


Includes all areas: Donor, patient, marker testing, component preparation




I originally adopted this technology because of the plethora of new emerging pathogens. In addition, I have been concerned about unknown pathogens that have not yet been discovered. It is not what we know, it is what we don’t know that bothered me.
It is now 10 years since I started using riboflavin-based pathogen inactivation. Our adoption of the technology was as follows:
Buffy coat platelet pools and whole-blood-derived plasma were both prepared with automated blood component technology, originally with the Terumo Atreus and later with Terumo Reveos system. We were the first site worldwide to use automated production with the Mirasol system.
After 10 years and over 300,000 donor collections, no documented infectious agent transmission has been noted. Our average platelet loss has been 4%. There has been no increase in adverse reactions to plasma or platelets compared the time before we adopted these technologies. Physicians accepted the products readily.
Mirasol adoption allowed us to discontinue irradiation of platelets and extend our outdate to 7 days. We did not need a specific bacterial detection system. Pending regulations in the USA will require stringent bacterial detection processes that are not necessary if a pathogen-inactivation system is being used.
Terumo sent its own engineers to set up and validate the system. They also trained all the staff in the actual pathogen-inactivation processes and helped us to perform the validations.
When adopting pathogen inactivation, we compared Mirasol with its competitor and selected it for the following reasons:
In our system, our goal was rapid processing of units. With Mirasol, we did not have remove the riboflavin from the final product. The competitor product requires at least 6 hours post-treatment to remove the psoralen agent. We could immediately use the Mirasol product after treatment!
In our Reveos-Mirasol system, we can process whole blood into packed red cells in SAGM, buffy coat platelets, and plasma in a total of 5 hours including all testing with Mirasol treatment and platelet additive solution PAS.
We originally used the system manually, but in 2013, Terumo in conjunction with Medinfo Hematos IIG developed an interface to the Mirasol illuminator. The latter device would transmit the successful completion of the illumination to the software. Any errors in the illumination would block release of the blood component from Medinfo. Medinfo also monitored the component volumes to prevent treatment of units outside Terumo’s recommendation ranges.
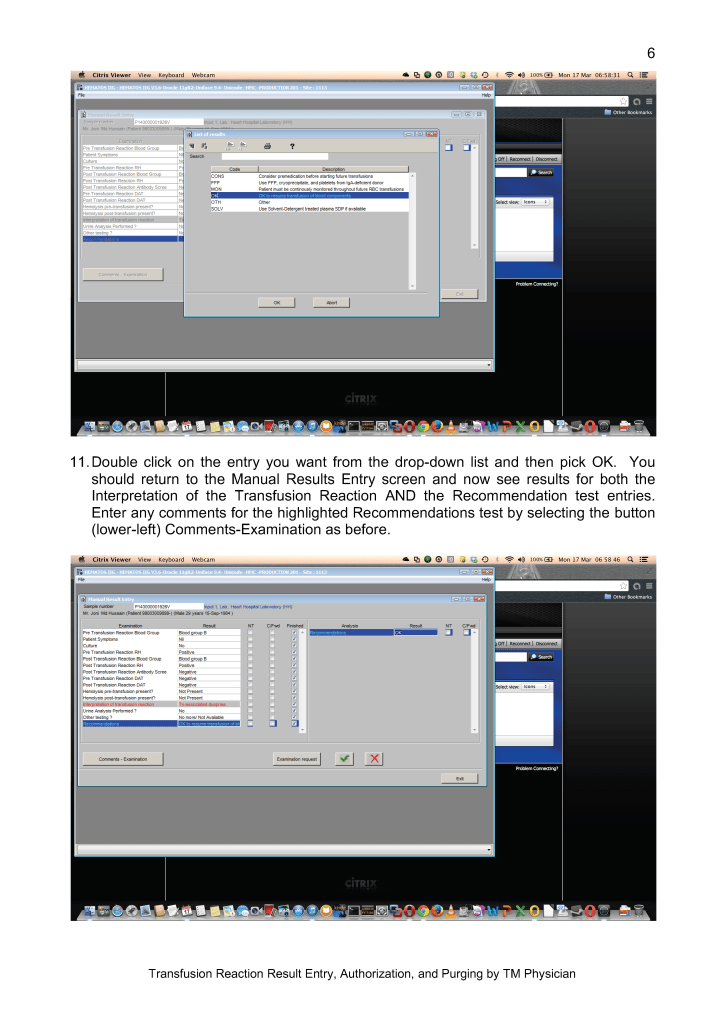
Adoption of platelet additive solution PAS gives us a final product with minimal residual plasma which potentially can reduce plasma reactions and TRALI/TACO. It also minimizes our need to reduce the volume of platelet components for pediatric patients, especially in cases with ABO-incompatible plasma
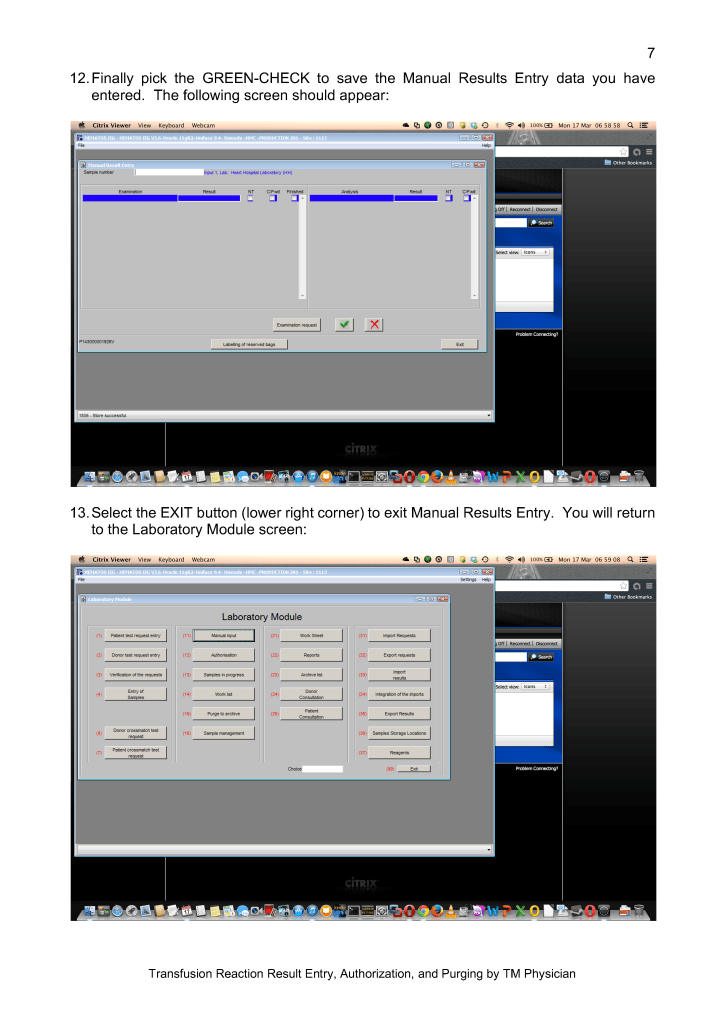
Adopting any system of pathogen inactivation requires meticulous monitoring of component volumes to ensure they are within the range for the treatment. The use of a blood bank software greatly facilitates this.
