Principle:
In accordance with the College of American Pathologists’ accreditation standards, all report structures (content, formatting) are reviewed at least biannually and upon modification by staff designated by the Chairperson, DLMP, here the Division Head, Transfusion Medicine/Laboratory Information System.
Policy:
- Responsibilities:
- The Chairperson, DLMP is ultimately responsible for the report formats, electronic and paper (if applicable) across all areas/divisions/sections of the department.
- The Chairperson, DLMP delegates the responsibility for this review to the Head, Laboratory Information Systems LIS.
- The Head, LIS reviews/approves the final reporting formats after review/acceptance of the formats by the Division and Section Heads for their respective areas.
- Content of the reports:
- Headers and footers as required, especially for paper reports
- Headers will include full patient name and a unique alphanumerical identifier, age, location, sex, date of testing/reporting, location, and ordering physician.
- Footers will include contact information for the site performing the testing and for the Chairperson, DLMP.
- Body of the report will include:
- Test results
- Flags
- Reference range
- Order and result comments
- Corrected/amended/appended results will be clearly marked, including any changes from the initial reports.
- Headers and footers as required, especially for paper reports
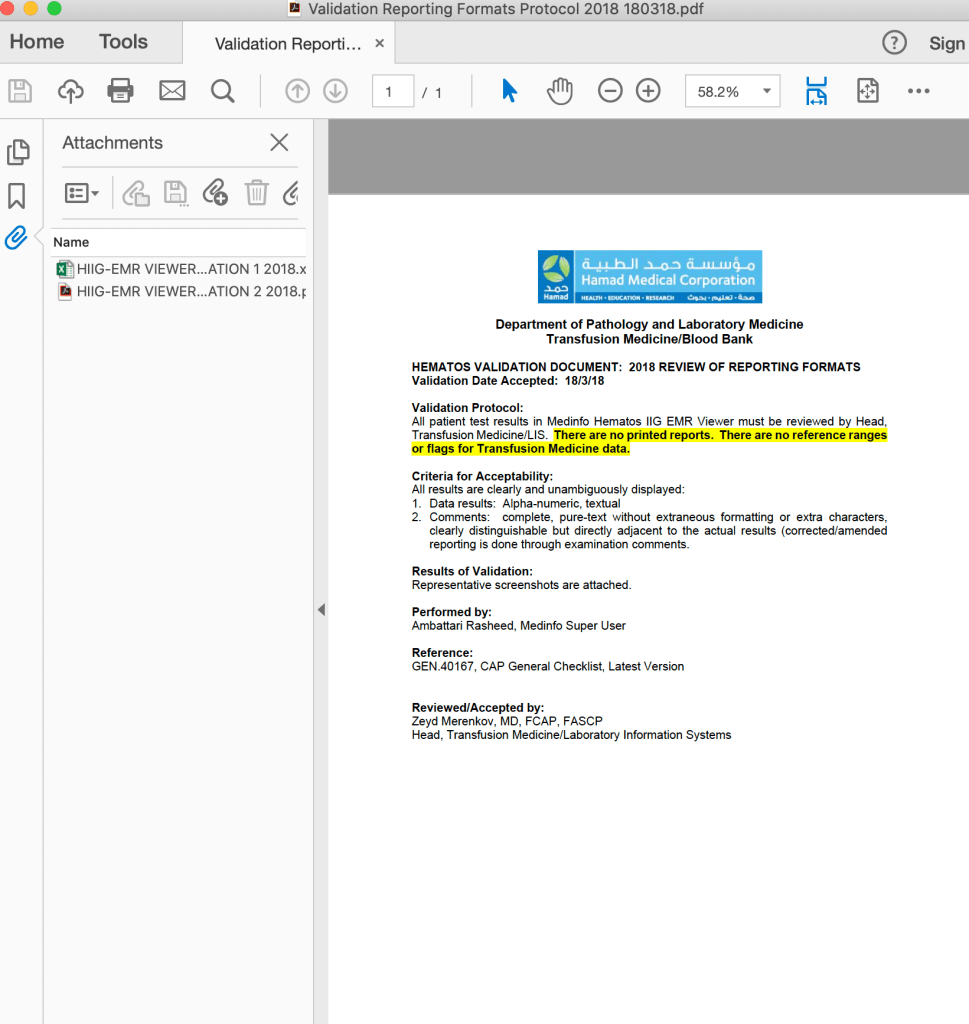
- Documentation of Review:
- Screenshots (electronic) or printout (paper formats) will be collated for one example of each test and reviewed by the Division/Section Heads and documented with a signature/stamp for each test result.
