In February 2020, I established COVID-19 plasma collection and distribution for the state of Qatar. Here is a research paper prepared in conjunction with the Qatar Communicable Disease Center about using it for patients there.








Includes manual and computer processes for donor, patient, and IT
In February 2020, I established COVID-19 plasma collection and distribution for the state of Qatar. Here is a research paper prepared in conjunction with the Qatar Communicable Disease Center about using it for patients there.









Principle:
In 1984 effective with the 13th Edition AABB Standards, the requirements for performing a direct antiglobulin test and autocontrol for compatibility testing were eliminated. The DAT is very important to detect delayed hemolytic transfusion reactions, certain autoimmune conditions, and drug-related hemolysis.
Since that time, the immediate-spin crossmatch and now the electronic computer paperless crossmatch may be used for most compatibility testing in place of the classic, antiglobulin-phase (indirect antiglobulin test) crossmatch.
If an antiglobulin phase (IAT) crossmatch is performed, an RBC unit with a positive DAT will cause a false-positive reaction. Since most crossmatching does not include the IAT, it will not be affected by the DAT status of a donor unit.
Policy:
Important: Don’t do a classic AHG/IAT phase crossmatch unless you have to do it (see conditions above.) A donor unit with a DAT is unlikely to be clinically significant and may be transfused safely to the patient in most situations. Patients receiving electronic-crossmatch and immediate-spin crossmatch are receiving units with positive DAT without incident.
References:
At HMC during my tenure, all plasma products—whole-blood and apheresis-derived were pathogen inactivated with riboflavin (Mirasol). In our software processes, I had options to release both Mirasol-treated and untreated (the latter in emergencies) and to aliquot either as needed. The same processes applied to COVID-19 convalescent plasma CCP except that they were performed in a quarantine production area. There were specific ISBT codes for CCP.
24/9/20
Principle:
Under AABB and FDA rules in the Uniform Donor History Questionnaire, unlicensed, investigational vaccines have a 12-month deferral or as indicated by a responsible physician. In light of the anticipated vaccination trials for COVID-19, this policy gives interim guidance until more definitive information is available.
For COVID-19 Convalescent Plasma CCP donation, investigational vaccine recipients should not donate COVID-19 convalescent plasma until further information is available about their antibody profile.
Policy:
Any donor who has received a COVID-19 (SARS-CoV-2) vaccine will be deferred as follows:
Reference:
Text from the AABB Weekly Report:
Novel Coronavirus Update, Regulatory Update: Investigational Vaccines and Deferral for Donor of Blood and Convalescent Plasma, AABB Weekly Report, 7 August 2020
“FDA recognizes AABB’s DHQ which includes unlicensed (experimental) vaccines on the medication deferral list as a 12-month deferral or as indicated by the responsible physician.
“For routine blood donation, the responsible physician may wish to consider the potential infectious risk associated with the vaccines, and the use of short deferral periods (e.g., 14 days) for live attenuated vaccines and no deferral for non-replicating, inactivated or RNA-based vaccines.
“We agree that no deferral is necessary for routine blood donors who might have received the mRNA-1273 Moderna vaccine.
“At this time, we suggest that individuals who have received a COVID-19 investigational vaccine should not donate COVID-19 convalescent plasma until further information is available about their antibody profile.”
Here is my recent interview with Terumo BCT showing how I rapidly set up the Covid-19 convalescent plasma program CCP at HMC this winter.
I resigned from HMC on 16/4/20. Here are a set of my major accomplishments during that period. None of my work after this date has any relationship to HMC.
2011
Established automated component production using Atreus technology, plasma and platelet pathogen inactivation (Mirasol)—made HMC component production Good Manufacturing System GMP compliant
Adopted non-PCR-based NAT technology (Grifols/Novartis Tigress) and Qatar becomes world reference site for this
Based on the above, Qatar can now completely process all whole blood into blood components (red cells, platelets, and plasma) in as little as 5 hours from collection!
2011-2020:
Prepared policies and procedures for the hospital blood banks/transfusion services, blood donor center, therapeutic apheresis, and laboratory information systems to bring HMC in compliance with the Council of Europe, international AABB, and other standards. I customized our own standards for our local needs based on them.
2012-2013
Implemented custom build of the multilingual blood bank computer system (Medinfo) for both patient and donor services, including development of interfaces to all production equipment including Atreus and Mirasol (world’s first) and a direct link to Ministry of the Interior to obtain patient demographics in English and Arabic—Qatar became the world’s first site to combine fully-interfaced, automated component production with pathogen inactivation: Qatar becomes world reference site for this.
2013-2014
Built, validated, and implemented laboratory build of hospital information system, Cerner Millennium
2015
Replaced and updated Atreus with Reveos automated component production to allow faster throughput and capacity with a full bidirectional interface (world’s first), introduced platelet-additive solution PAS with pathogen inactivation (Mirasol)—Medinfo interfaces updated to Reveos for all equipment: this doubles the capacity to process whole blood into components using the same physical space
2015-2019
Updated dedicated blood bank software Medinfo Hematos IIG by several versions using Division Head, LIS, and internally trained Super Users—at great cost savings to HMC by not using outside consultants (e.g. Dell Consulting)
2019
Established column absorption technology using Terumo Optia therapeutic apheresis machine for treatment of ABO-incompatible renal transplants: I validated using the Ortho Vision MAX to perform ABO antibody titers for this system and correlated it with the reference method at Karolinska Institutet in Stockholm (manual gel) to bring rapid throughput and labor savings—Qatar being the first-site in the world to do this. We saved money by using the same apheresis machine to use this column absorption technology (no need for second machine to use the columns)
2020
Expedited setup (two weeks total) of COVID-19 convalescent plasma production, initially manual and then fully integrated into the Medinfo computer system as a customized module with separate quarantine collection, production, and transfusion service functions
Other:
I was awarded two HMC Star of Excellence Awards:
2013—Liver Transplantation Transfusion Support
2019—ABO-Incompatible Renal Transplantation Support
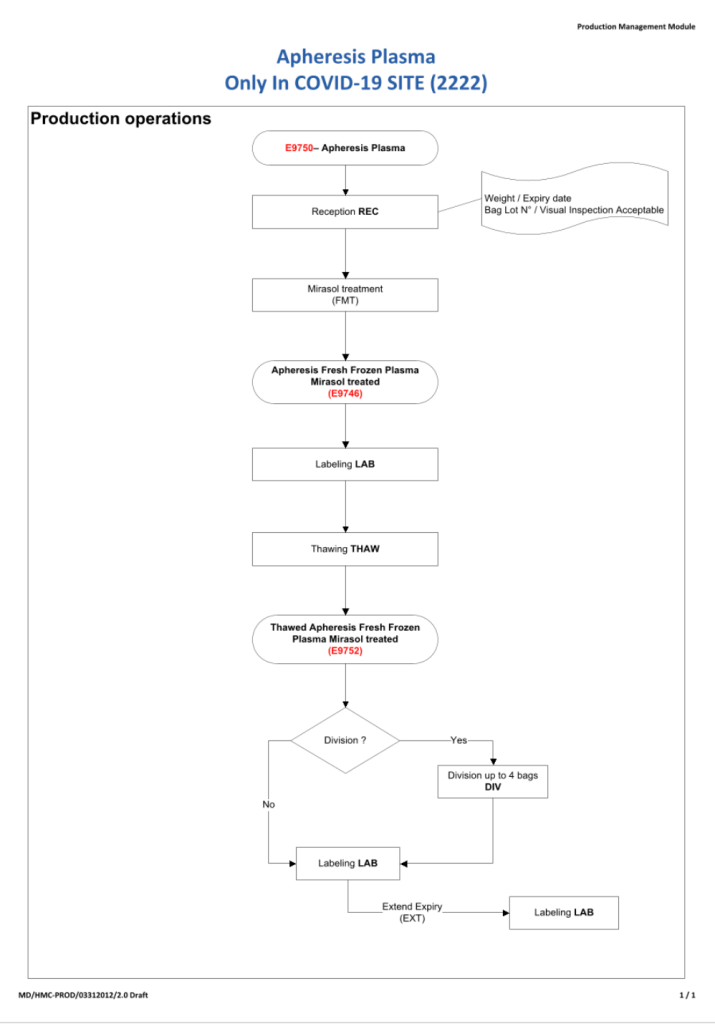
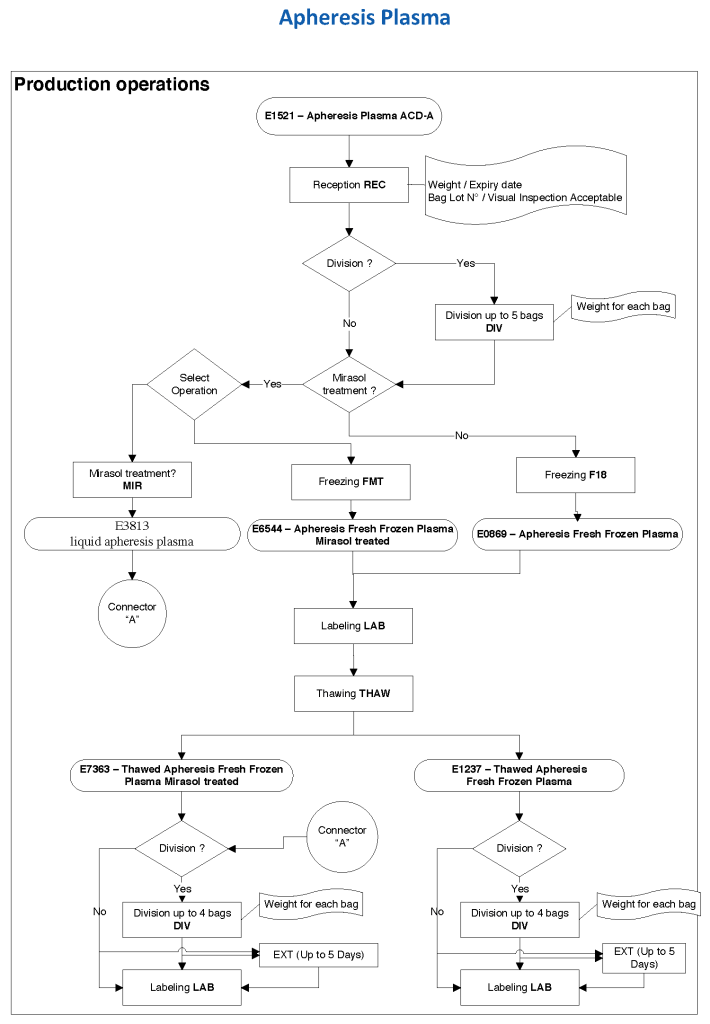
After the initial manual setup of the CCP program, the Medinfo process was set up. The following workflow shows the production of CCP from the raw apheresis collection, including division into aliquots based on the total volume. The plasma volumes were kept within the range for riboflavin pathogen inactivation (Mirasol).