We make both buffy coat and apheresis platelets. The change from plasma-suspended to PAS-suspended platelets went smoothly. Special training for Trima apheresis staff to use the new processes was provided by Terumo.
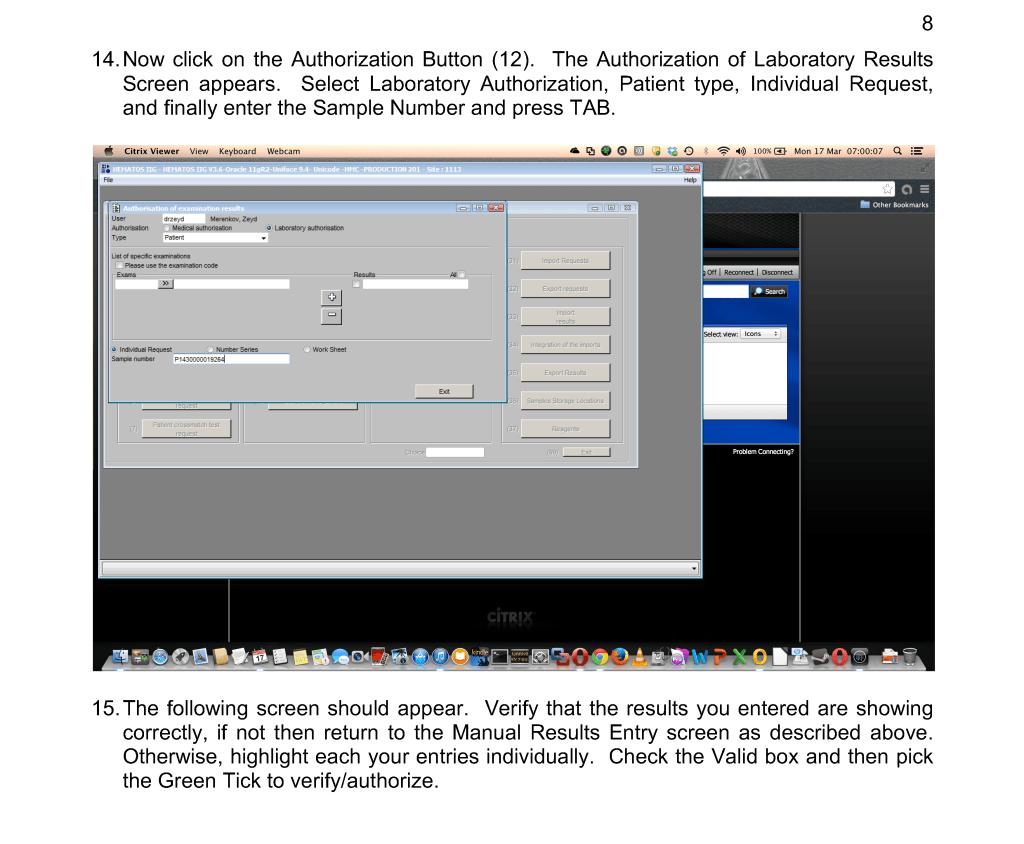
Throughout this time interval, Terumo has provided excellent technical support and educational activities for all staff. Despite the COVID pandemic, Terumo has been able to deliver supplies to meet our needs so there was no interruption in production.
We started COVID convalescent plasma CCP production at the end of winter 2020. We set up a parallel but separate quarantine system of collection and processing, originally manual but later controlled by the dedicated blood bank software Medinfo Hematos IIG. All CCP units have been treated with Mirasol.
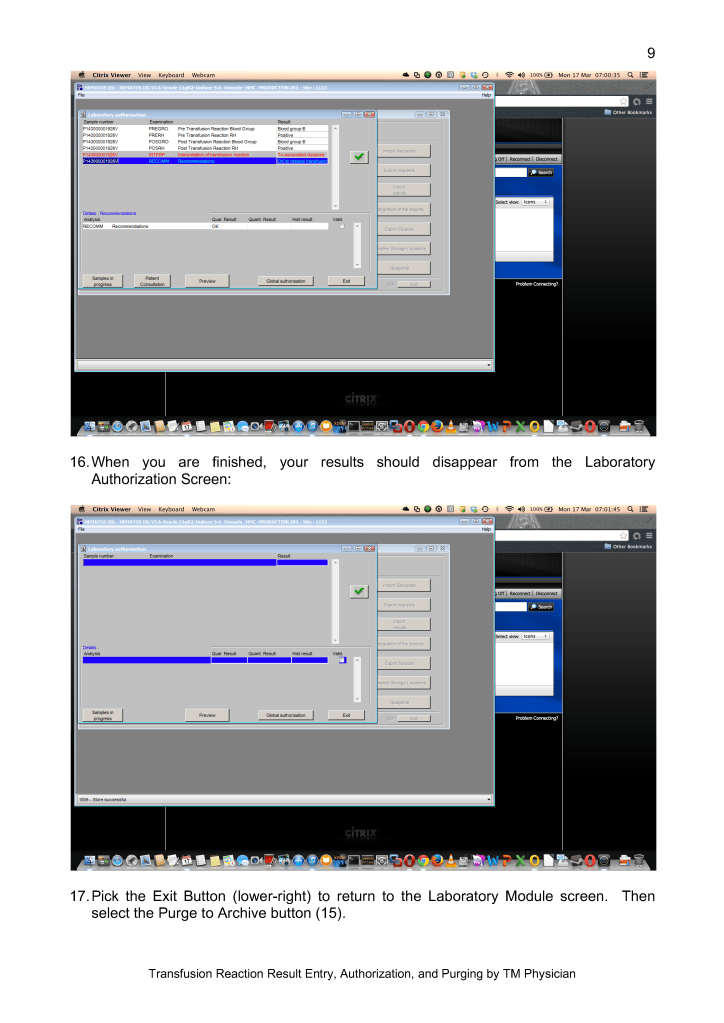
In the future, I hope Mirasol will close the loop by providing a pathogen-inactivation process for red blood cells so all components can be treated. The CE mark for pathogen inactivation of whole blood is exciting and I hope that component preparation from this product will be offered.
In summary, our blood bank system had an excellent, synergistic relationship with Terumo and Medinfo to provide the highest quality product that is currently licensed. I hope we will all continue to work together to improve the patient care.


