Processes and Software Building—Part 20
My practice across the globe has exposed me different rationales to performing antibody titration. In my American training and practice (and also at international institutions following the American version of AABB accreditation), I only routinely performed titration of anti-D for Rh(D) hemolytic disease of the newborn and anti-A/anti-B for ABO-incompatible stem cell transplants AND ABO-incompatible renal transplants.
I have had heated arguments with some physicians who insisted they wanted titers for other antibodies. The AABB Standards do not require this but leave it to the discretion of the Transfusion Service Medical Director.
In my entire career, I never worked in a blood bank or blood center which had optimal staffing or resources. I focused on what was medically/technically necessary and even then still had shortages. If performing a test will not change the clinical treatment, why perform it unless you are doing a research project!
Titration is a time-consuming, and until recently, a tedious manual task. Recently some of the automated immunohematology analyzers offer a titration program. We used the Ortho Vision Max which could perform both IgG and IgM titers within one hour—walk away!! However, during that time, the titration procedure monopolized the analyzer.
The ABO-incompatible renal transplant program at HMC Qatar was modelled after Sweden’s Karolinska Institute. However the latter site performed manual IgG and IgM titrations using Biorad/Diamed gels.
I did not have sufficient resources to commit staff to manual titration at HMC so I did a comparison study between the Ortho Max and the Biorad methods. We were able to get good correlation and used the automated method for the transplant.