The usual safeguards for production were also in effect for CCP. The product could not be labelled without all criteria (donor screening, collection, marker testing) being met. Furthermore, the inter-depot and transfusion service processes still applied. However, all steps were done in quarantine at a location separate from the regular processes. Also, the actual ordering and release of CCP was restricted to the quarantine hospital blood bank site.
The following outline the production process:
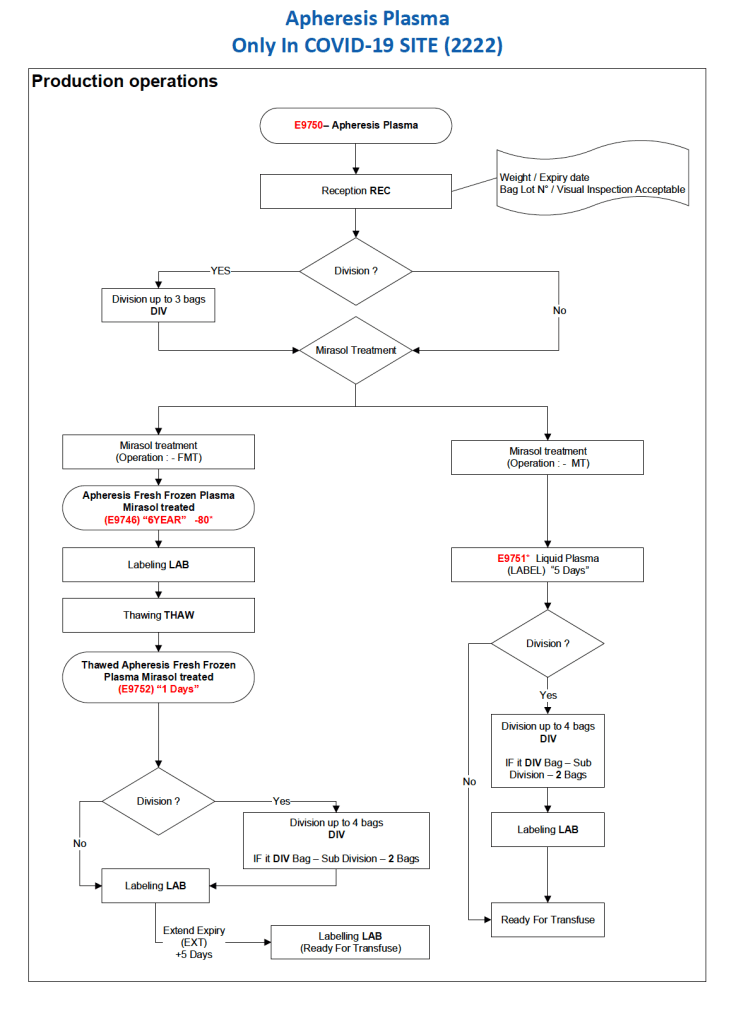

Principle:
Due to the pandemic, we will initially MANUALLY collect an experimental, investigational-use-only plasma product from apheresis donors and