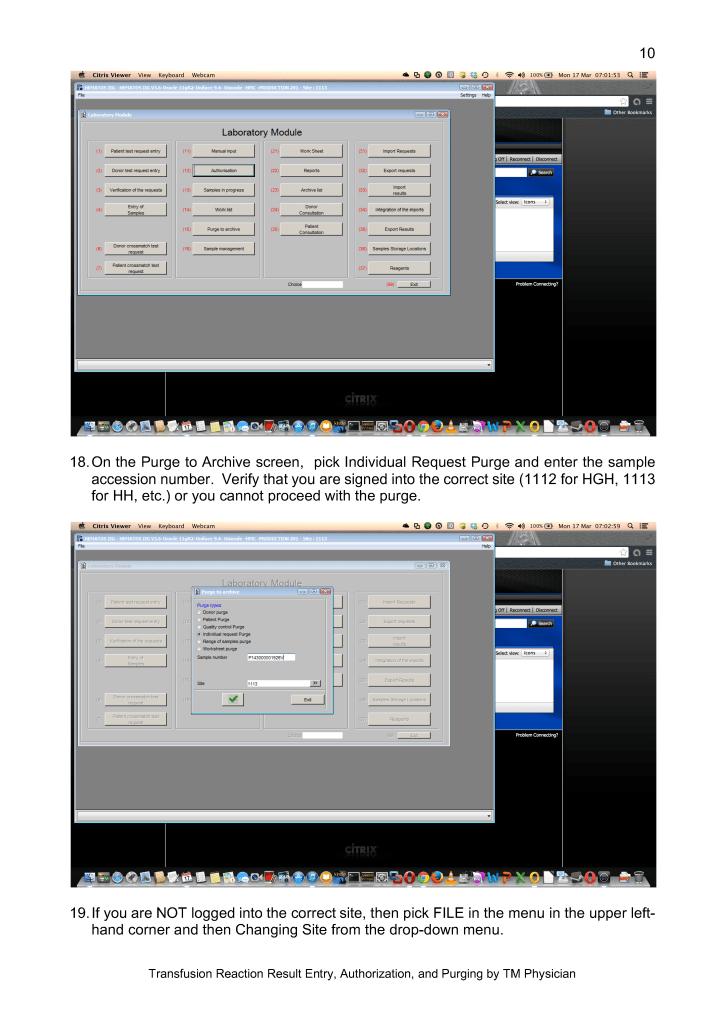
The COVID-19 pandemic imposed new challenges to our system. In general, these could be divided into:
There were fewer donors in the early phase and the nurses also had to add a large number of donor plasmapheresis collections for COVID convalescent plasma CCP. Still they had to maintain all donor and therapeutic apheresis services with no increase in staff. Although elective procedures had been cancelled, there were still obstetrical, oncologic, and trauma services in full action.
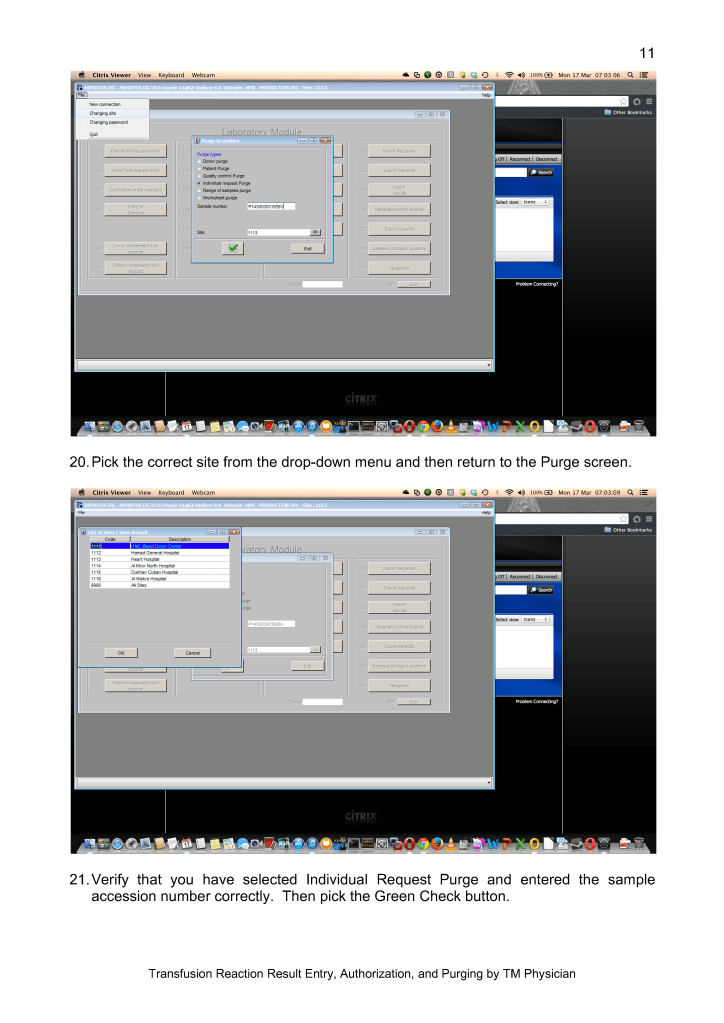
Many of our staff were on leave when the borders were closed. Some had to wait months before they could return to work. Others had COVID-19 infection and were quarantined for several weeks. This further reduced staffing. We could not just hire outside staff since considerable training is involved in these processes.
I dedicated a separate donor collection space for the CCP program away from the regular donors as well as a quarantine processing area. Similarly, the CCP plasma was kept segregated from the regular plasma supply and a specially designed location was identified for release of this product. Working for this program diverted resources from blood collection to this special project, again without increasing resources.
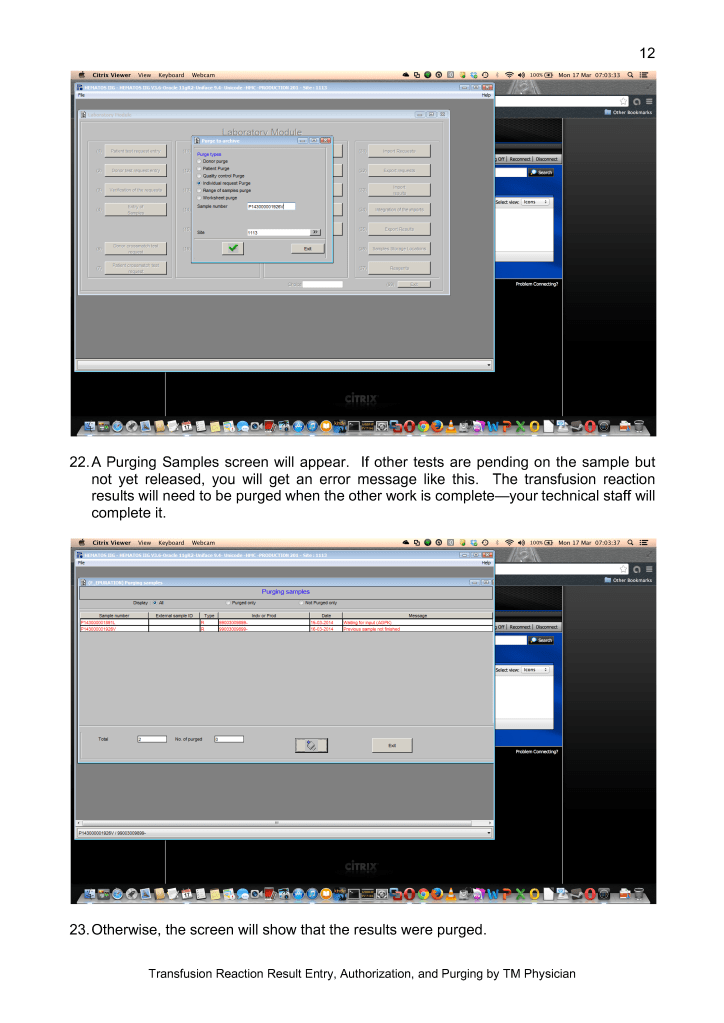
With disruptions to shipments of supplies, including the Reveos whole blood kits and Trima donor apheresis sets, we had to rely on our large in-home inventory until the situation stabilized. We prescreened the CCP donor candidates before we would collect them to avoid wastage of kits.