- Upon acceptance by the Division/Section Head, a cover letter summarizing the acceptance will be signed, stamped, and dated.
- The completed documentation will then be submitted to the Head, LIS for final review and approval.
Reference:
GEN.41077, Content/Format Report Review, CAP Checklist, Current Edition
The following is a sample report I prepared during my tenure at HMC Doha:
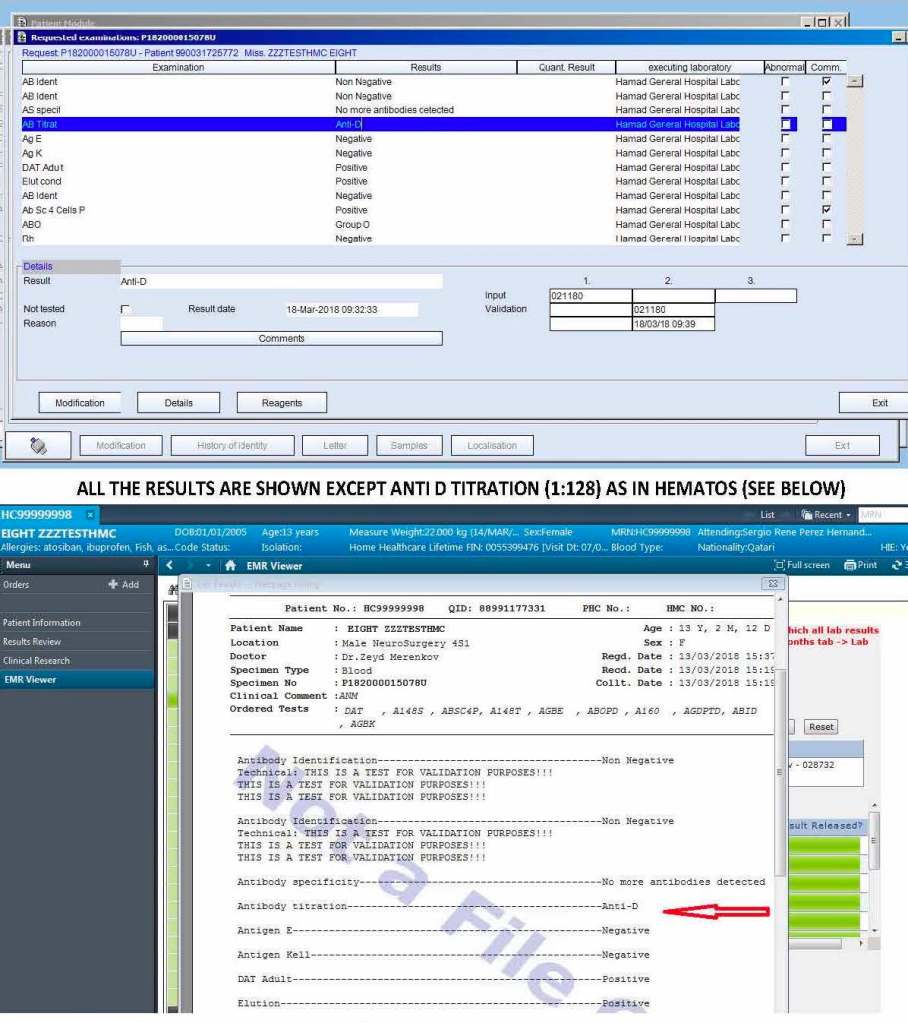
On the left-hand side of this composite PDF there are embedded attachments, which can be accessed by clicking on each one. In this document, I have shown a sample page from the actual screenshots generated. The data showing is a dummy test patient (no real patient data is exposed).
Note that our design of Medinfo did not include printing copies of reports. The only available reports were the screens. Staff outside Transfusion Medicine viewed the blood bank reports through a separate database viewer. In the example below, the Medinfo screen appears first followed by the EMR Viewer report.