treat it with Mirasol. THIS IS A EMERGENCY INTERIM PROCESS UNTIL THE MEDINFO HEMATOS IIG PROCESSES ARE PREPARED AND VALIDATED.
Policy:
8/4/20
This is a sample Medinfo overview document for the blood collection process for HMC Doha that I designed in conjunction with Medinfo France and Medinfo Doha. This includes, registration, donor consent, questionnaire, physical examination, and collection.
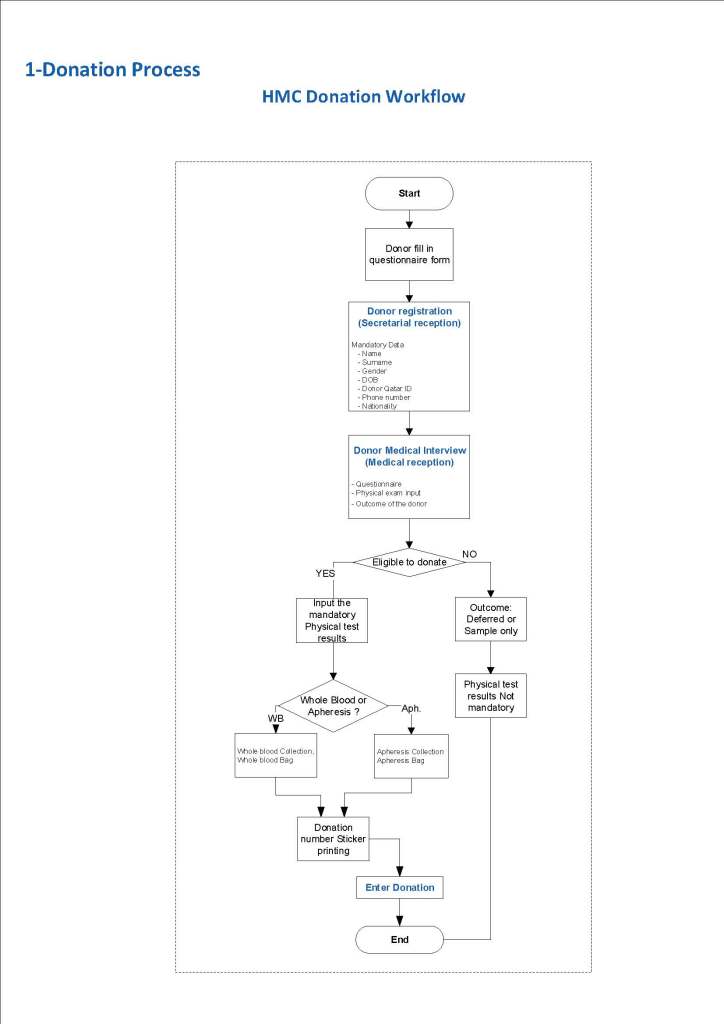
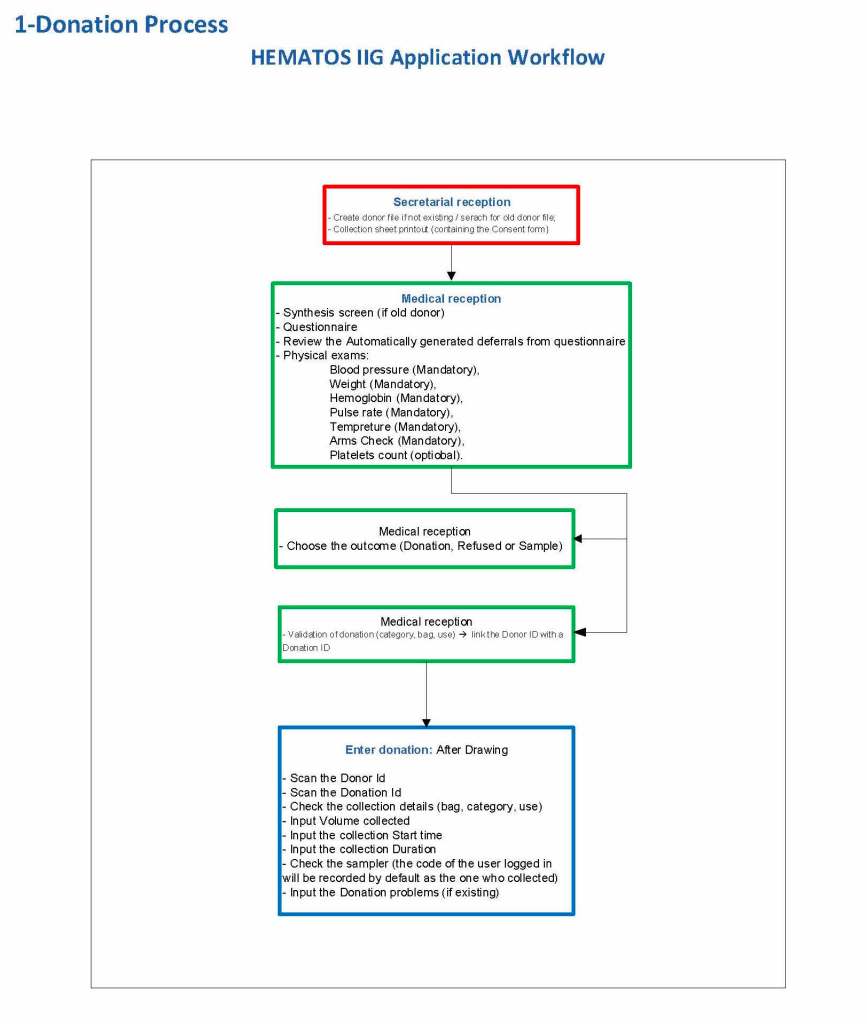

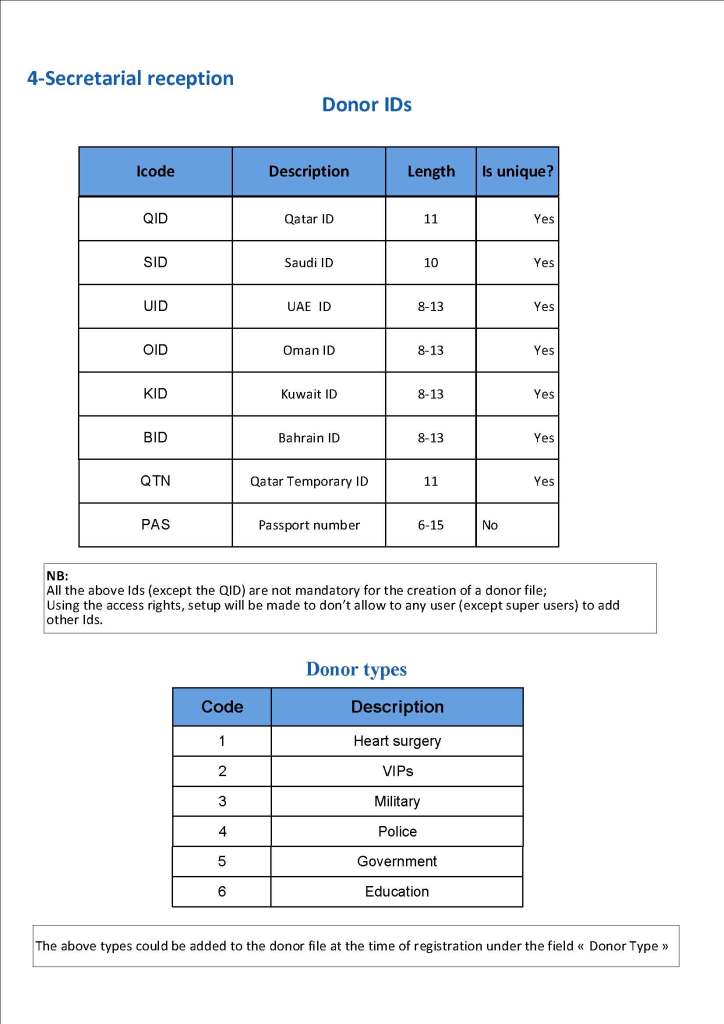


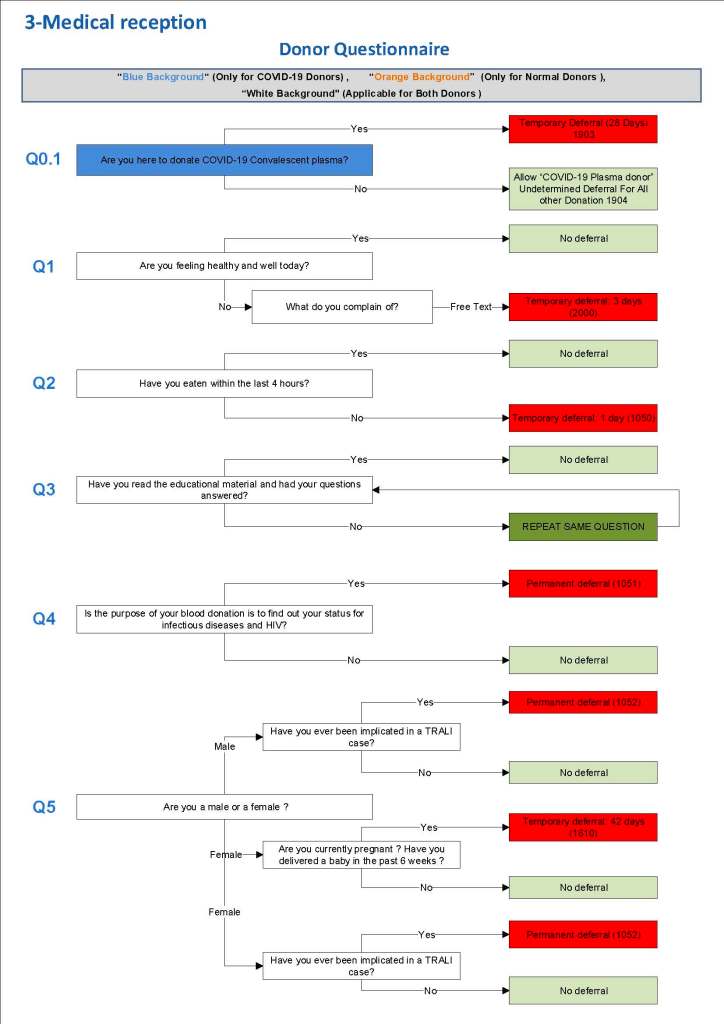


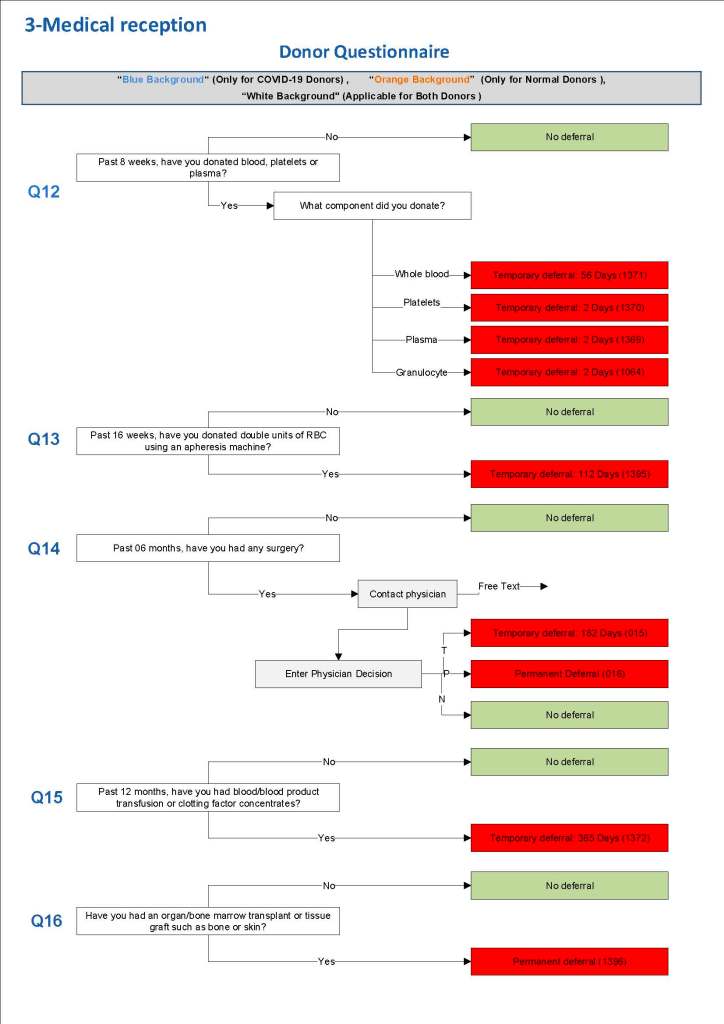
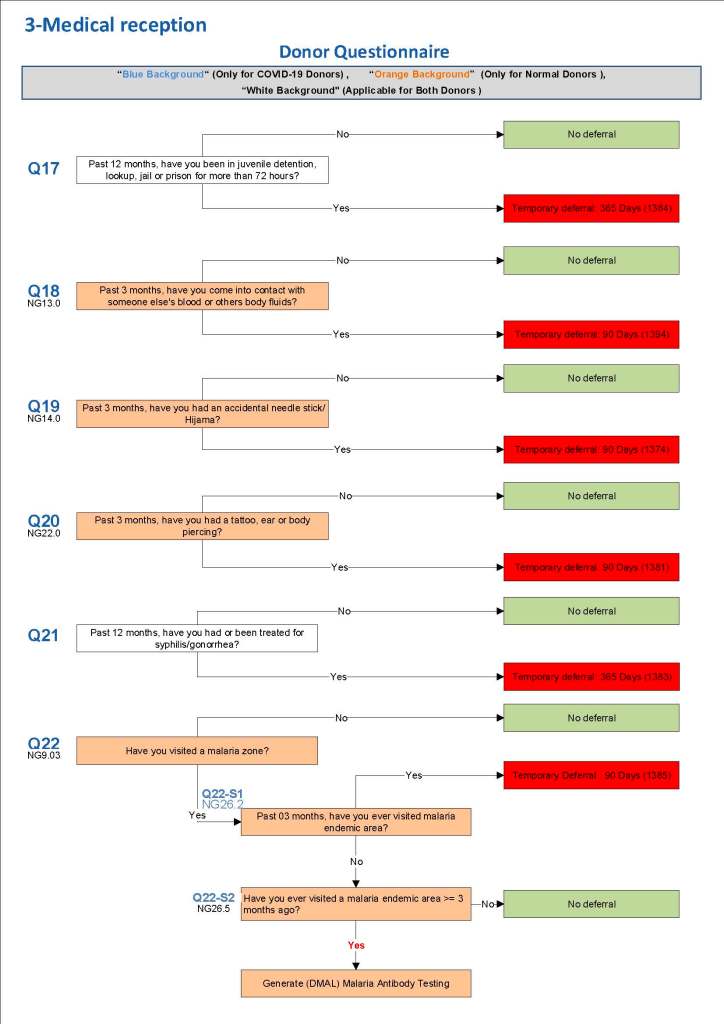
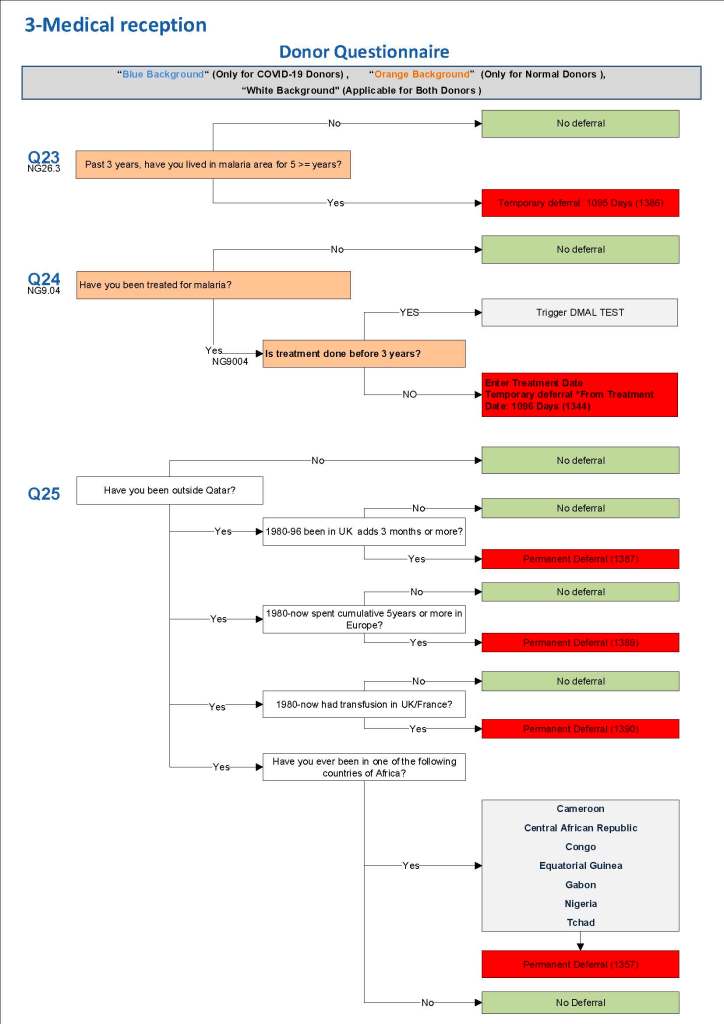