Fortunately, our throughput was minimally affected because our equipment and processes had always stressed speed. We used single-well NAT testing to minimize the need of additional runs. Also, we used Reveos automated component processing to greatly speed production (one Reveos can process four whole blood units in about 23 minutes or about 12 units in 75 minutes.) One technologist could operate all 4 of our machines simultaneously and perform other tasks while the machines were working.
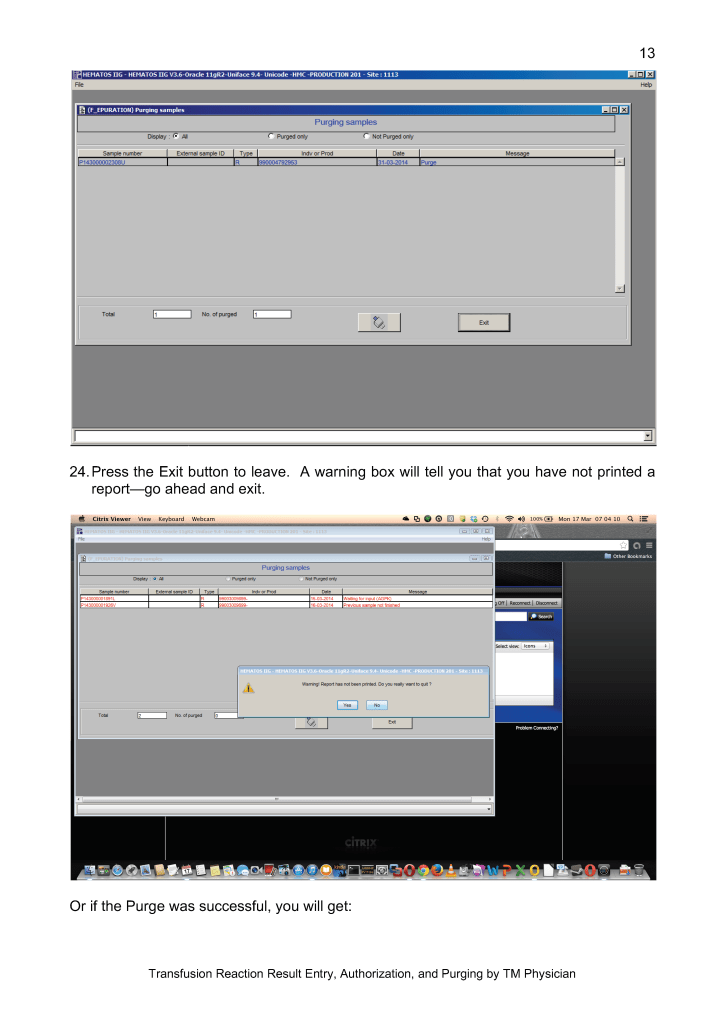
In the system I developed in Qatar, we could complete processing into components (RBCs, buffy coat platelet pools, leukodepleted plasma), all marker and immunohematology testing, leukoreduction of the pools and RBCs, Mirasol pathogen inactivation, and platelet additive solution in as little as five hours.
In rapid turn-around events, it is most helpful to have a robust blood bank computer system that can scale to the challenge. Also, it must mercilessly enforce all the rules starting with donor qualification, screening, collection through testing and production. At times of emergency, it is difficult to meet Good Manufacturing Processes manually.
I had built parallel separate donor collection, donor processing, and transfusion service/hospital blood bank processes specifically for CCP and had to staff them with available personnel, limited our capability to process regular donors. The blood bank computer software restricted CCP use to designated physicians and transfusing locations. For those interested, there is a separate series of posts about the CCP project and its implementation in the dedicated blood bank Medinfo HIIG.
COVID-19 vaccinations should have minimal effect in donor qualification since mRNA or antigen-based ones do not cause donor deferral. Live attenuated COVID vaccines will defer donors for 2 weeks by current rules—the same as other live vaccines. Donors who had previously received CCP will be deferred for three (3) months after last receiving this product.
In summary, the COVID pandemic reduced staffing and affected donor recruitment. We had production mitigations to maximize throughput. The system was stressed by the reduced staffing and special demands to produce CCP. However, the extent of our automation allowed us to maintain throughput throughout the crisis.



Principle:
There are several different types of comments in HIIG:
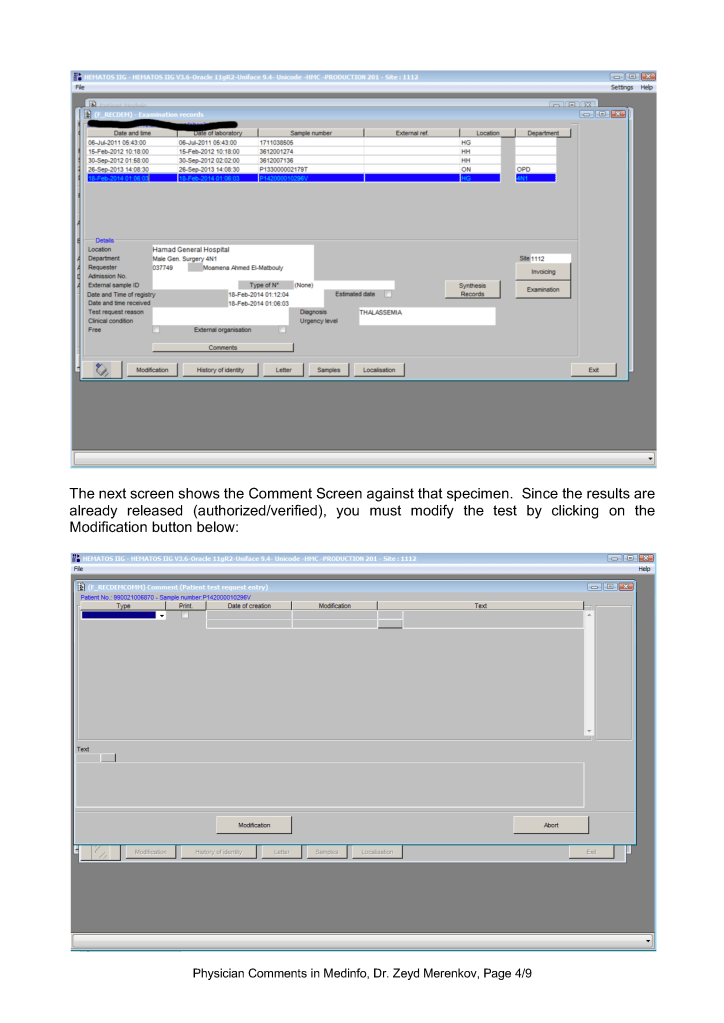
Physicians may enter any of these comment types. Comments may be entered before or after a test is authorized/verified. If entered after authorization, the test must be modified to accept the comment. Only result comments are visible in the patient’s medical record.