I am still biased against performing titrations for other antibodies. I always ask, ‘Does the titration correlate with clinical severity?’ Unlike anti-D, antibodies such as anti-Kell and anti-c may be low titer but cause death. Can anyone show me a definitive study that titers are useful except for transplants and Rh(D) hemolytic disease of the fetus/newborn?
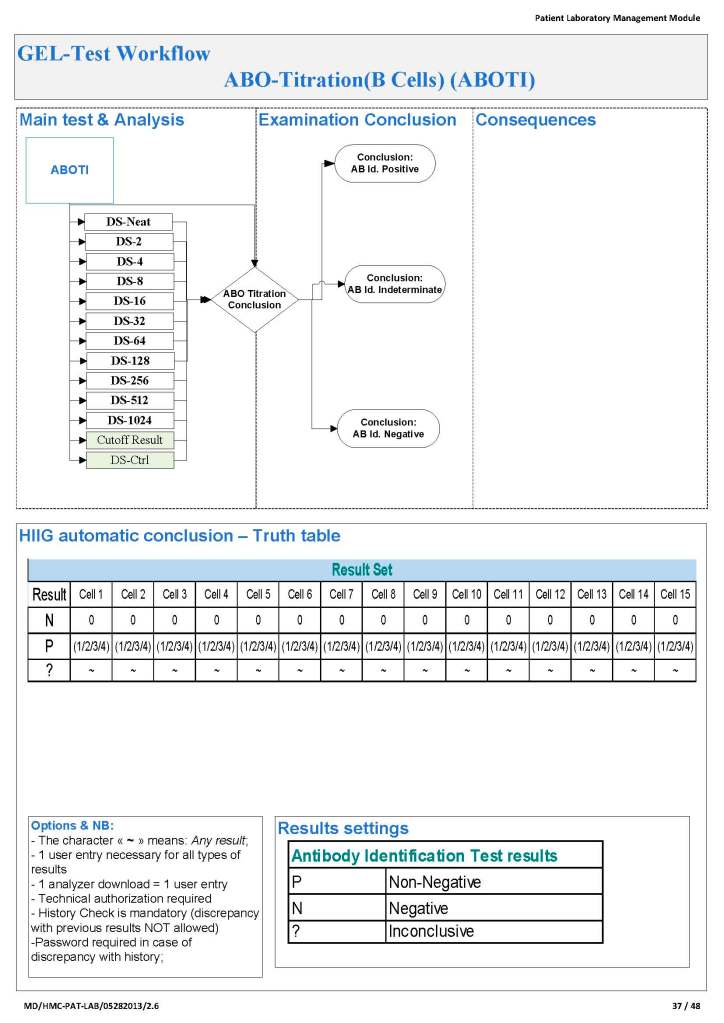
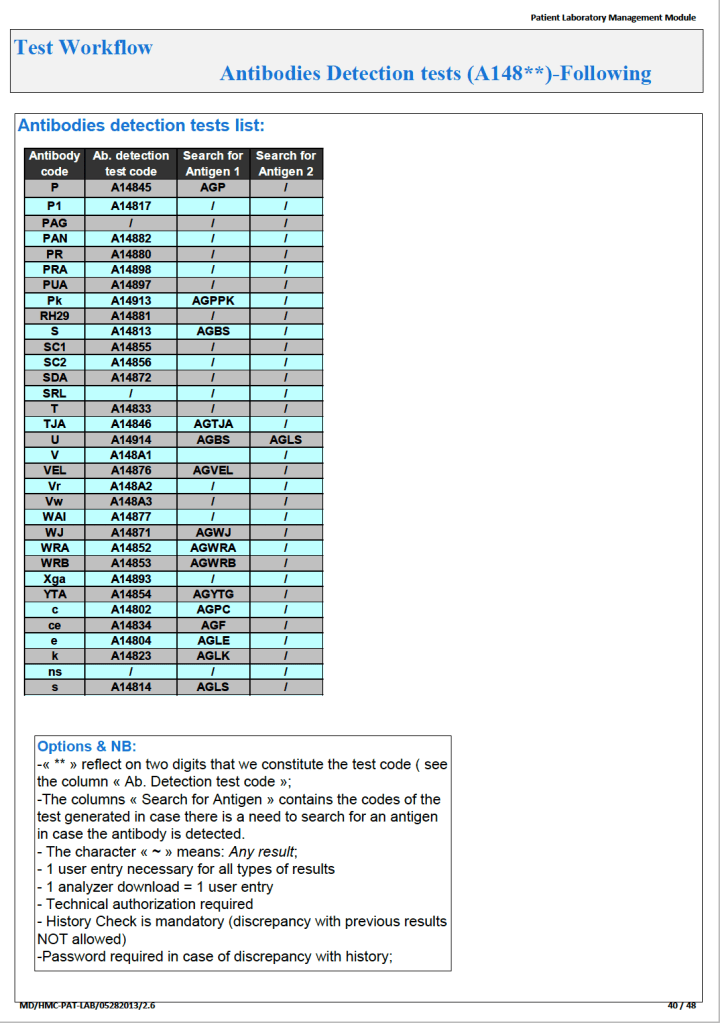
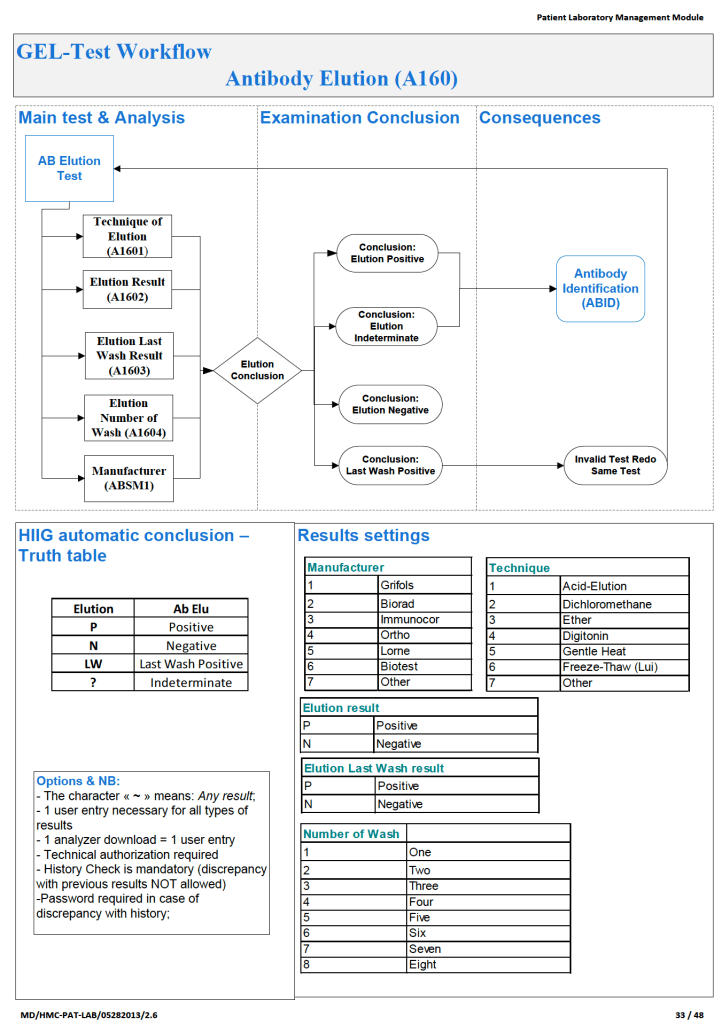
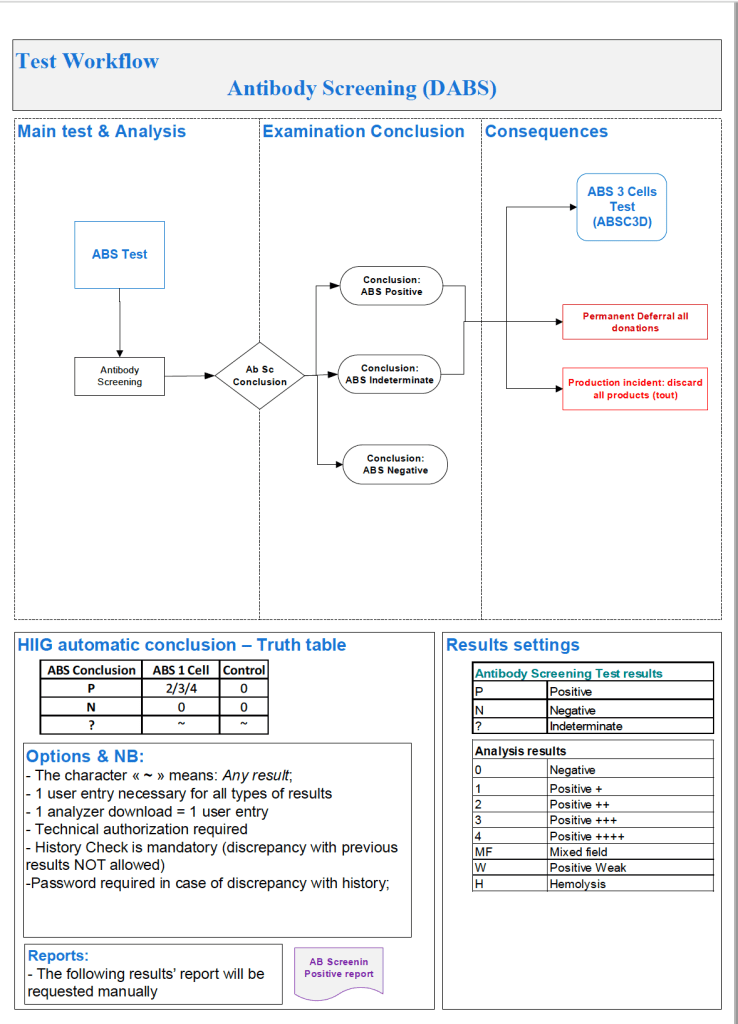
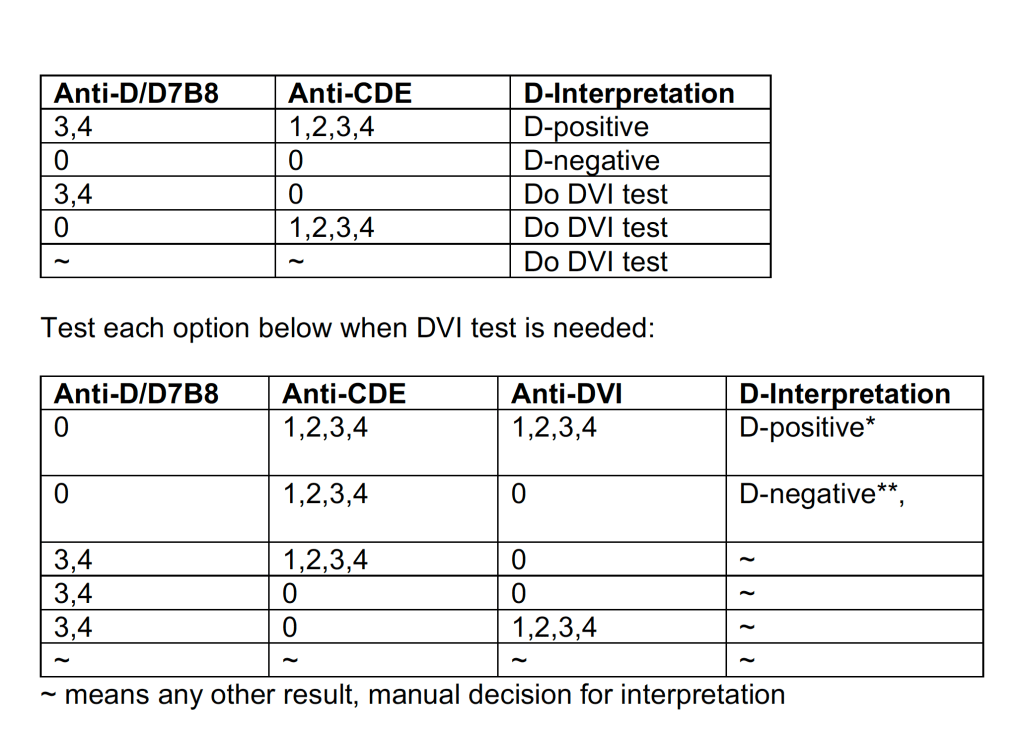
Since the method was working well on the Ortho equipment, I next established an interface to Medinfo. The test was performed separately for IgG and IgM antibodies. Medinfo recorded the reactions in all the wells. The last well showing a 1+ reaction was interpreted as the titer (e.g. if 1:64 were the last 1+ reaction, then the titer was 64 in Medinfo).
The Medinfo process is shown below.
20/7/20