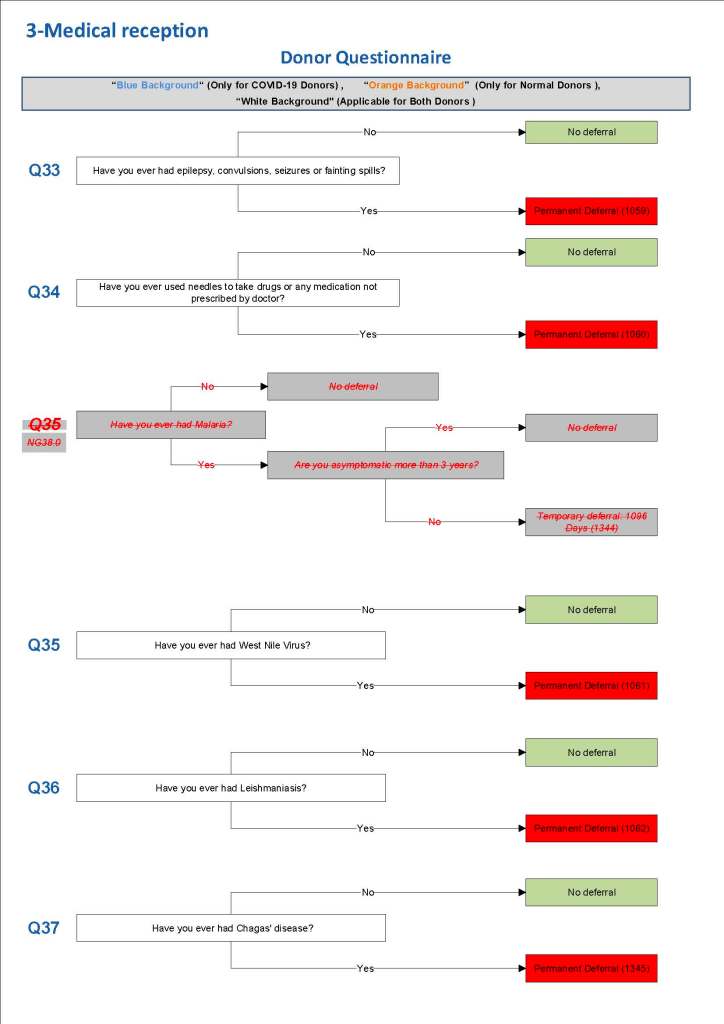
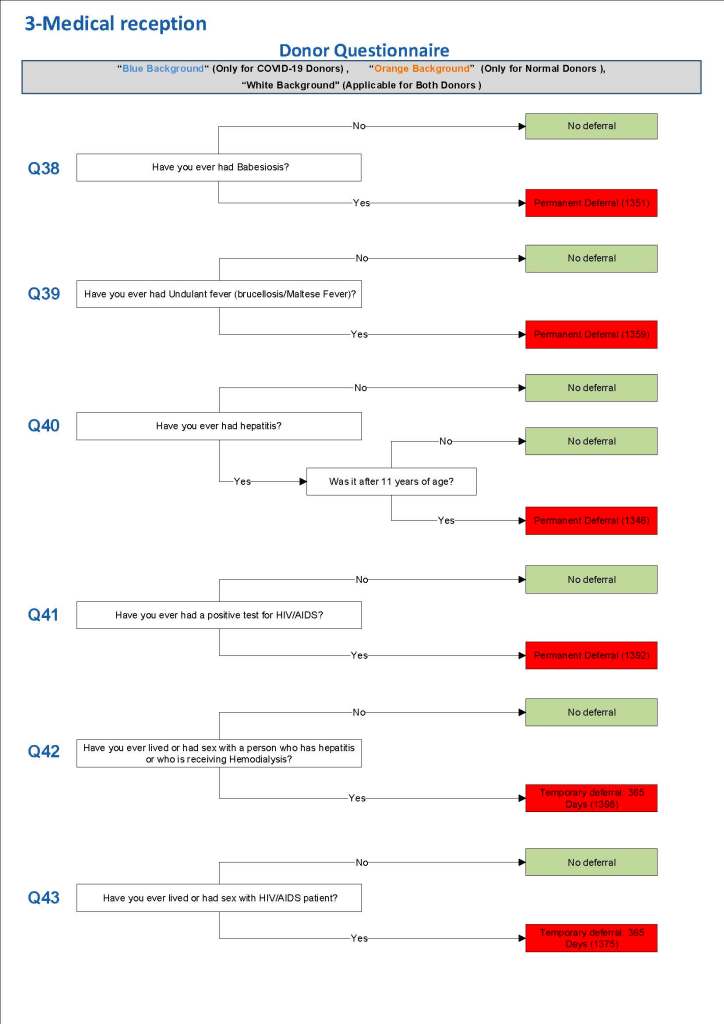

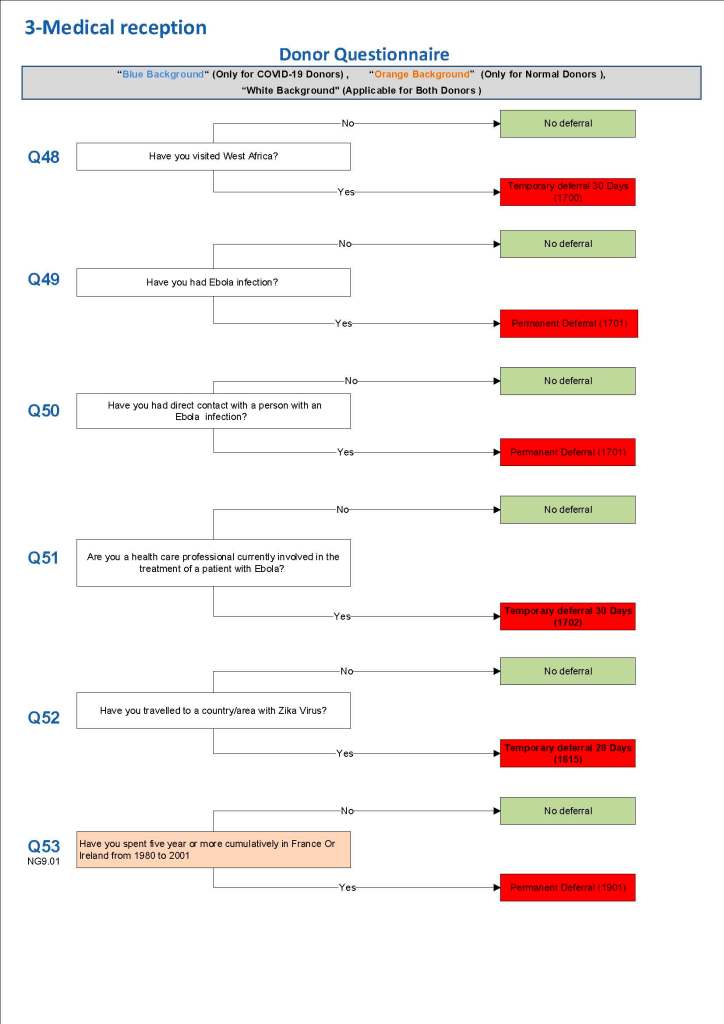
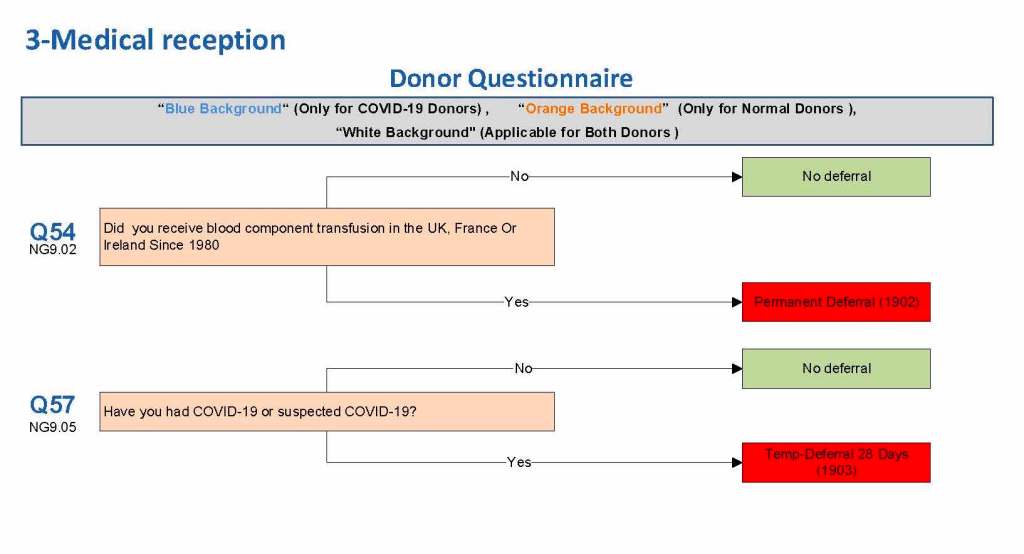
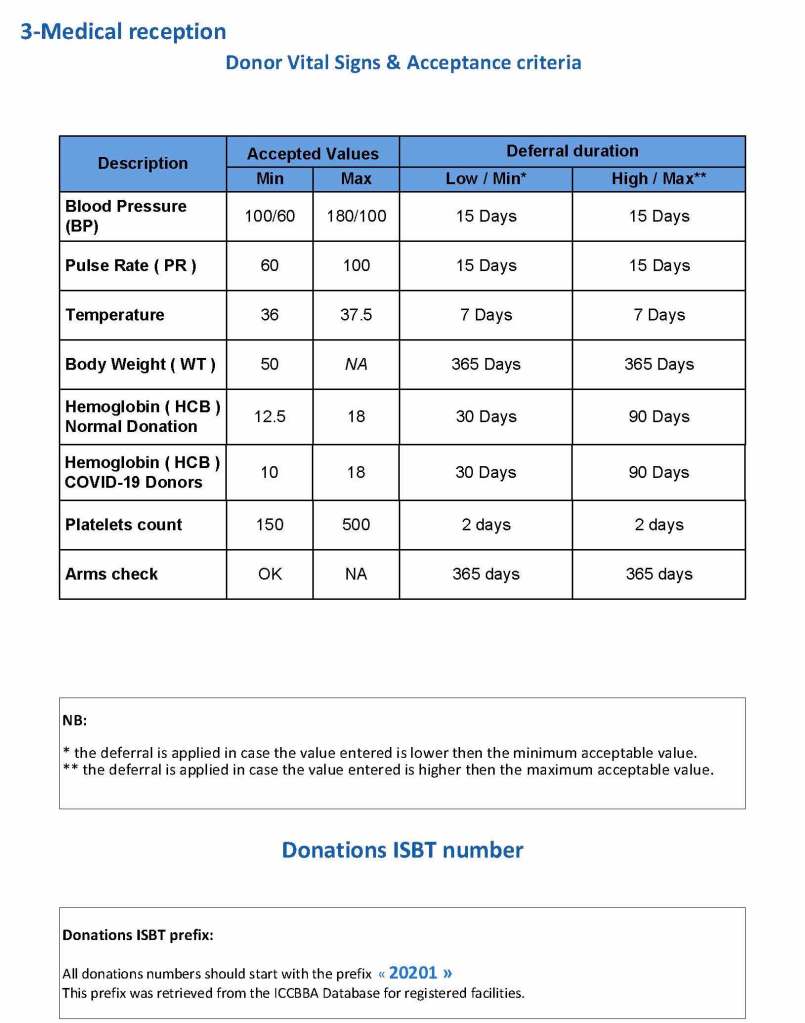
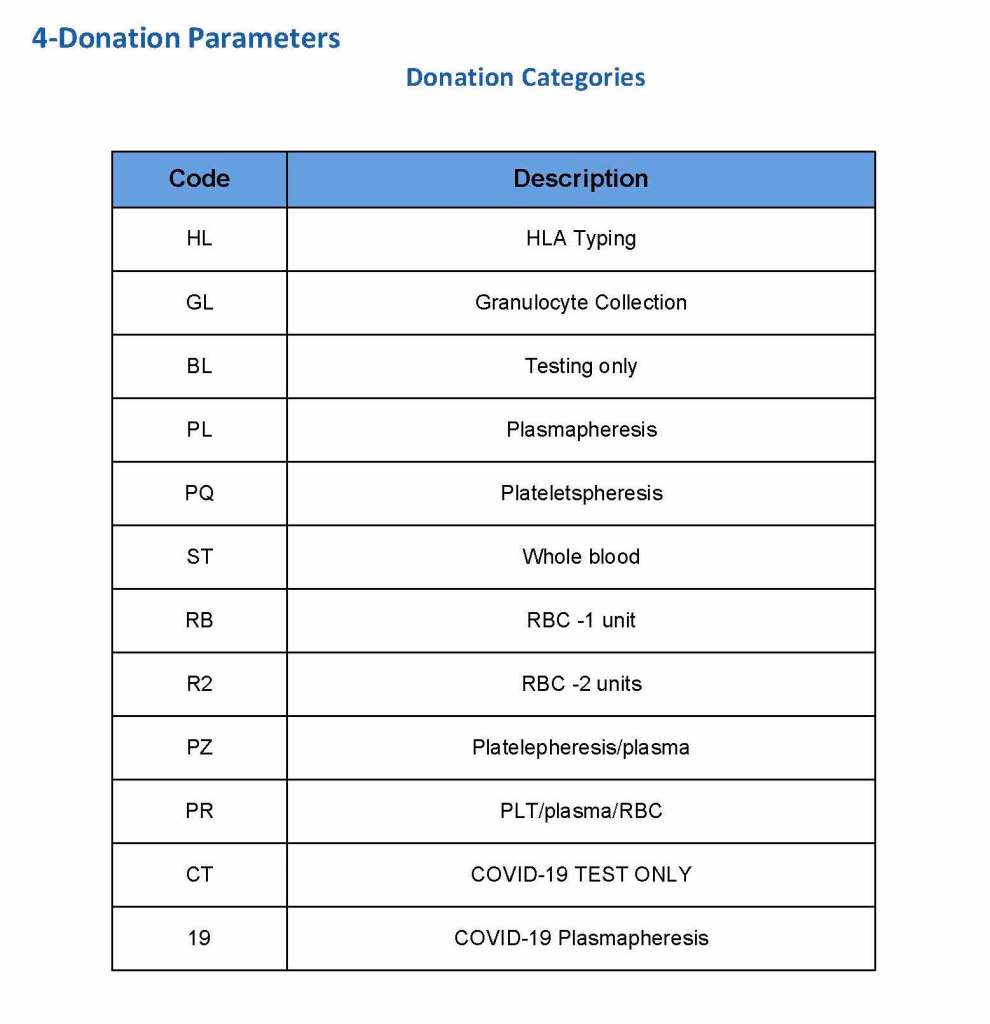
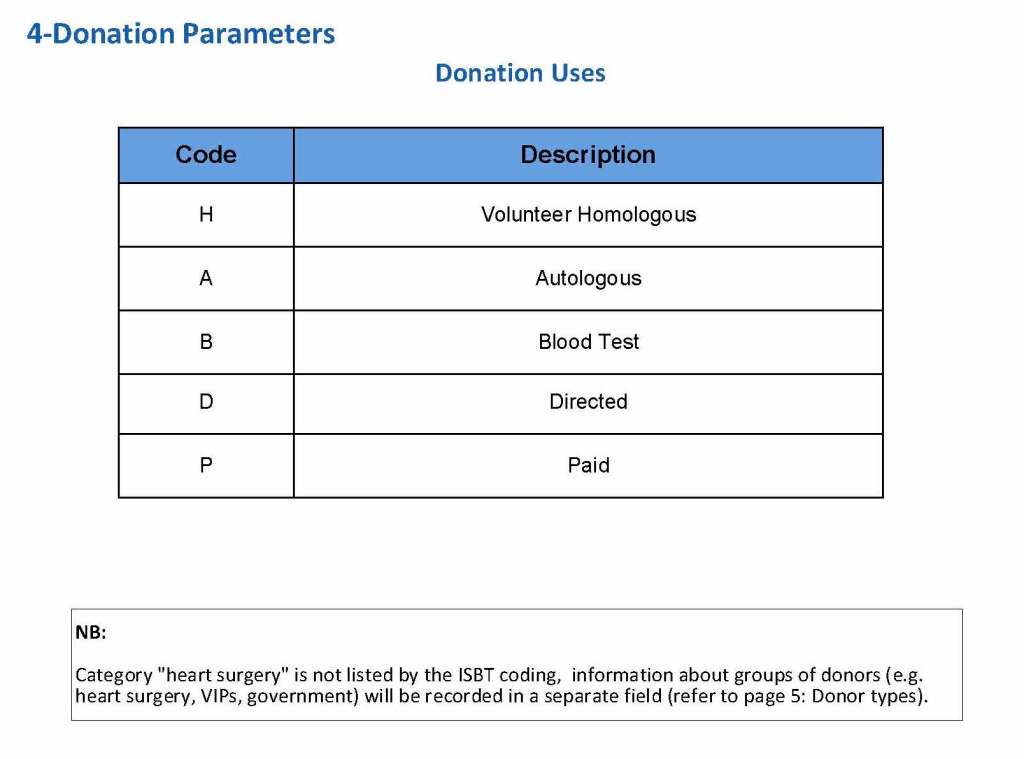
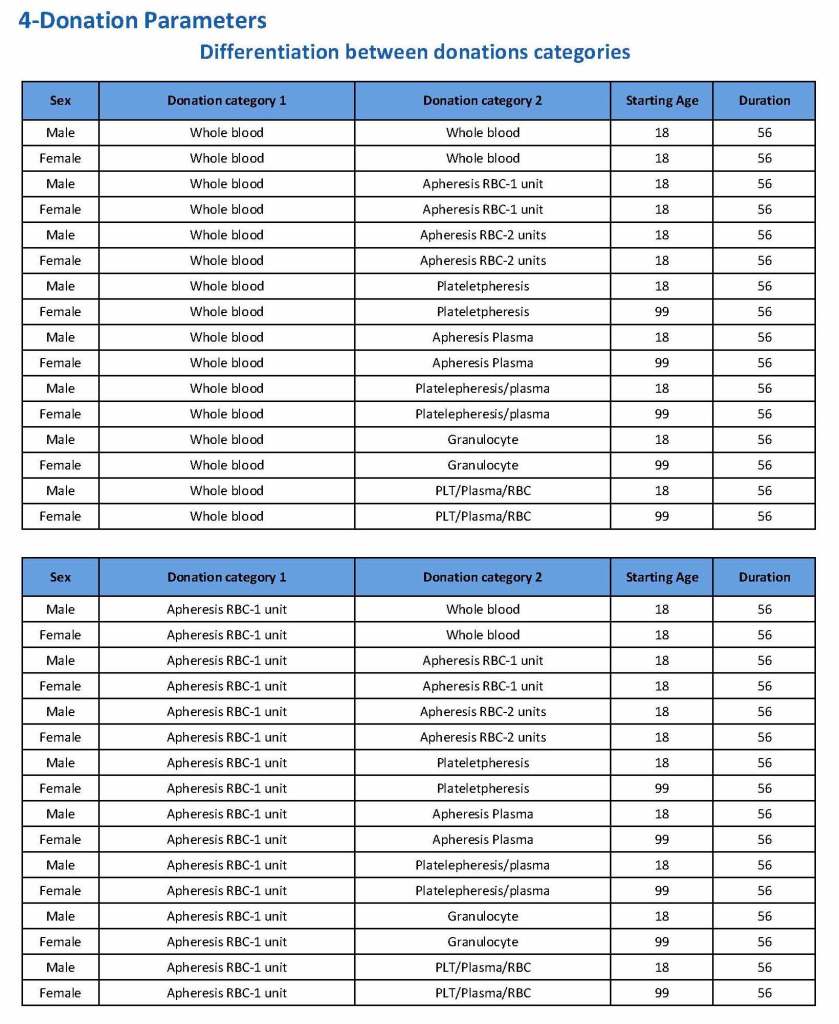
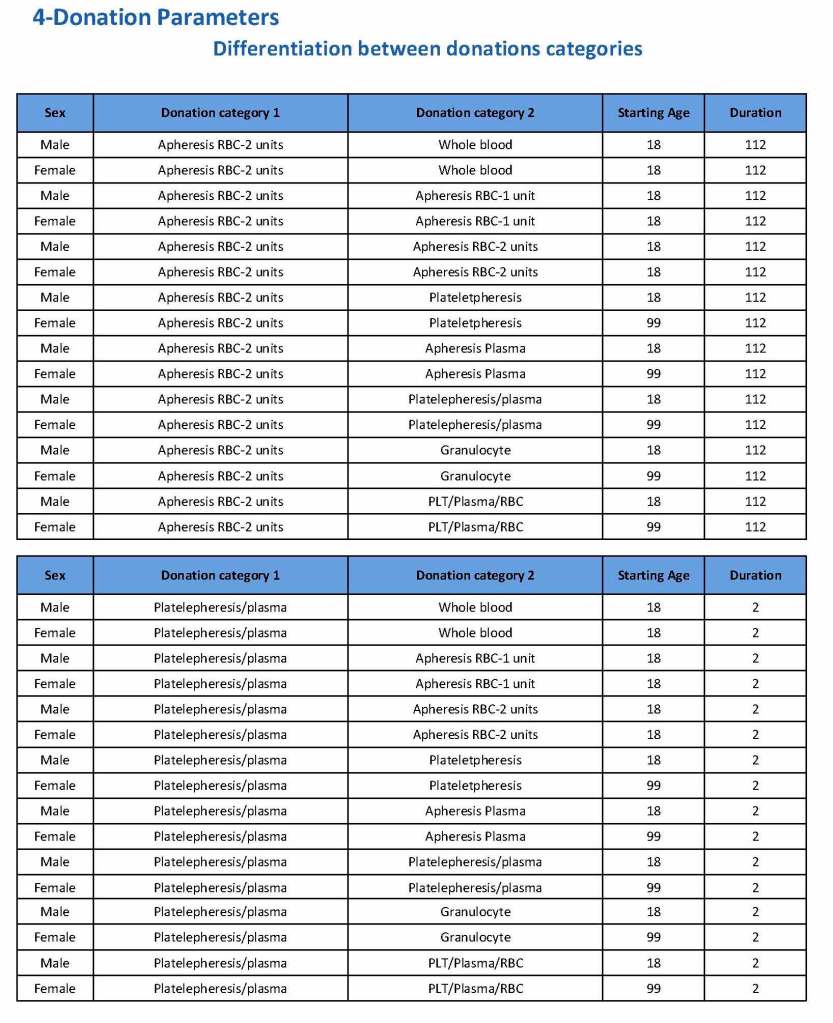