Policy:
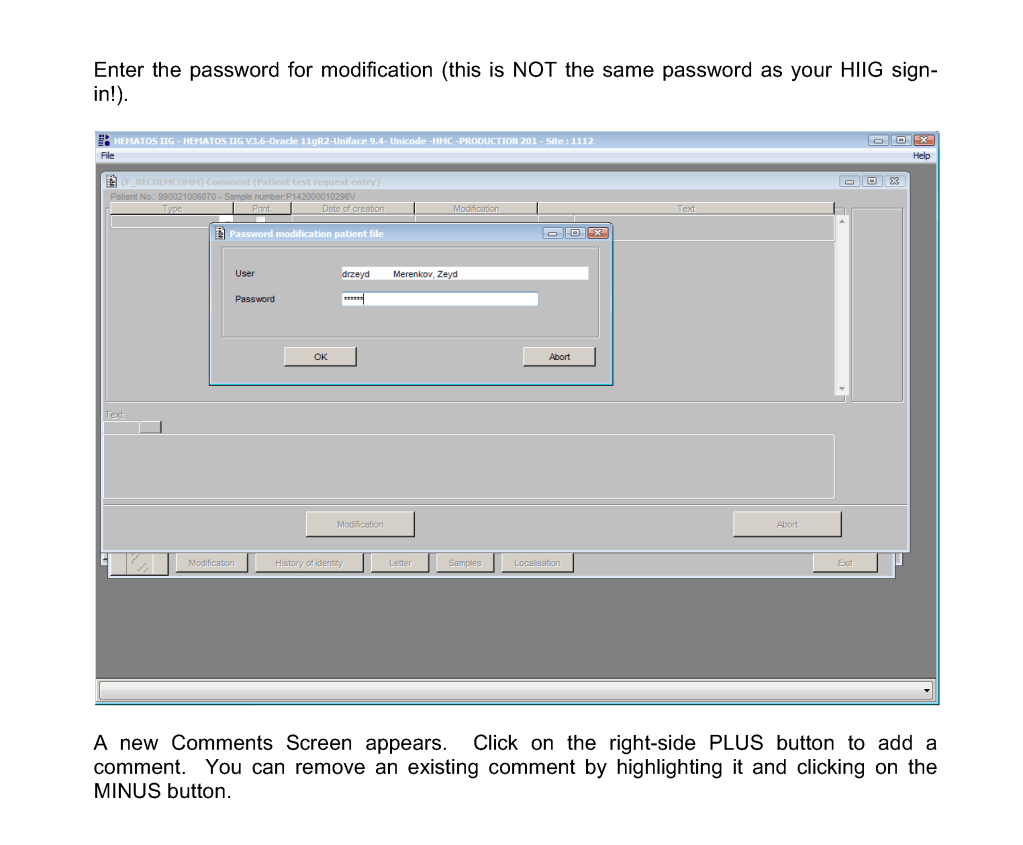
Procedure—Patient Result Comments After Results are Authorized/Verified:
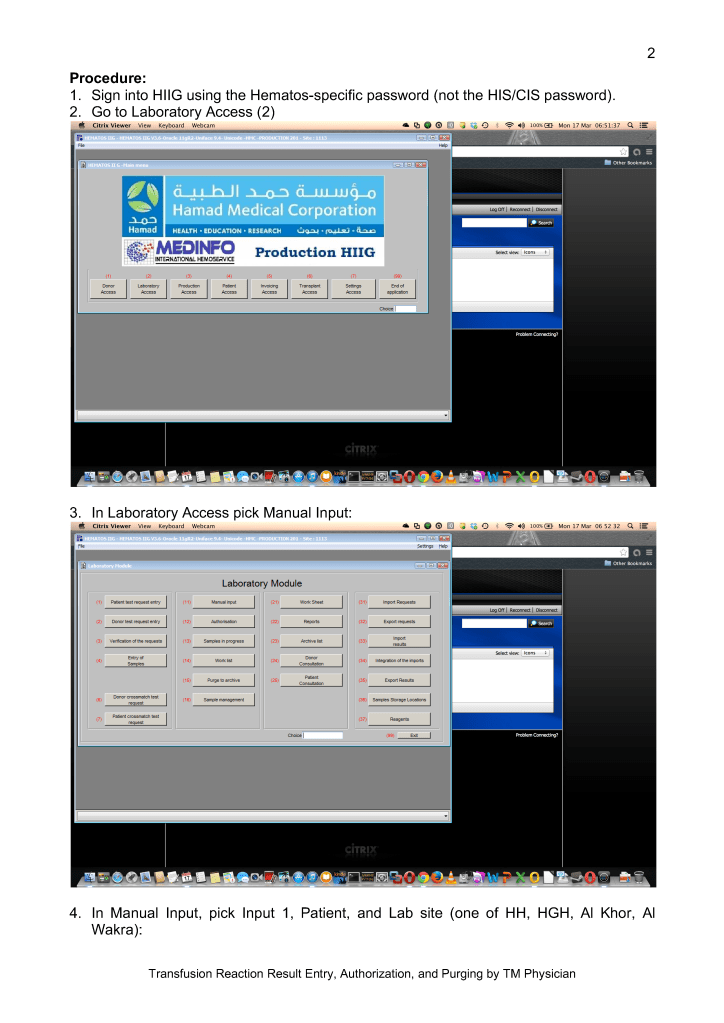
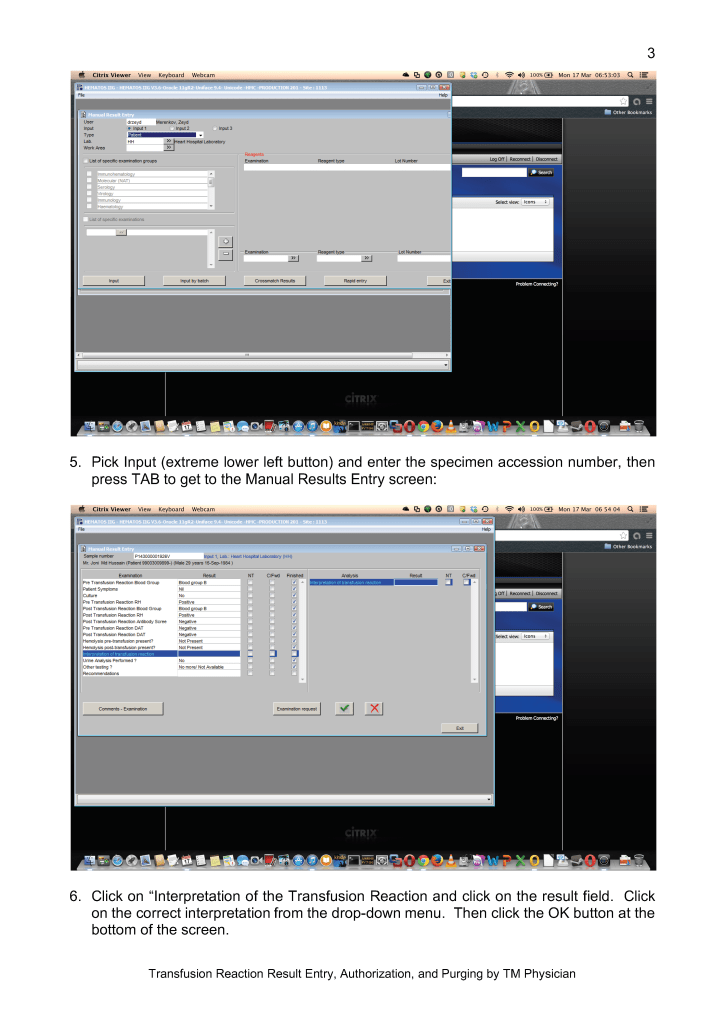
