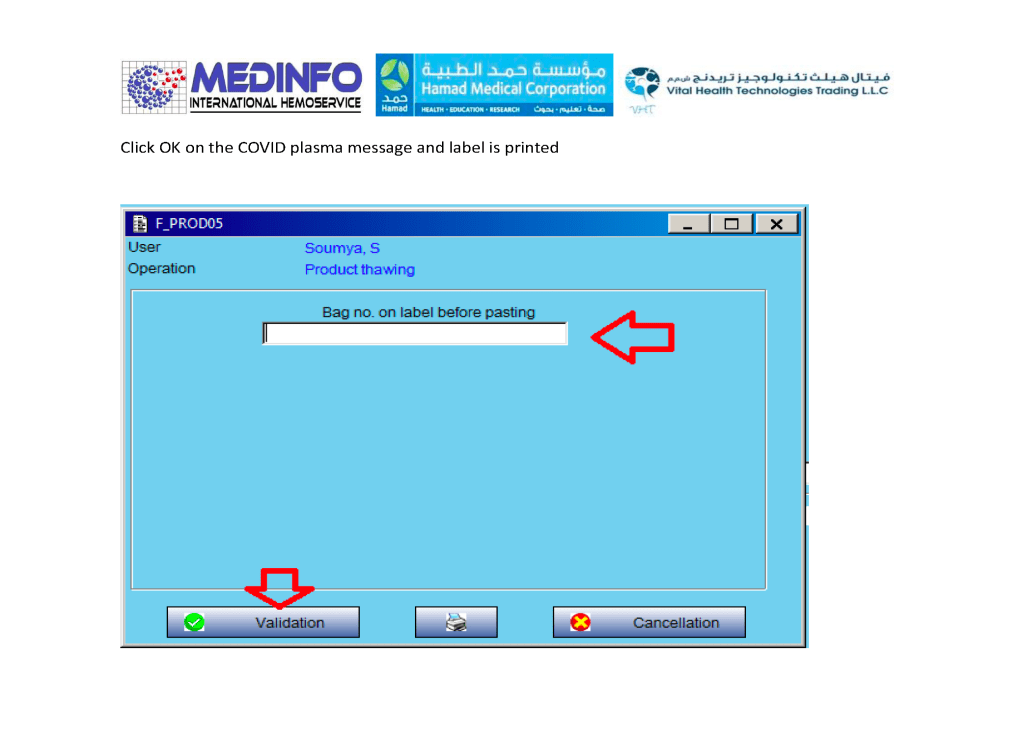
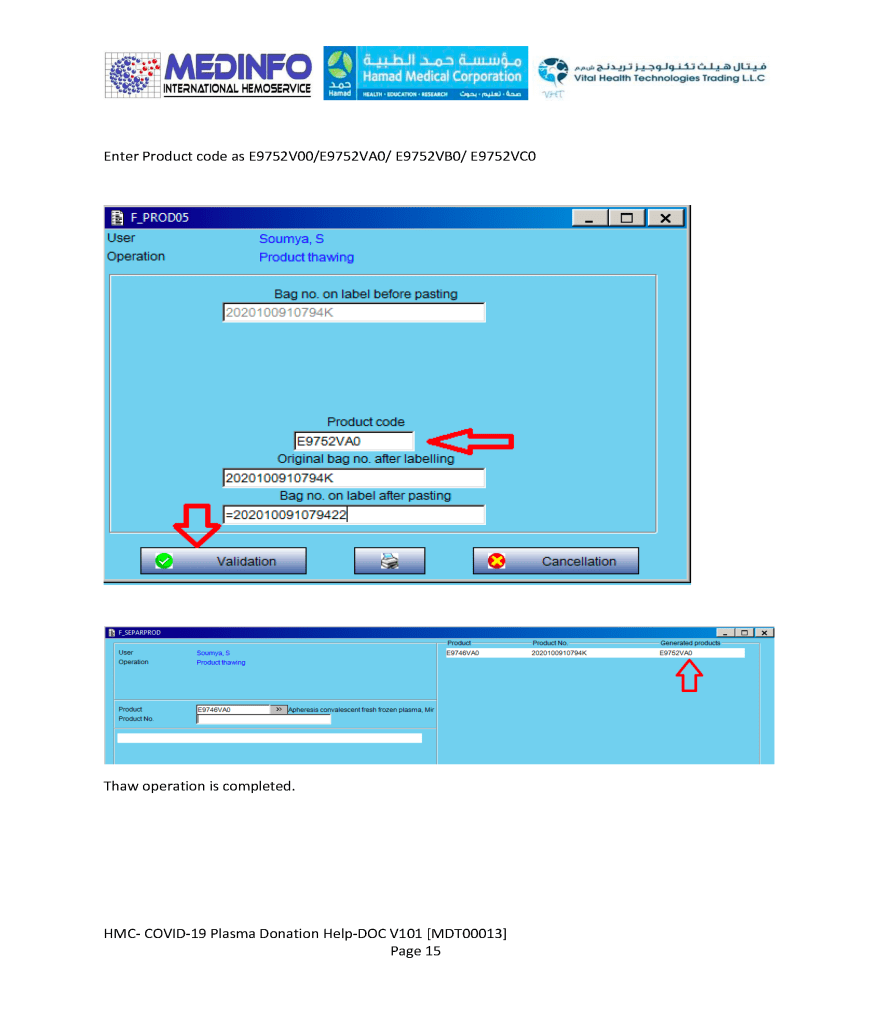
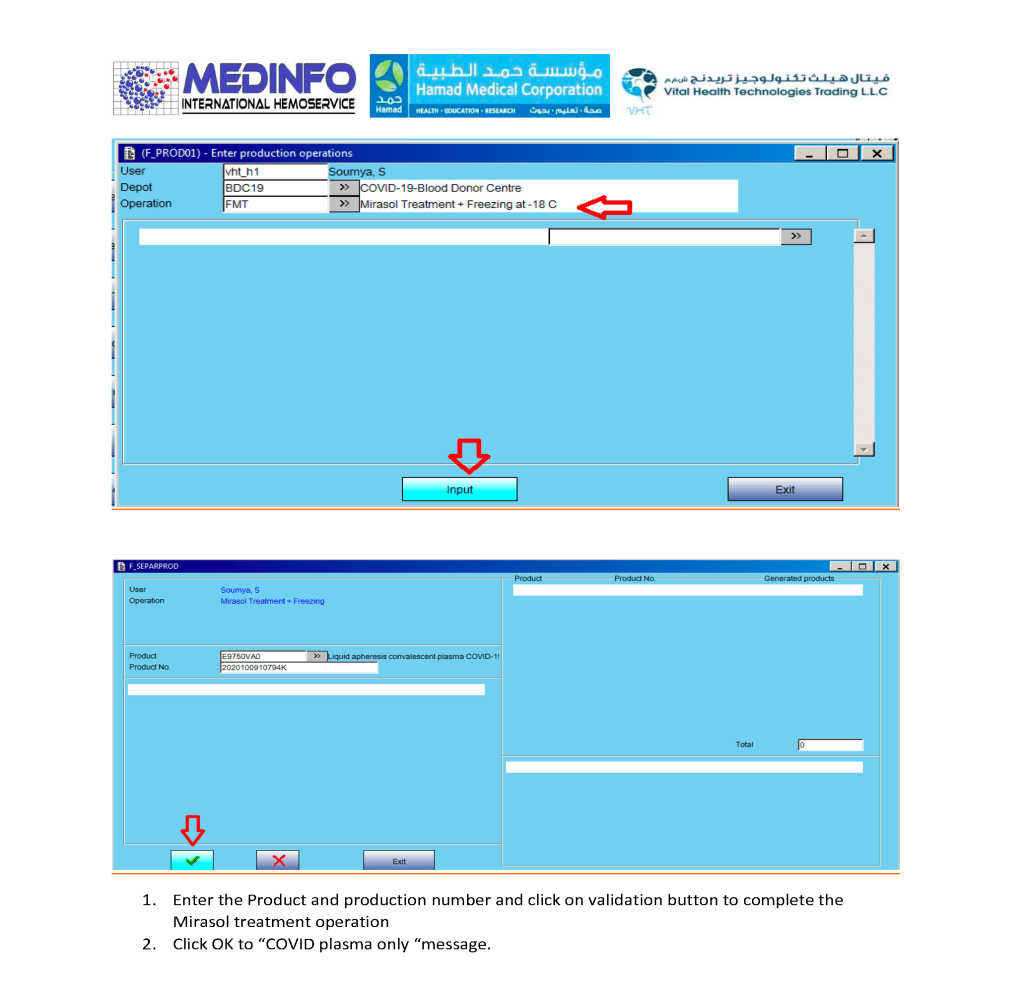
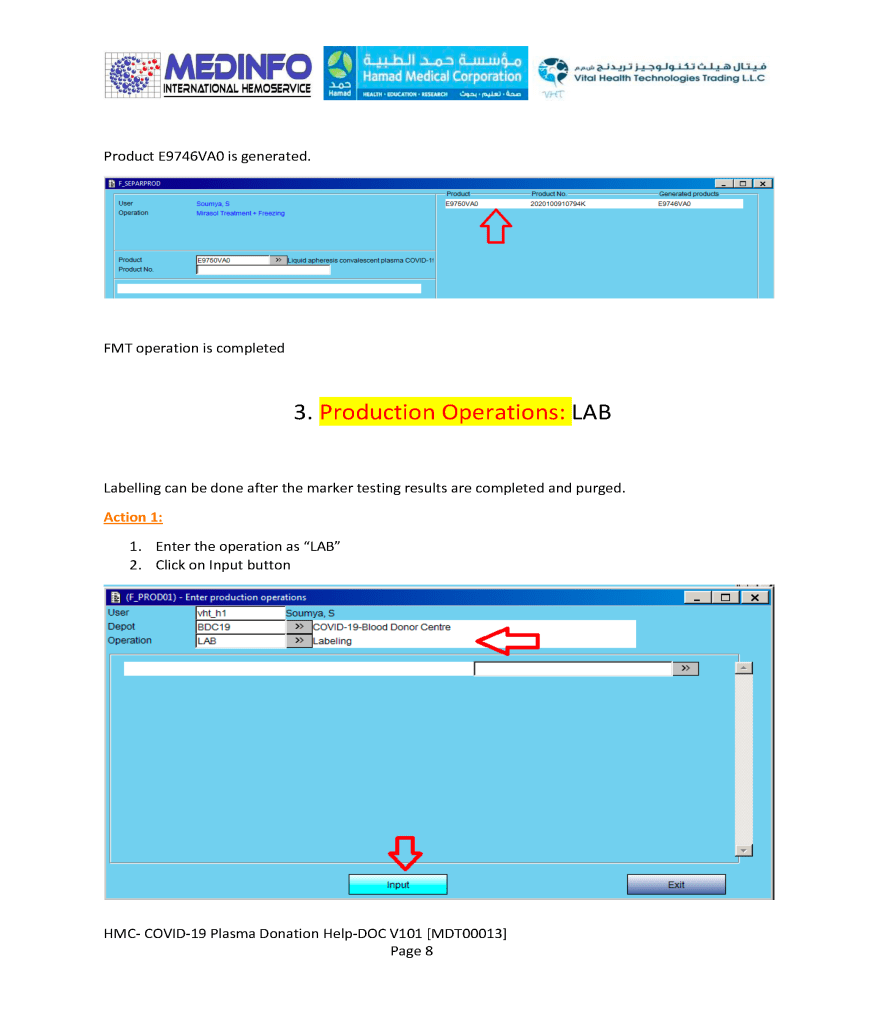
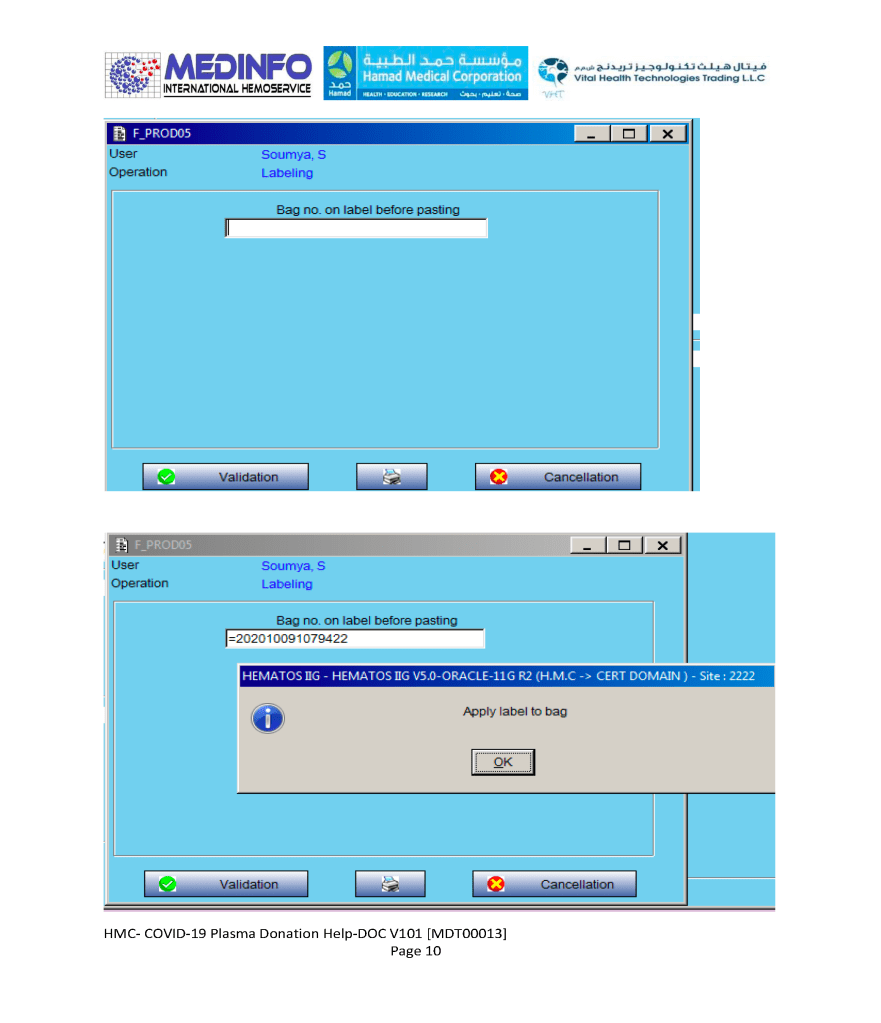
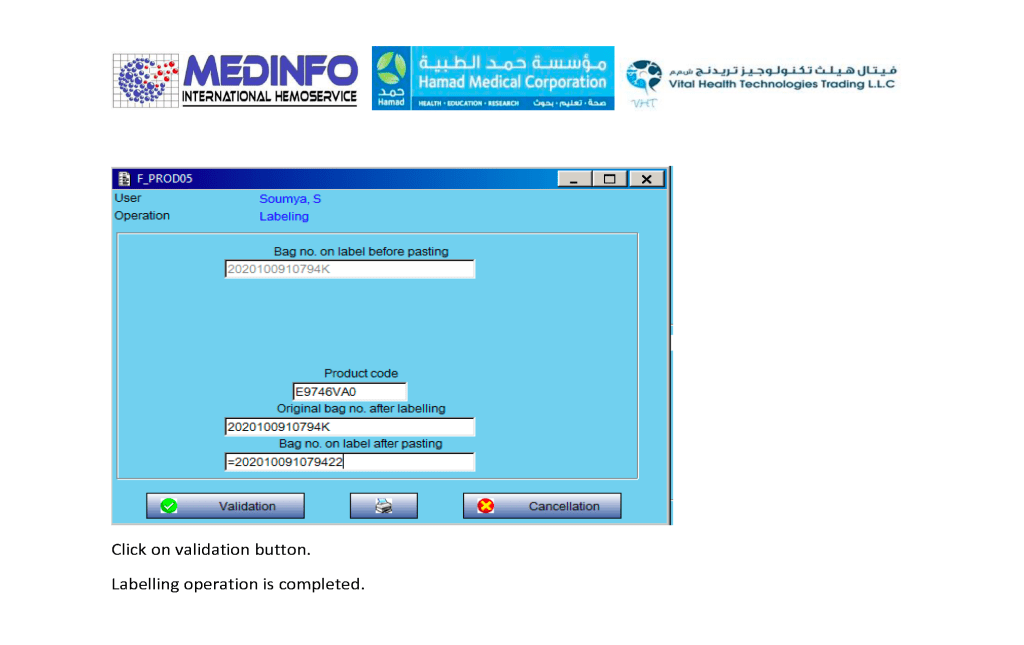
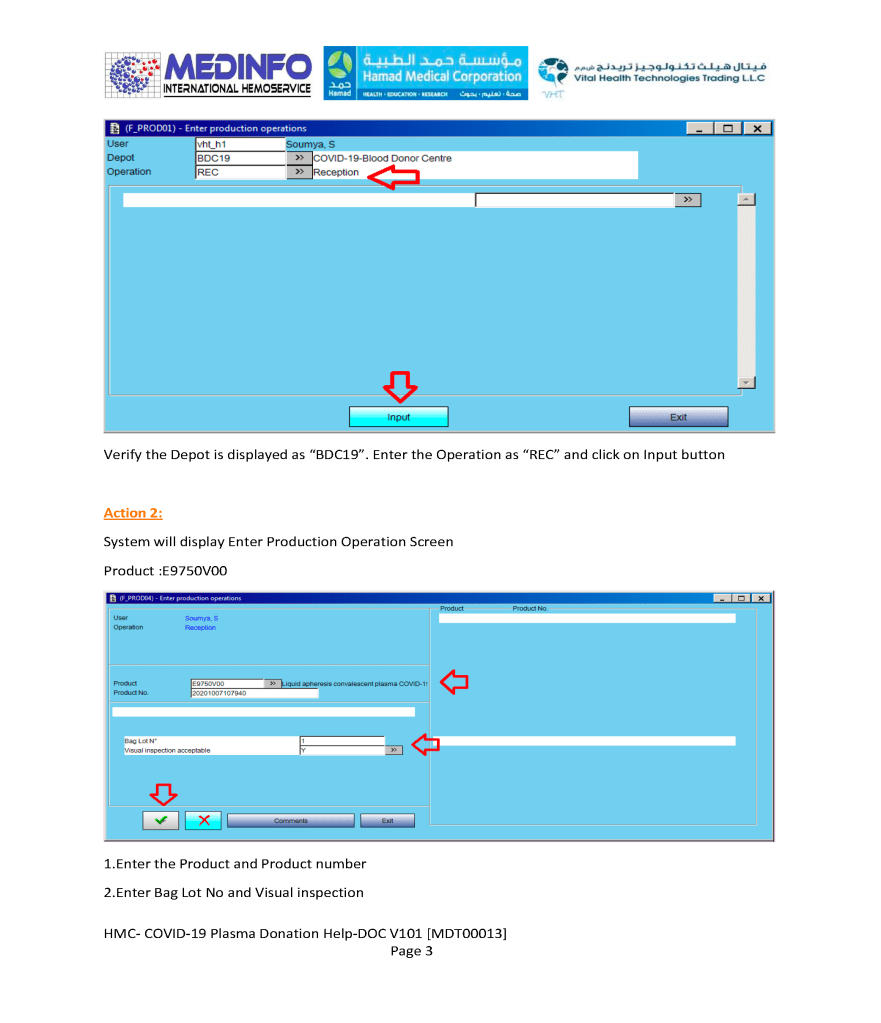
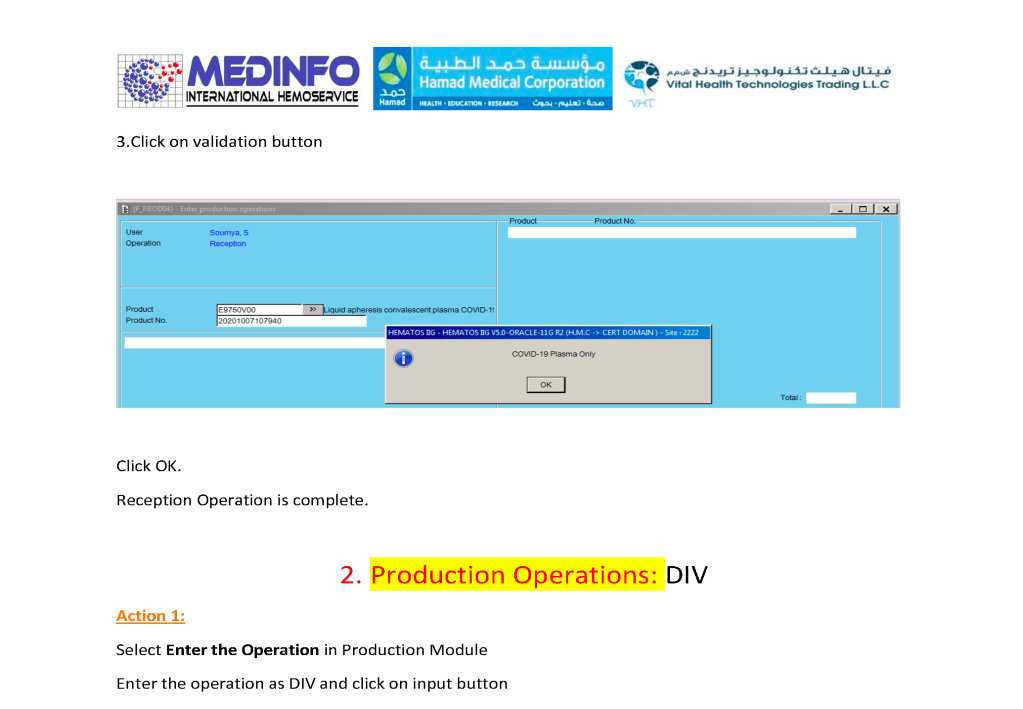
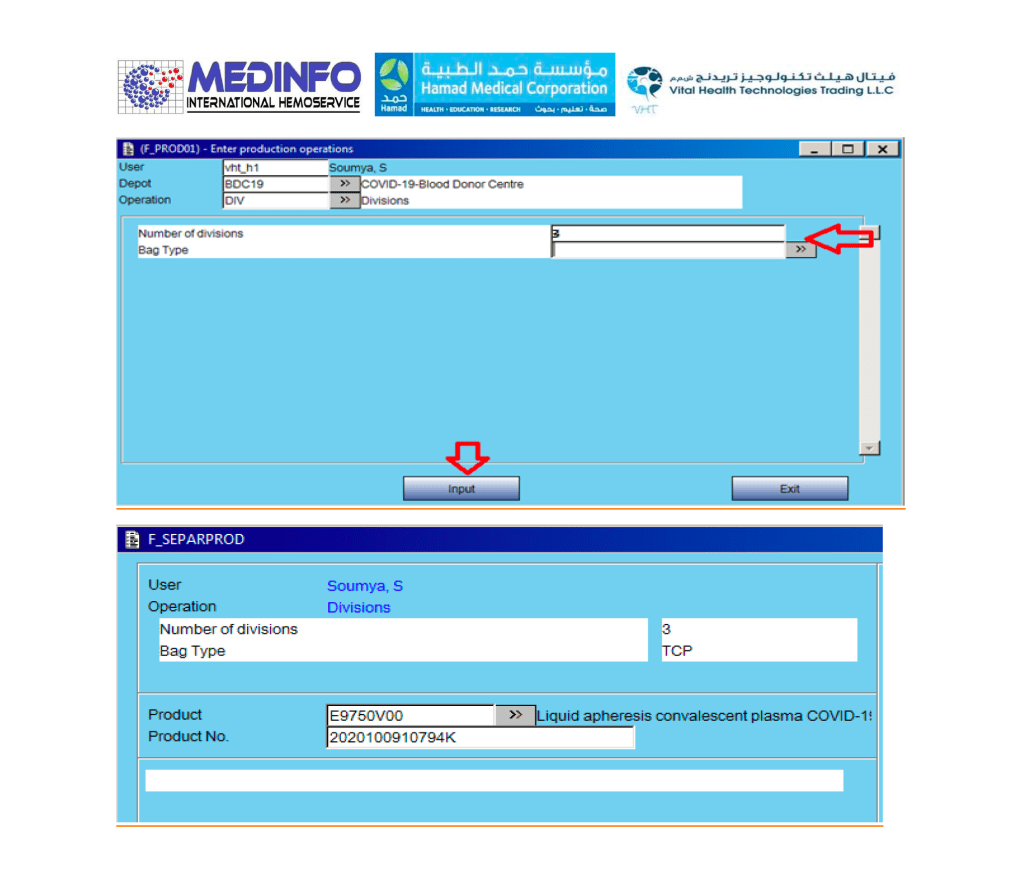
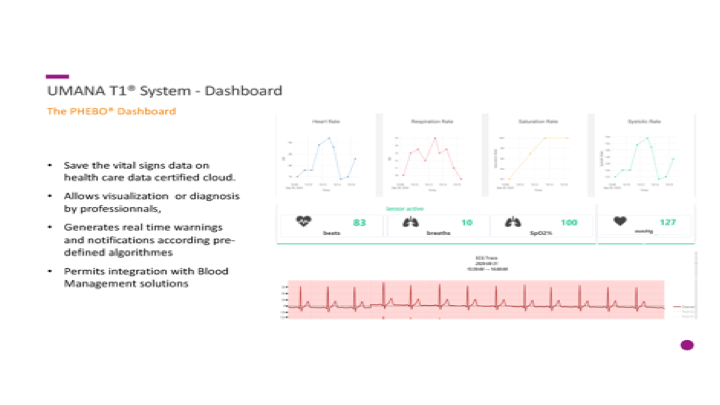
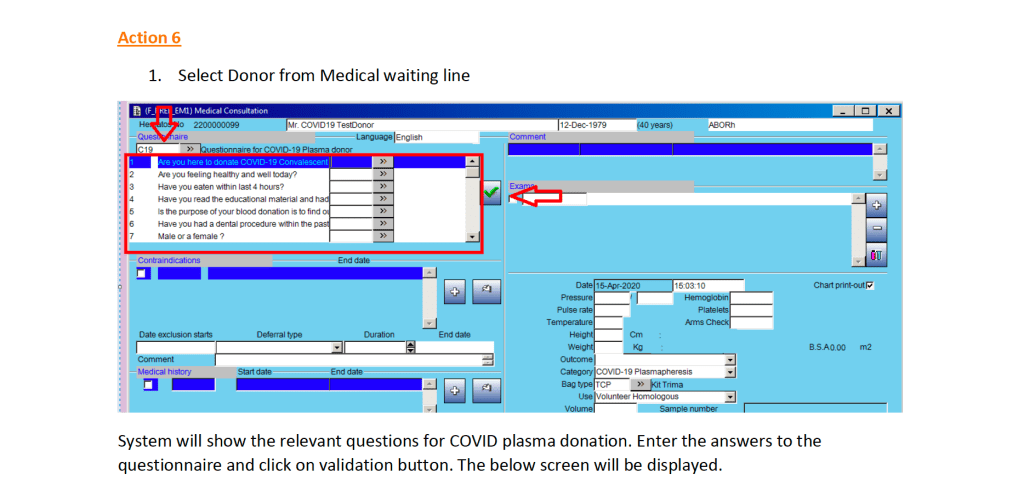
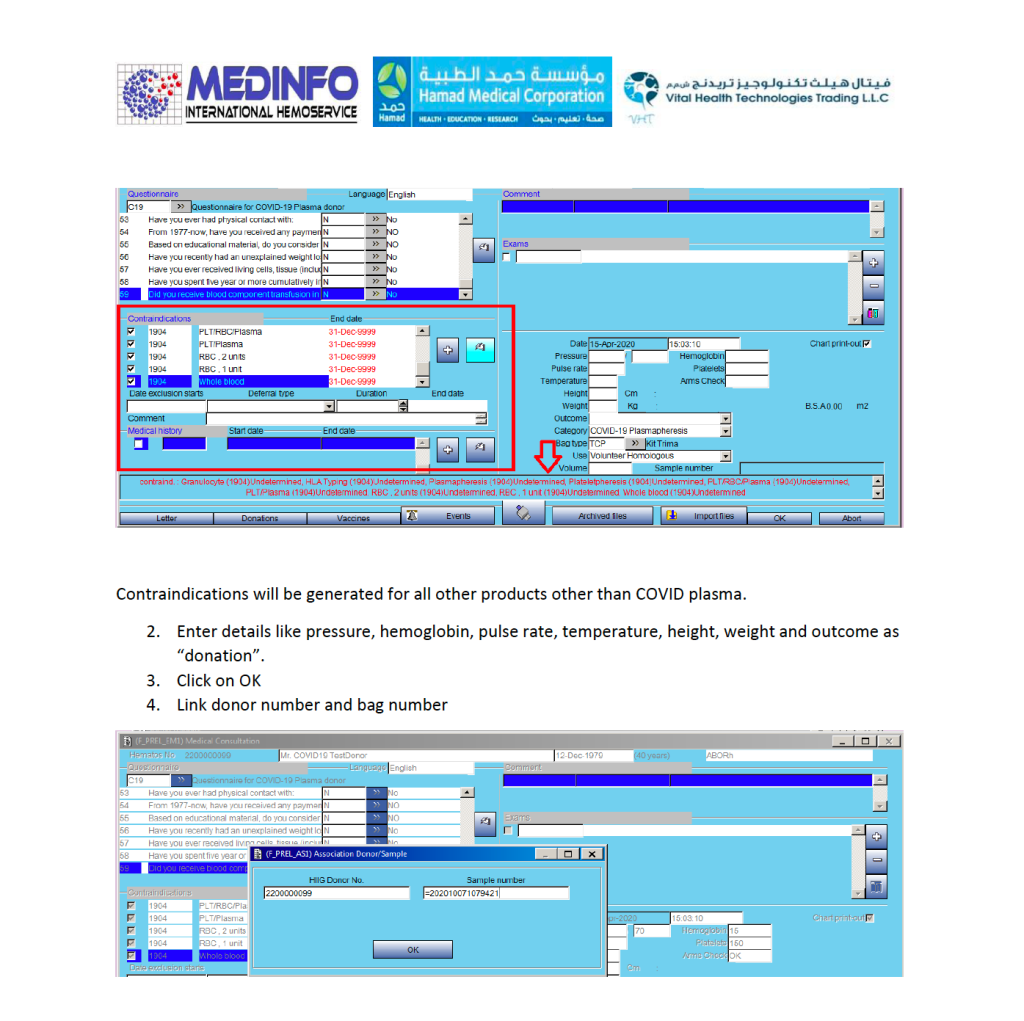
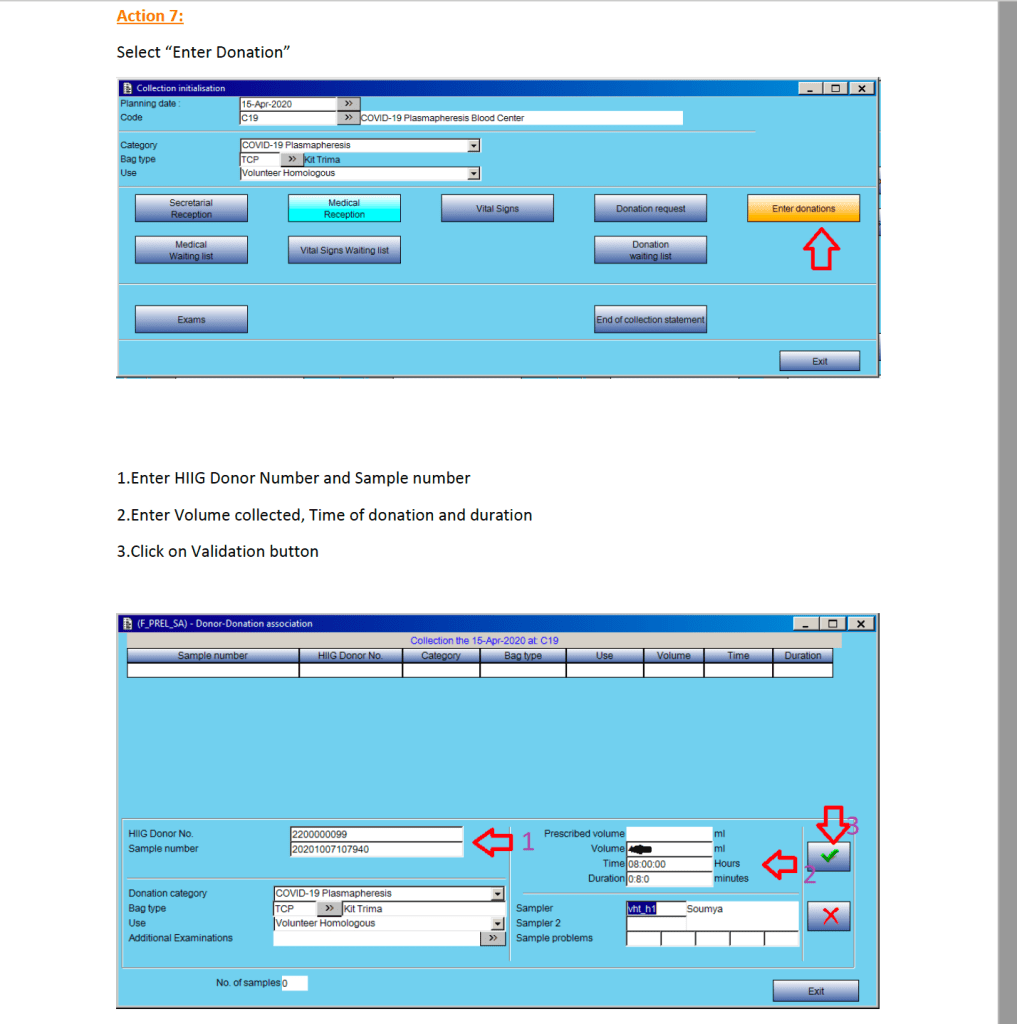
This is the conclusion of a continuing series of posts on the actual Medinfo design of the CCP donation and release processes and covers the transfer of completed units to the hospital blood banks. It highlights specific changes made for the parallel CCP system I developed at HMC Doha.
A blood component is either located at a production site, a destination hospital blood bank site, or in transit. Here a quarantine production site is specified. The actual release process is documented in this post.
In summary, with the exception of the donor marker testing and immunohematology testing, all other CCP processes are handled by special quarantine processes. There are abbreviated marker testing specific for plasma and a special Predonation screening to minimize wastage of the expensive apheresis kits.