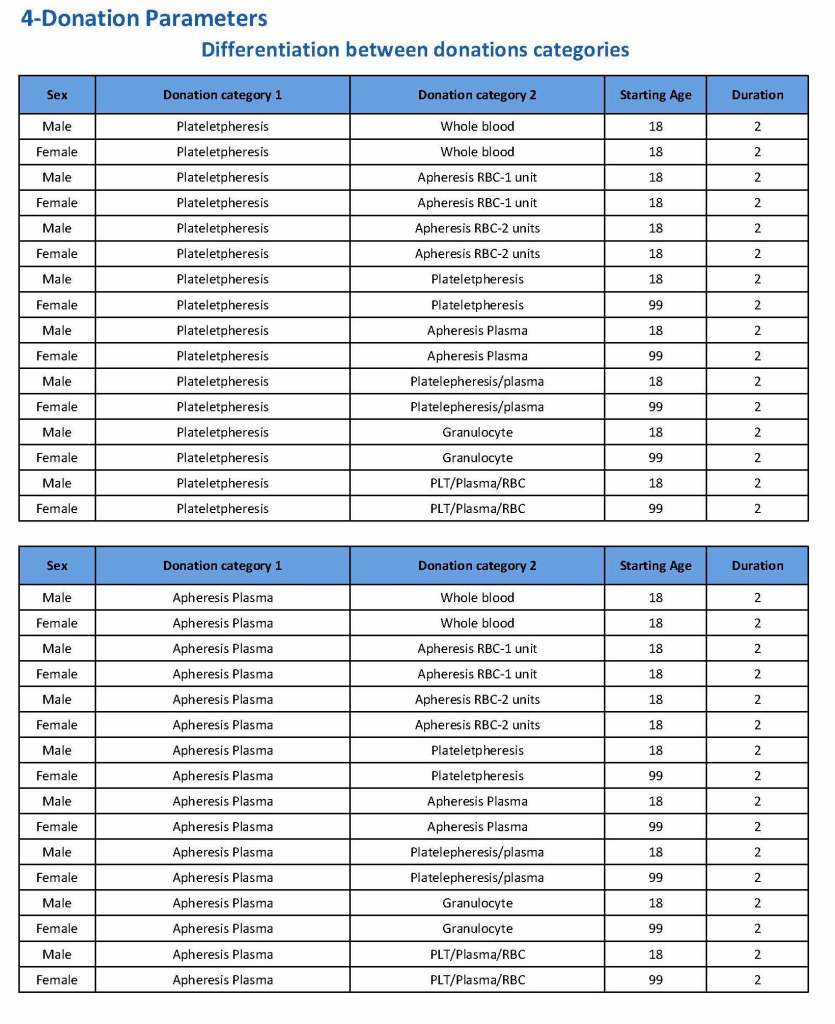
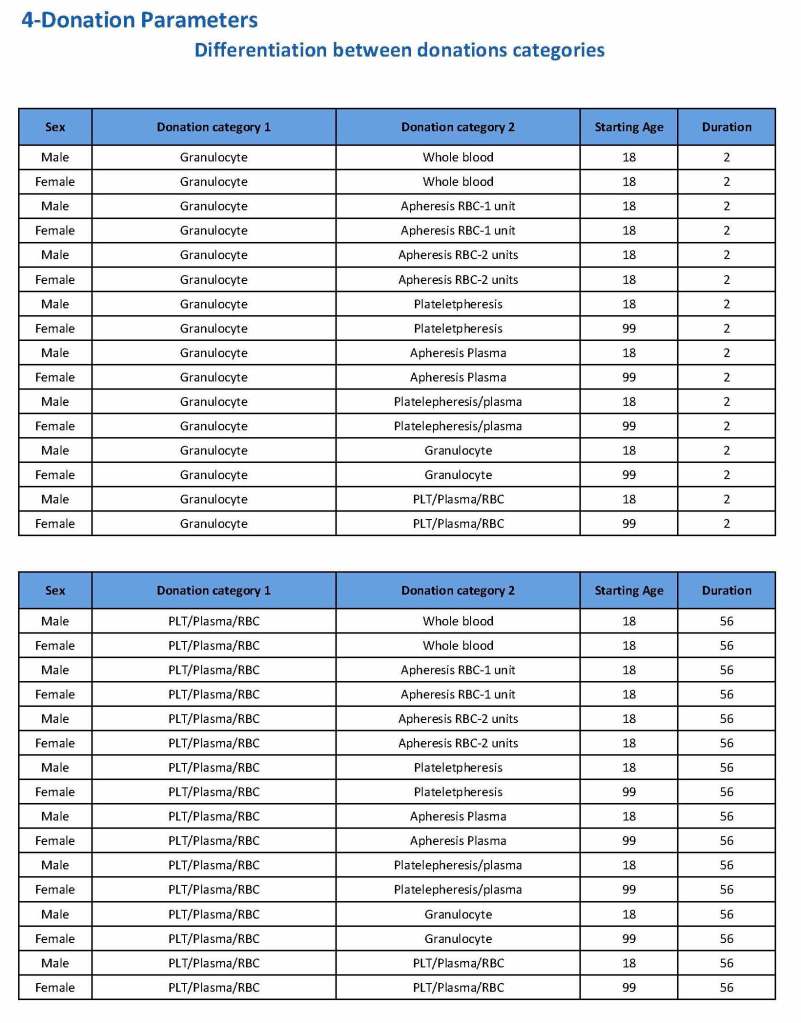
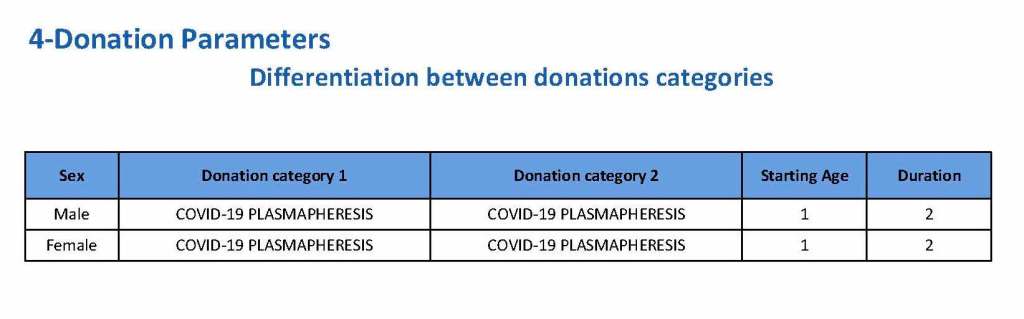
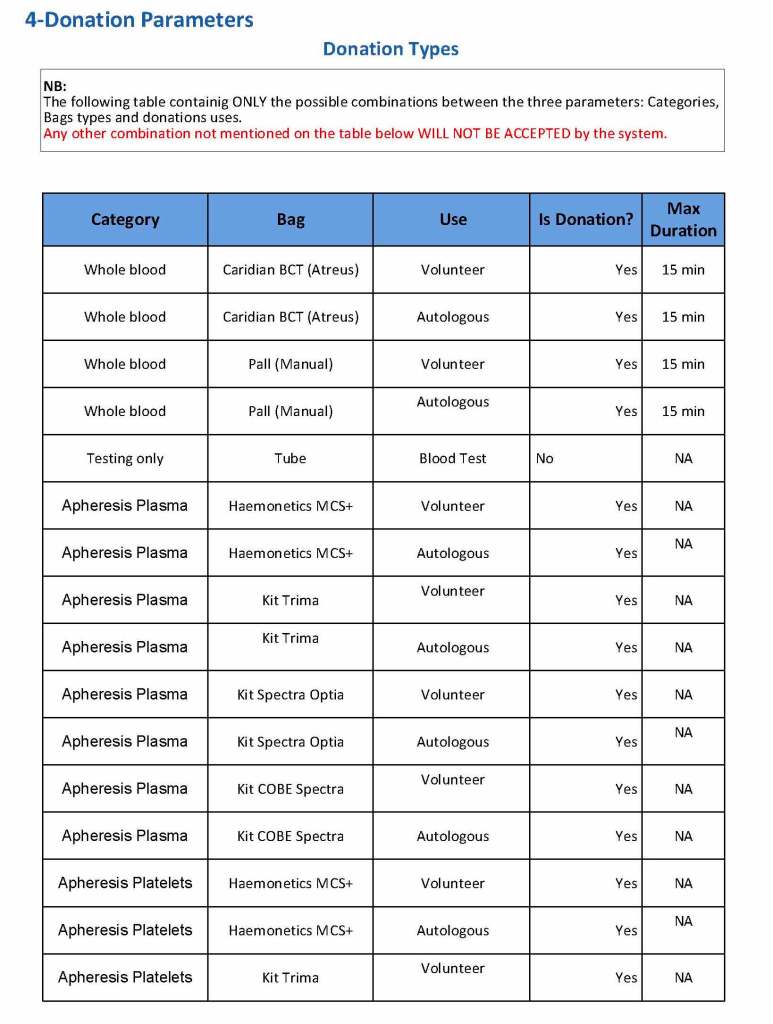
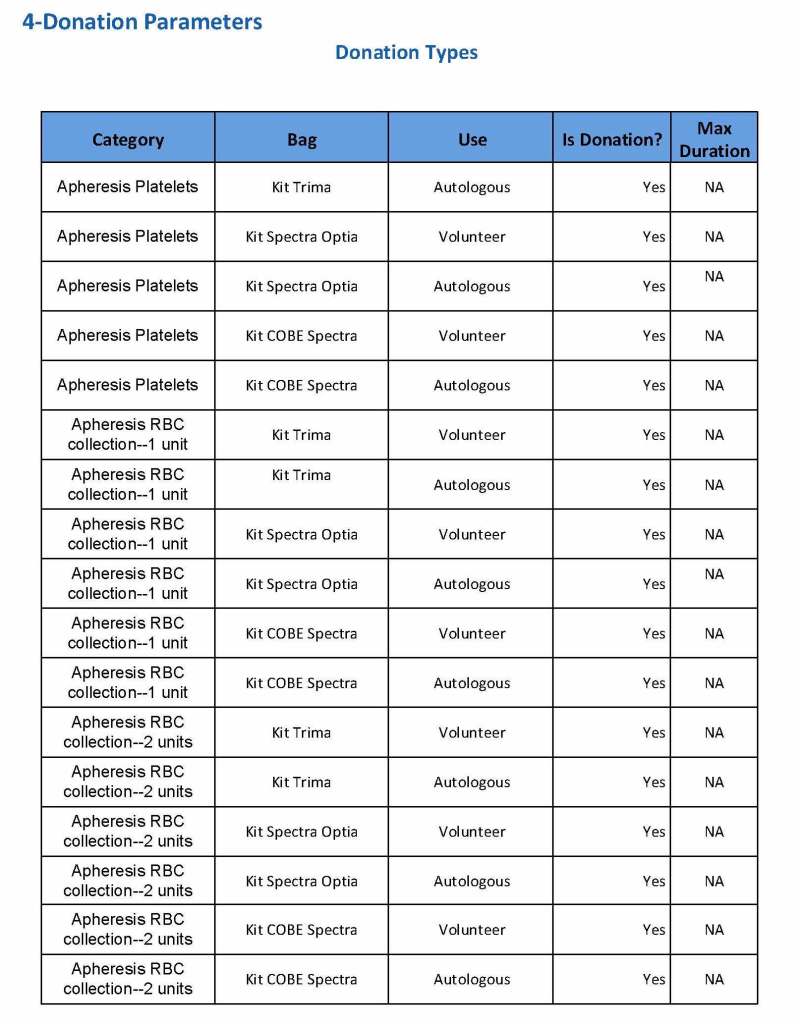
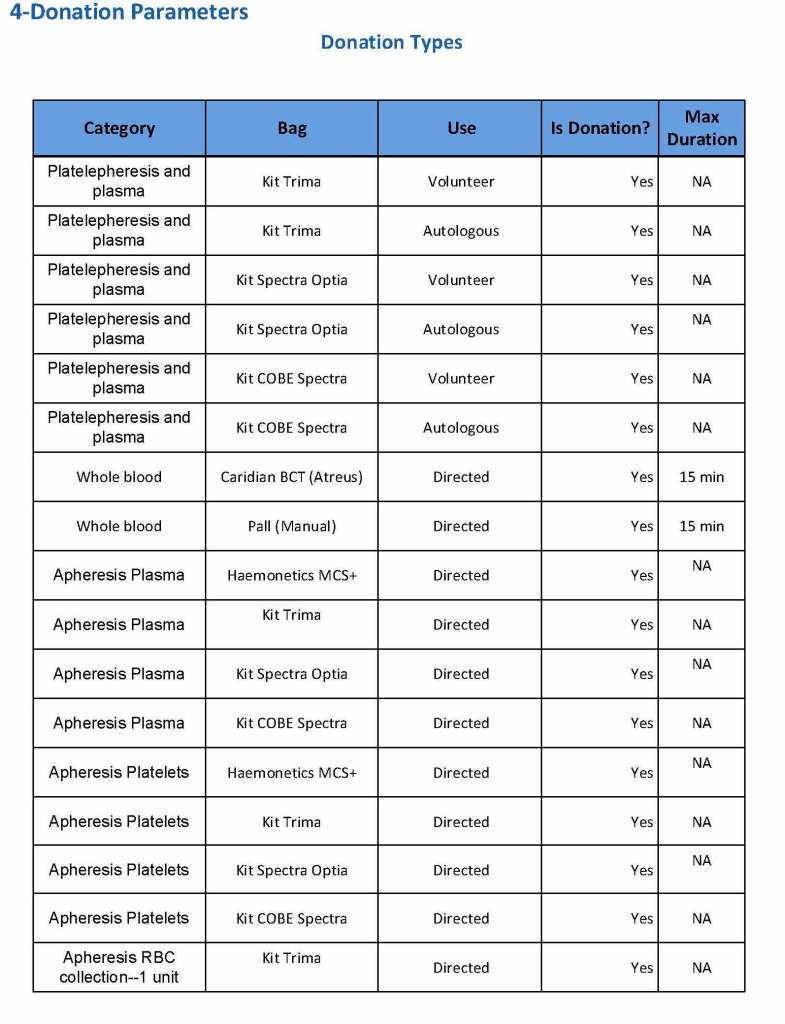
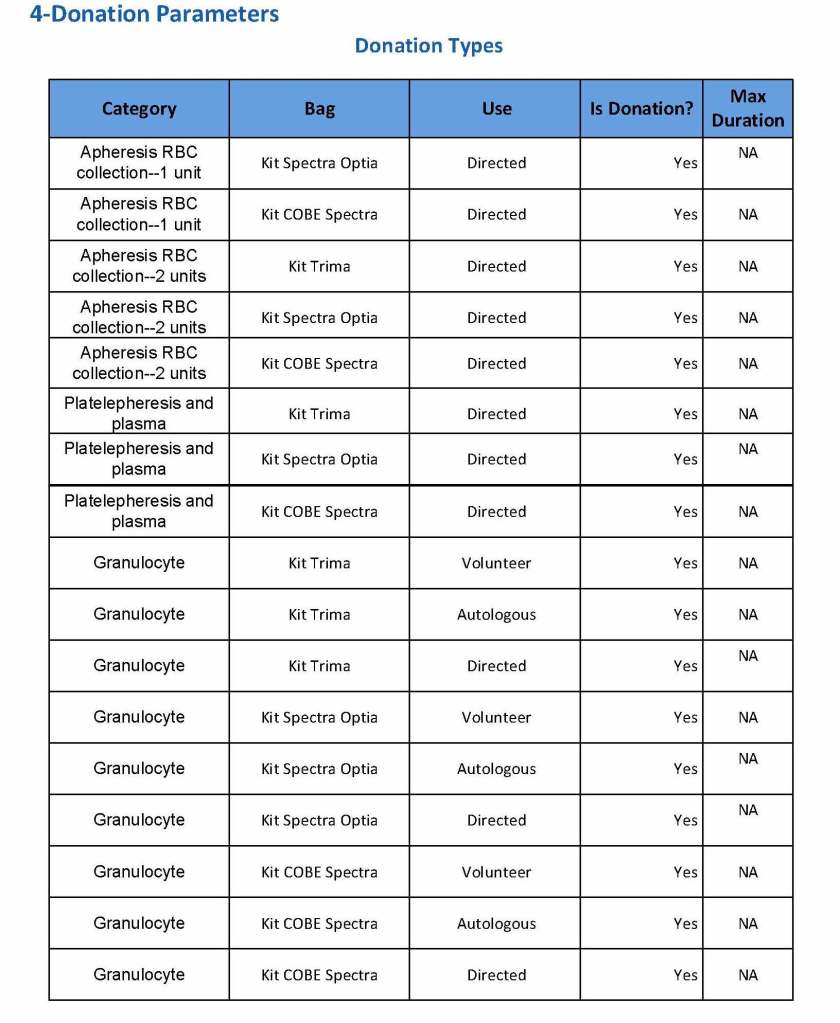
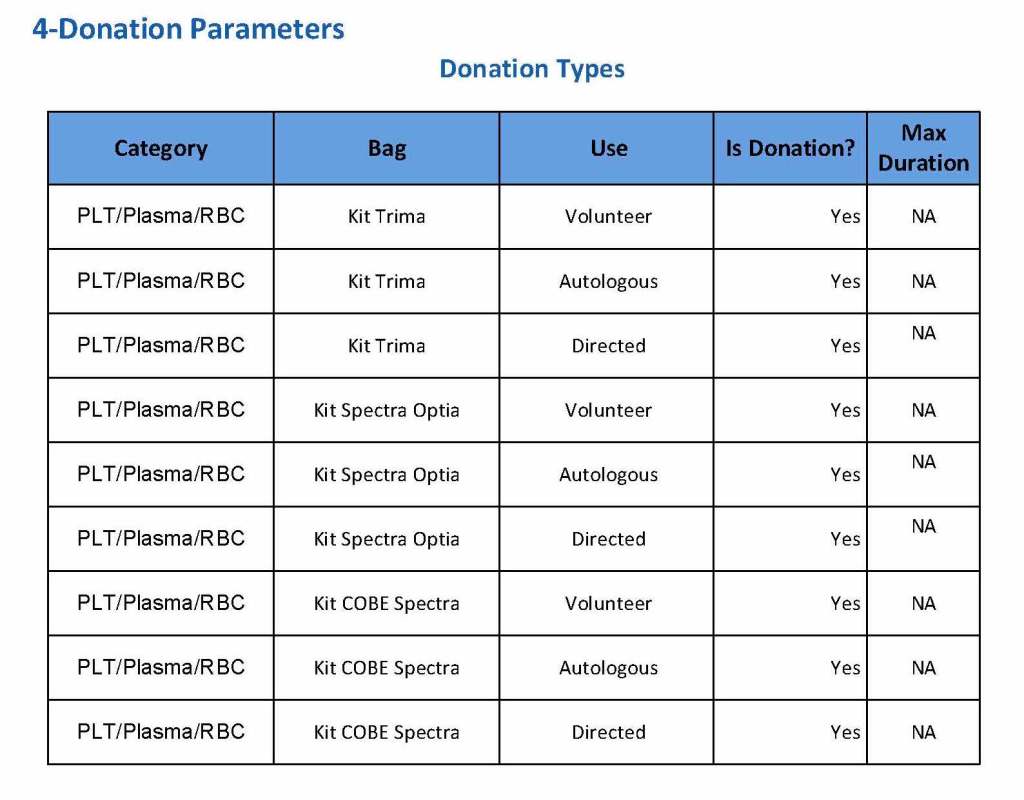
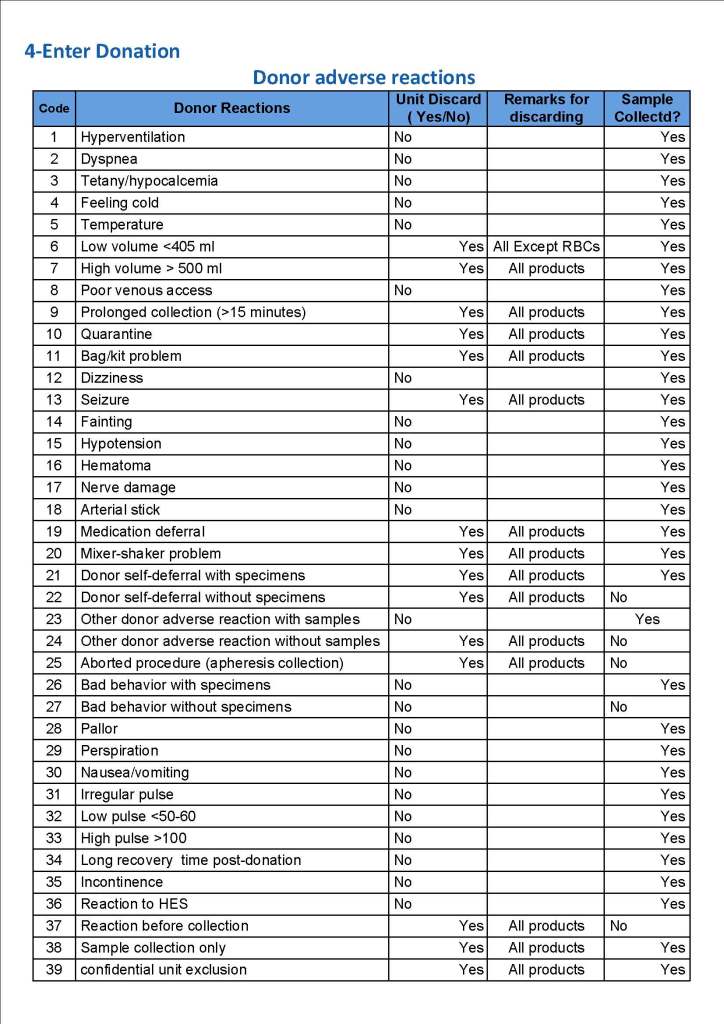
This is the basic COVID-19 convalescent plasma processing workflow specified by me and designed in Medinfo. I wish to thank Medinfo France and Medinfo Qatar for their fantastic support in completing the entire parallel but quarantined workflow in less than two weeks!! This was an extraordinary collaboration.