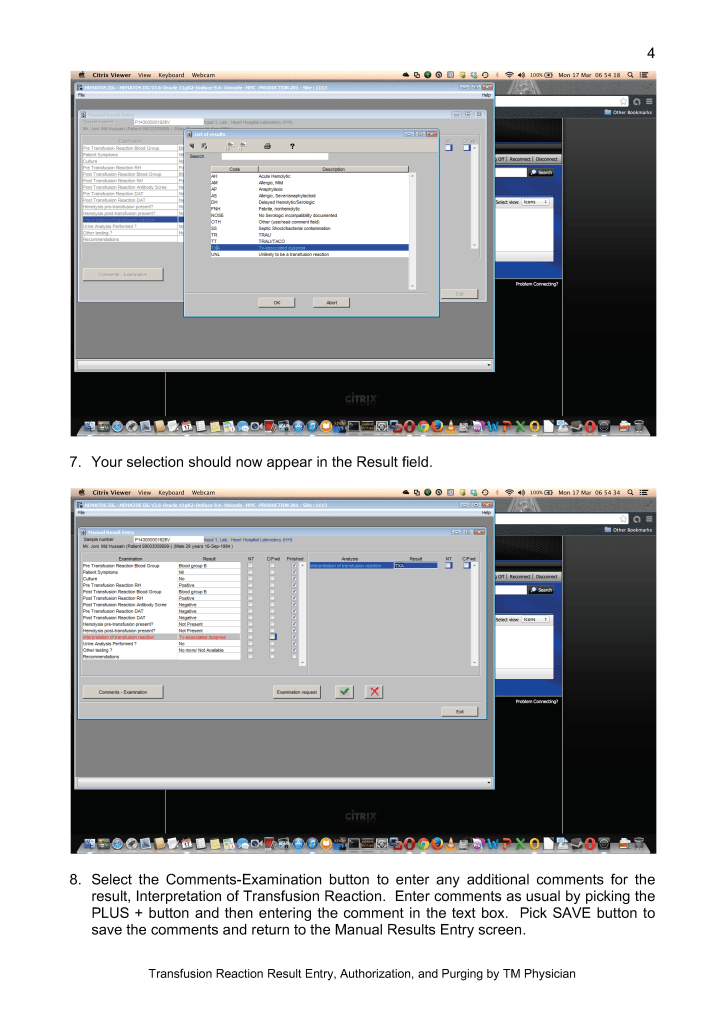
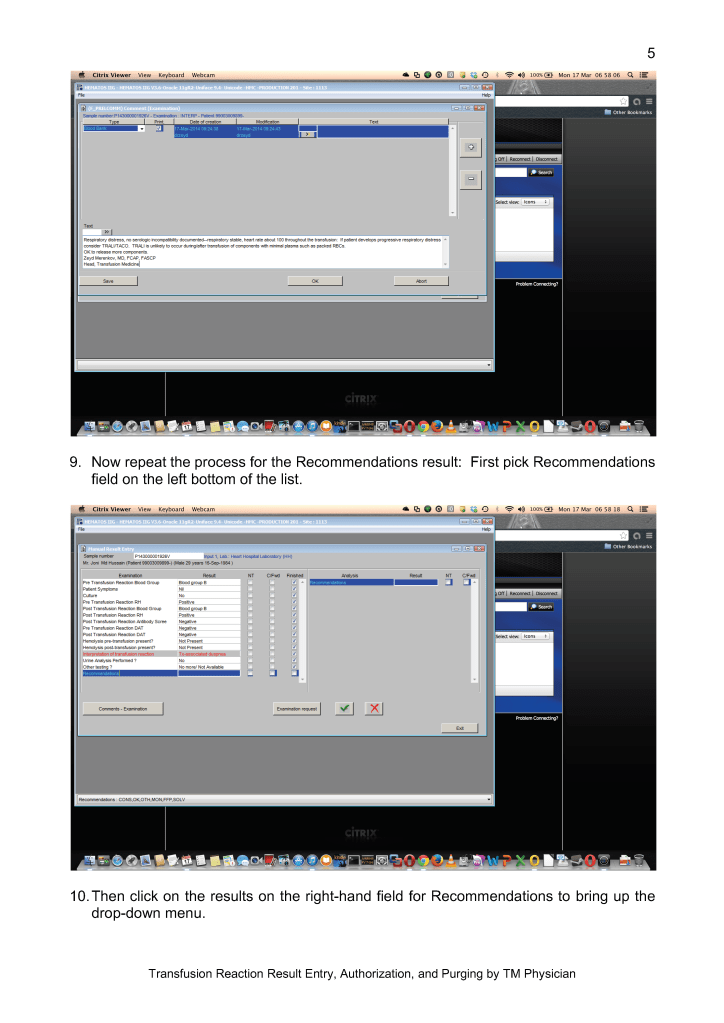
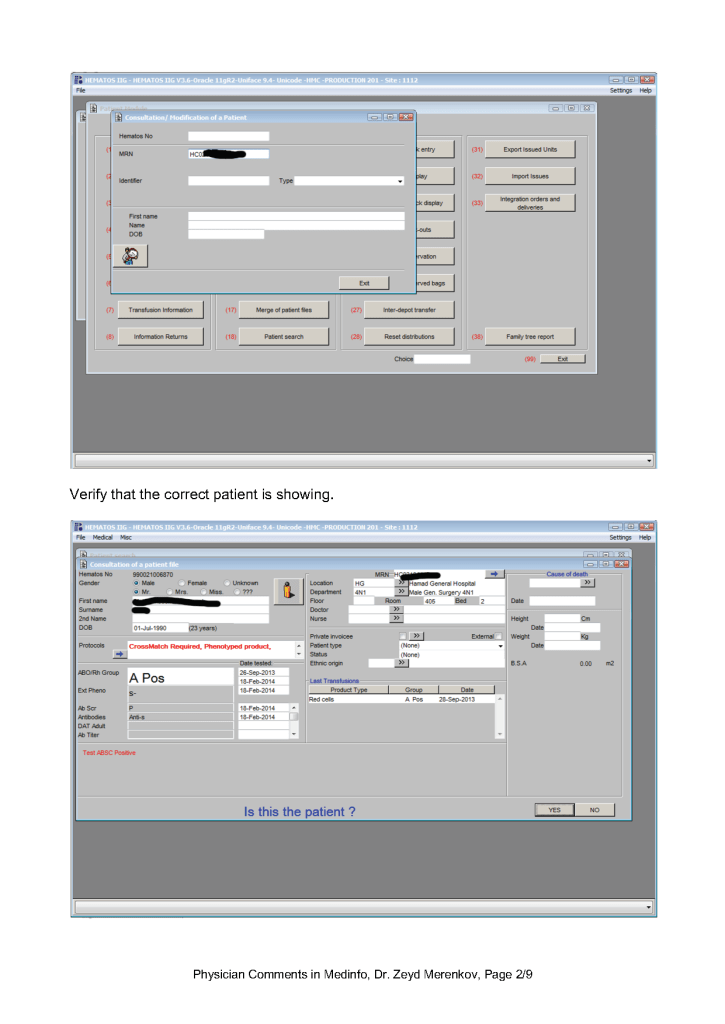
Sign into HIIG and select Patient module, Patient Consultation, and enter the patient hospital HC number.
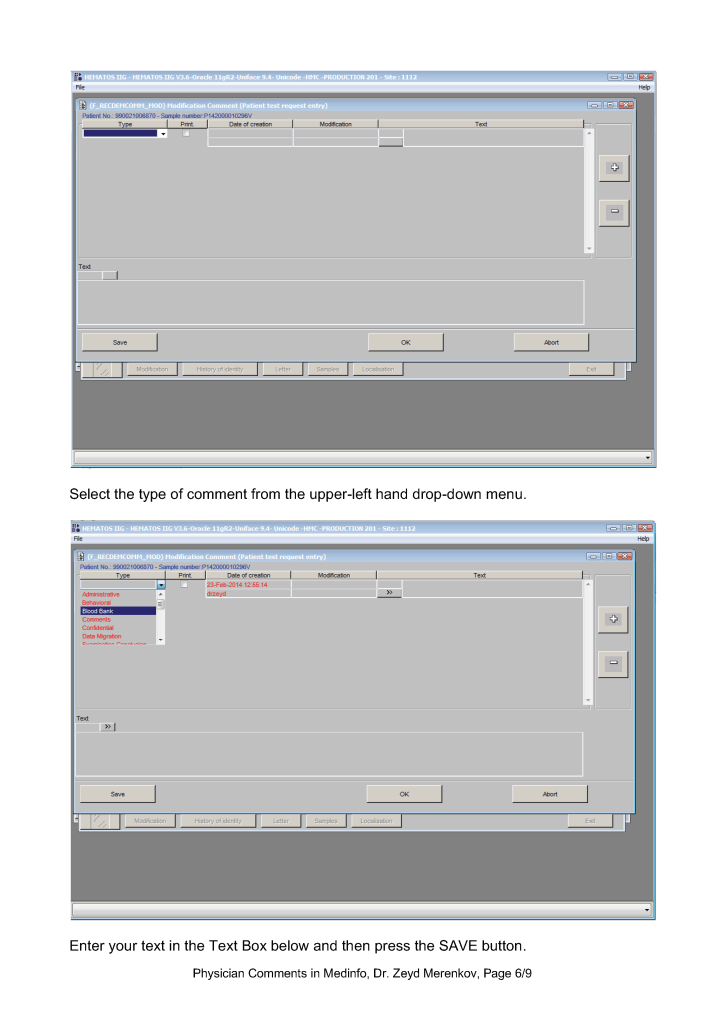

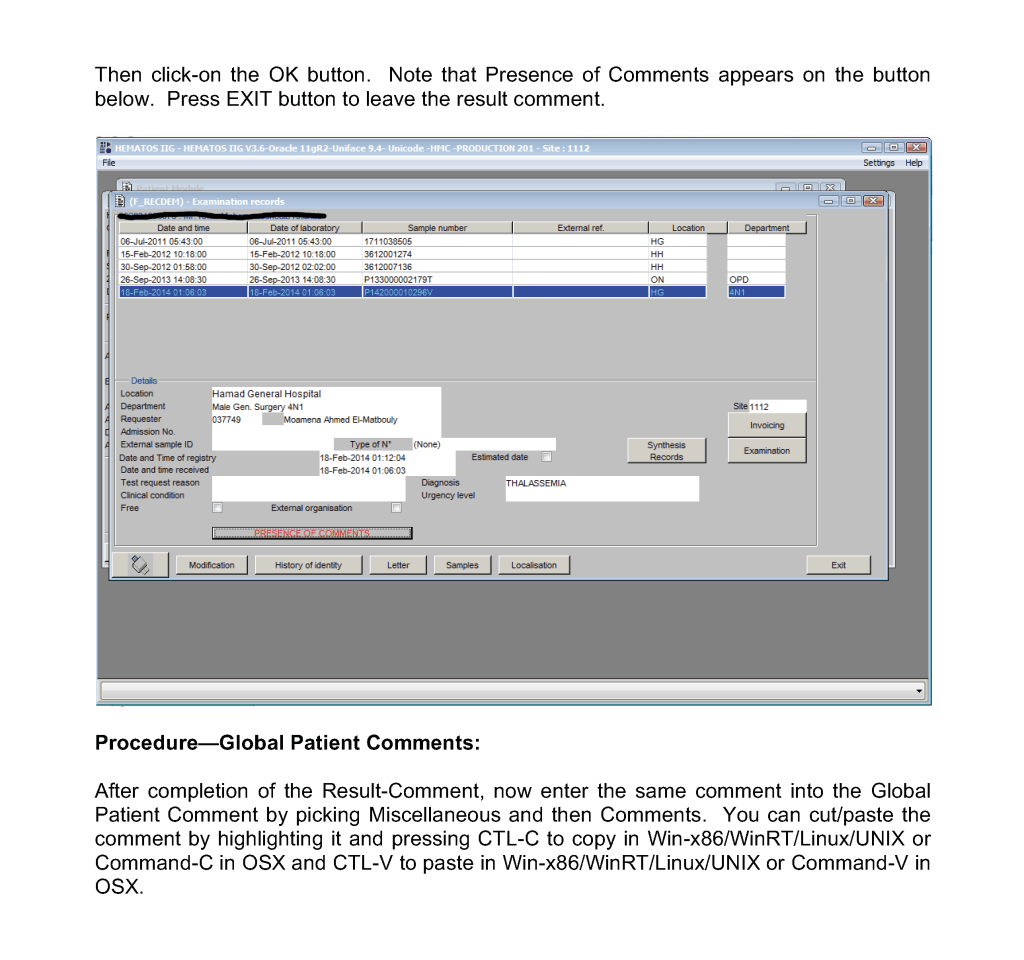
This post is the policy for using comments in Medinfo software. A subsequent post will show the process of entering comments.
Principle:
There are several different types of comments in HIIG:
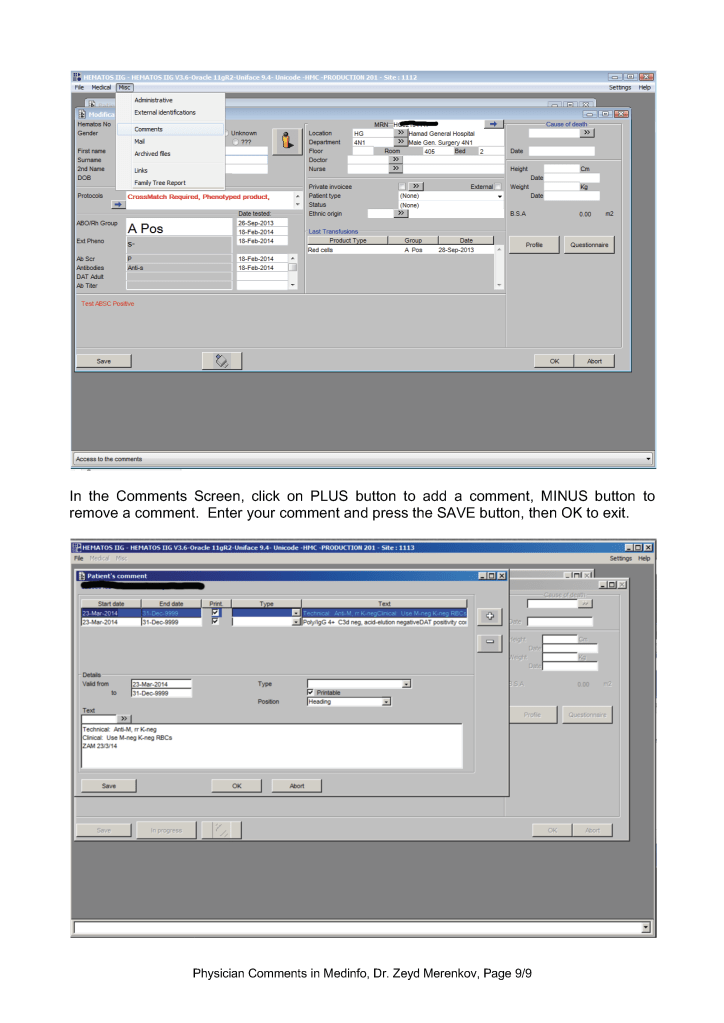
Global Comments appear on the first main screen of either the donor or patient record. The presence of comments is indicated by a bar at the bottom of the screen (in yellow or blue saying Presence of Comments. Double-clicking opens the list of entered comments.
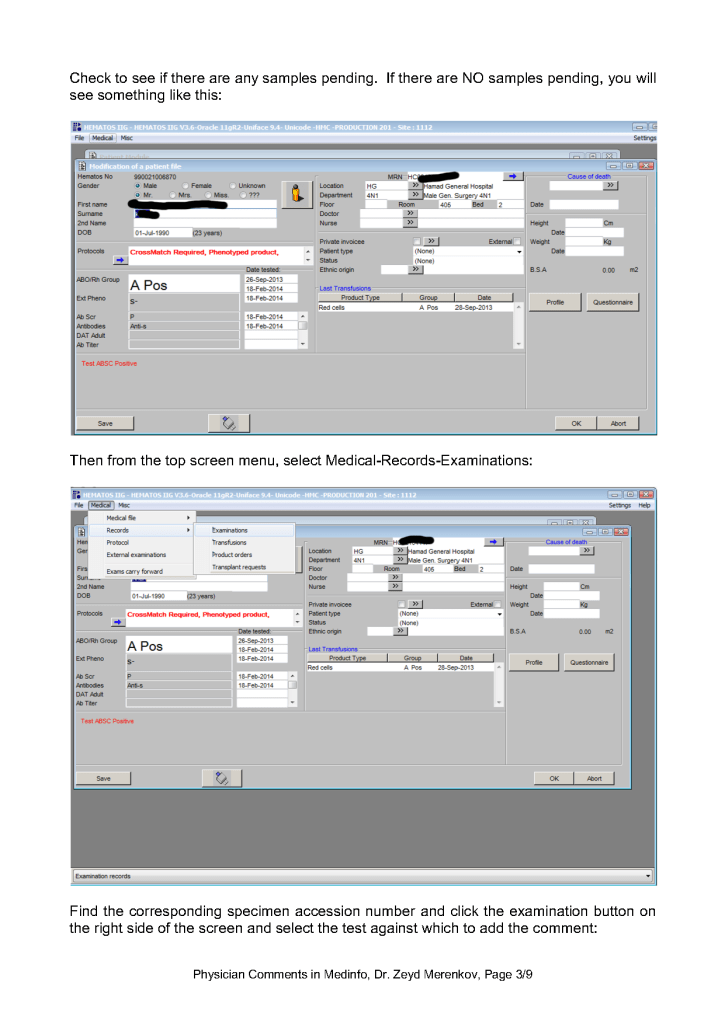
Examination/Results Comments appear only when you open the result to which it is attached. You must know in advance to which result they are linked to find them.
Contraindication Comments appear when entering a donor deferral code
At HMC, we will enter examination/results comments again as global comments (donor or patient) so it is easy for staff to retrieve them and see them with all other comments. You can do this by cut and paste.
Physicians may enter any of these comment types. Comments may be entered before or after a test is authorized/verified. If entered after authorization, the test must be modified to accept the comment and require a special password (not the user sign-into HIIG). Only results/examination comments are visible in the patient’s medical record. Global, analysis, and contraindication comments are visible only in HIIG! Donor comments are only visible in HIIG.
The presence of comments documents physician review of abnormal results as required by the various accreditation standards.
Policy:
References: