This is an updated version of a previous post.




Immunohematology testing and processes
This is an updated version of a previous post.



2011
Established automated component production using Atreus technology, plasma and platelet pathogen inactivation (Mirasol)—made HMC component production Good Manufacturing System GMP compliant
2011
Qatar is the first to adopt non-PCR-based NAT technology (Grifols/Novartis Tigress) and becomes world reference site for this
2011
Based on the above, Qatar can now completely process all whole blood into blood components (red cells, platelets, and plasma) in as little as 5 hours from collection!
2011-2020:
I established policies and procedures for the hospital blood banks/transfusion services, blood donor center, therapeutic apheresis, and laboratory information systems to bring HMC in compliance with the Council of Europe, international AABB, and other standards. I customized our own standards for our local needs based on them.
2012-2013
Implemented custom build of the multilingual blood bank computer system (Medinfo) for both patient and donor services, including development of interfaces to all production equipment including Atreus and Mirasol (world’s first) and a direct link to Ministry of the Interior to obtain patient demographics in English and Arabic—Qatar became the world’s first site to combine fully-interfaced, automated component production with pathogen inactivation: Qatar becomes world reference site for this.
2013-2014
Built, validated, and implemented laboratory build of hospital information system, Cerner Millennium
2015
Replaced and updated Atreus with Reveos automated component production to allow faster throughput and capacity with a full bidirectional interface (world’s first), introduced platelet
additive solution PAS with pathogen inactivation (Mirasol)—Medinfo interfaces updated to Reveos for all equipment: this doubles the capacity to process whole blood into components using the same physical space
2015-2019
Updated dedicated blood bank software Medinfo Hematos IIG by several versions using Division Head, LIS, and internally trained Super Users—at great cost savings to HMC by not using outside consultants (e.g. Dell Consulting)
2019
Established column absorption technology using Terumo Optia therapeutic apheresis machine for treatment of ABO-incompatible renal transplants: I validated using the Ortho Vision MAX to perform ABO antibody titers for this system and correlated it with the reference method at Karolinska Institutet in Stockholm (manual gel) to bring rapid throughput and labor savings—Qatar being the first-site in the world to do this. We saved money by using the same apheresis machine to use this column absorption technology (no need for second machine to use the columns)
2020
Expedited setup (two weeks total) of COVID-19 convalescent plasma production, initially manual and then fully integrated into the Medinfo computer system as a customized module with separate quarantine collection, production, and transfusion service functions
Other:
I was awarded two HMC Star of Excellence Awards:
2013—Liver Transplantation Transfusion Support
2019—ABO-Incompatible Renal Transplantation Support
This presentation was originally given to third-year medical students at NGHA-Riyadh as their first lecture, Introduction to Transfusion Medicine. It has been updated for new component types and will be presented in multiple parts.








These are the answers to the previous post.



This is the third-year hematology fellow examination I prepared for NGHA Riyadh:

This is a post of an old teaching presentation for pathology residents, hematology fellows, and transfusion medicine fellows from my time at NGHA-Riyadh.



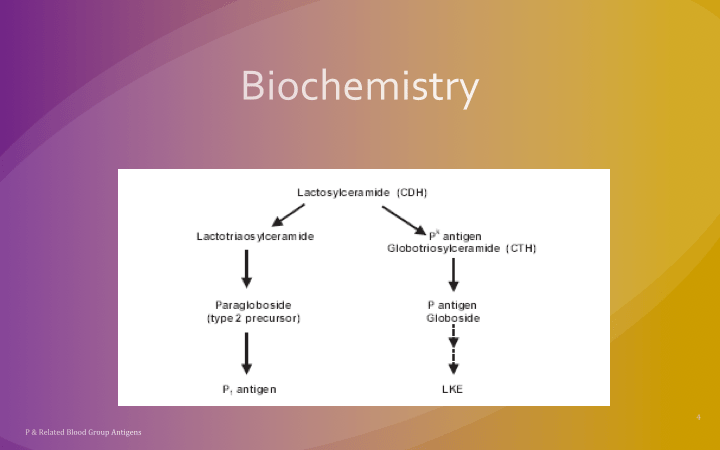
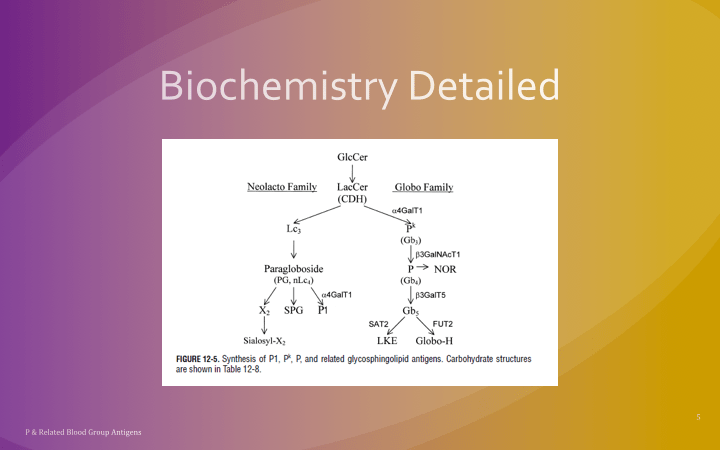



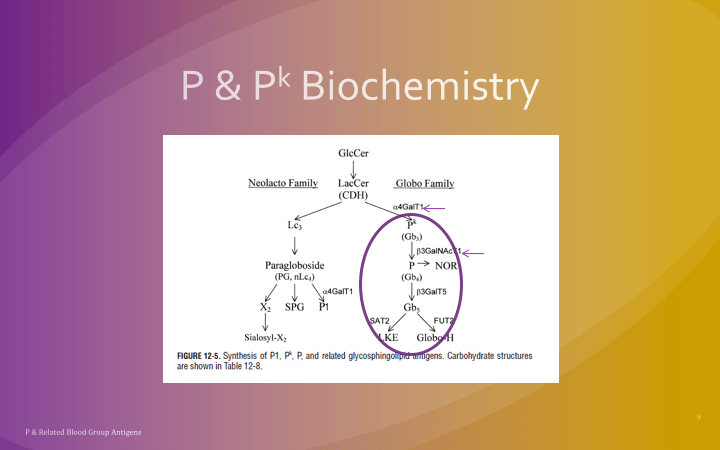

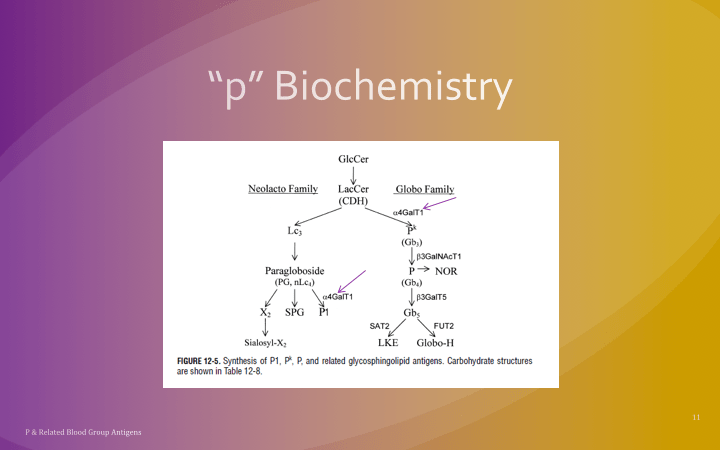

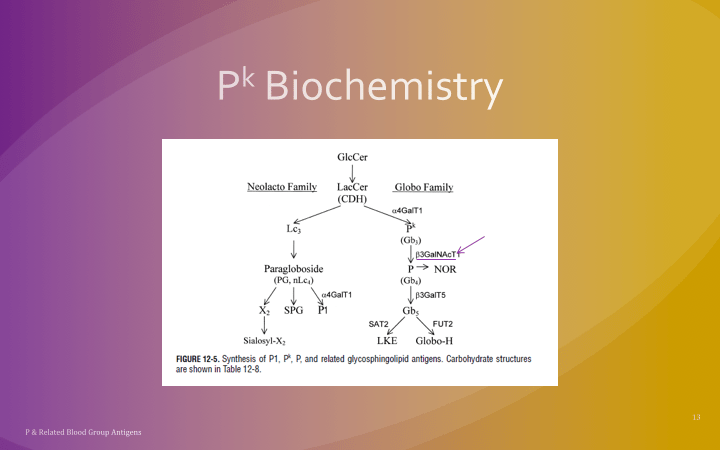

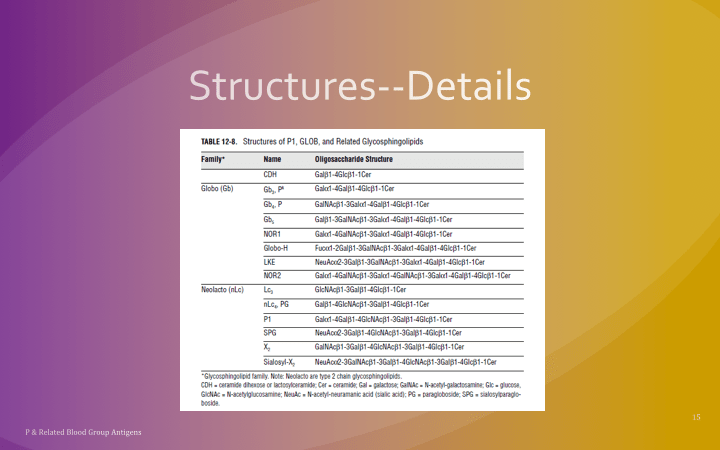
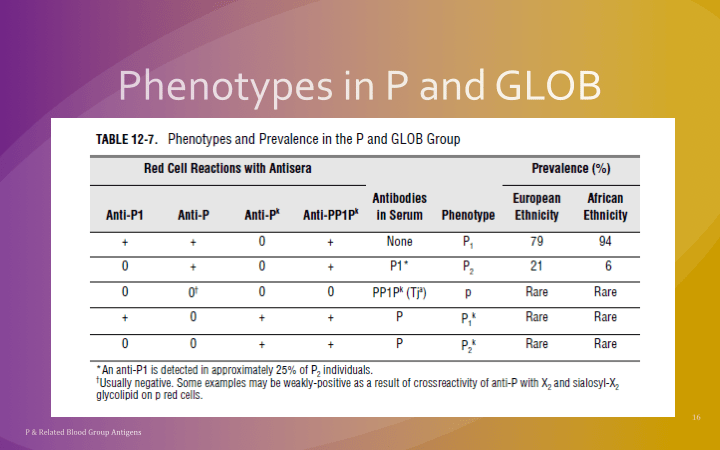
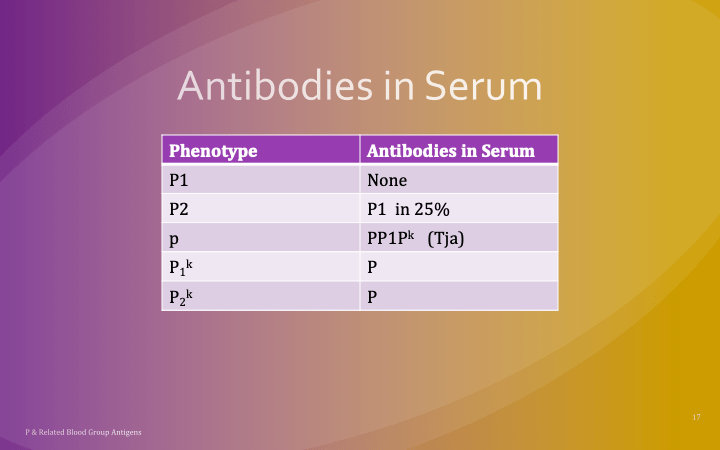











This is a reprint of a previous post.



This is an updated version of a previous post.

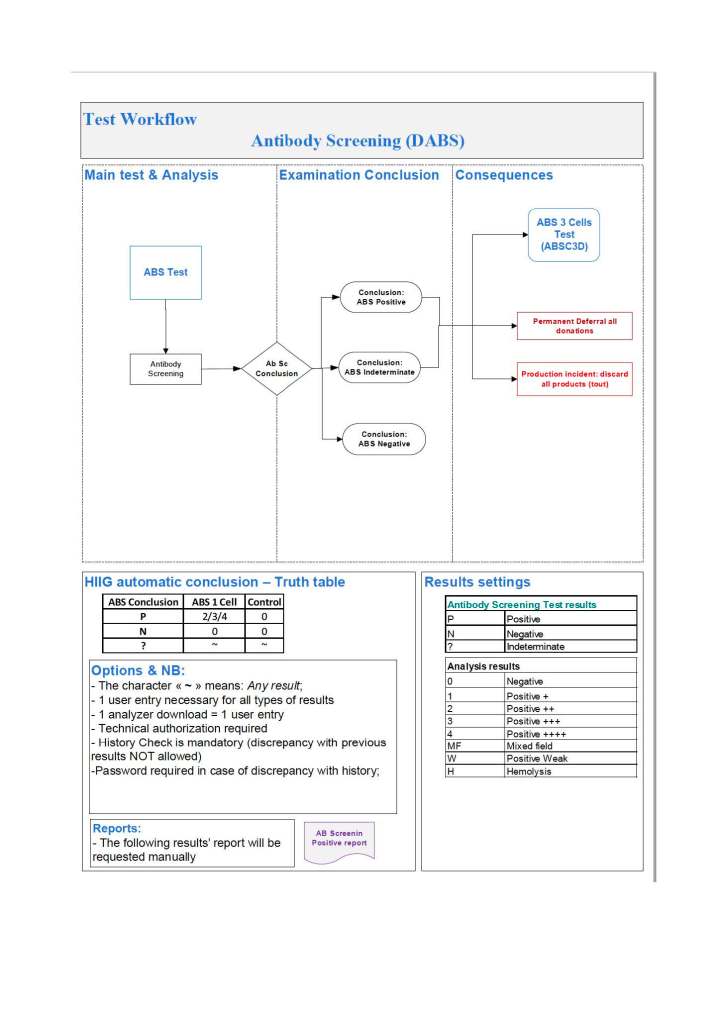
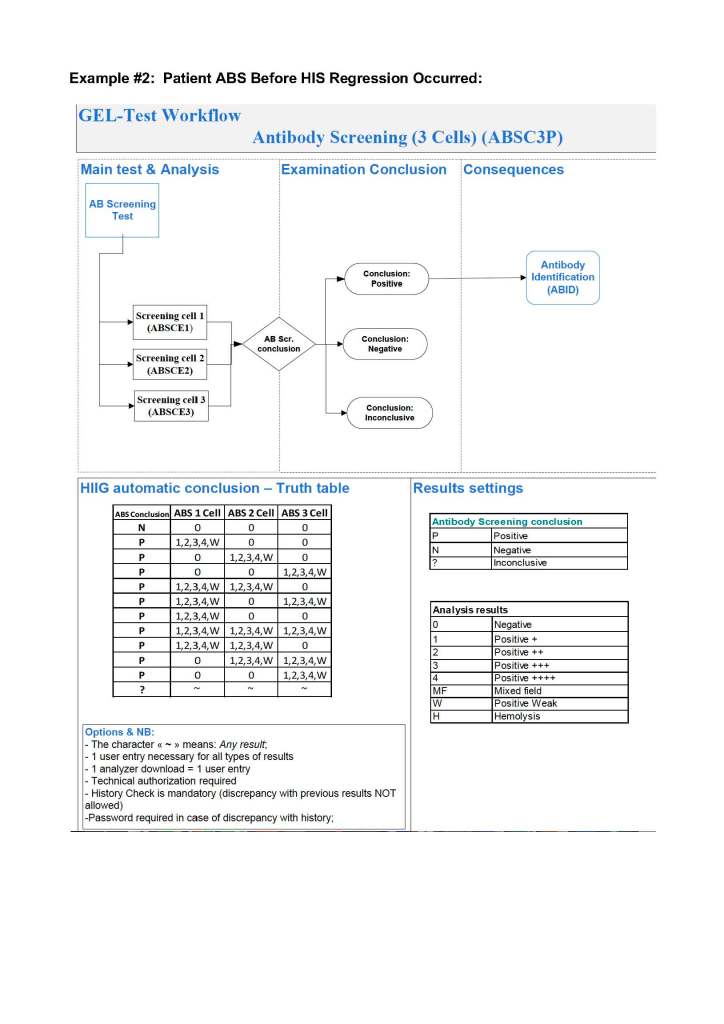
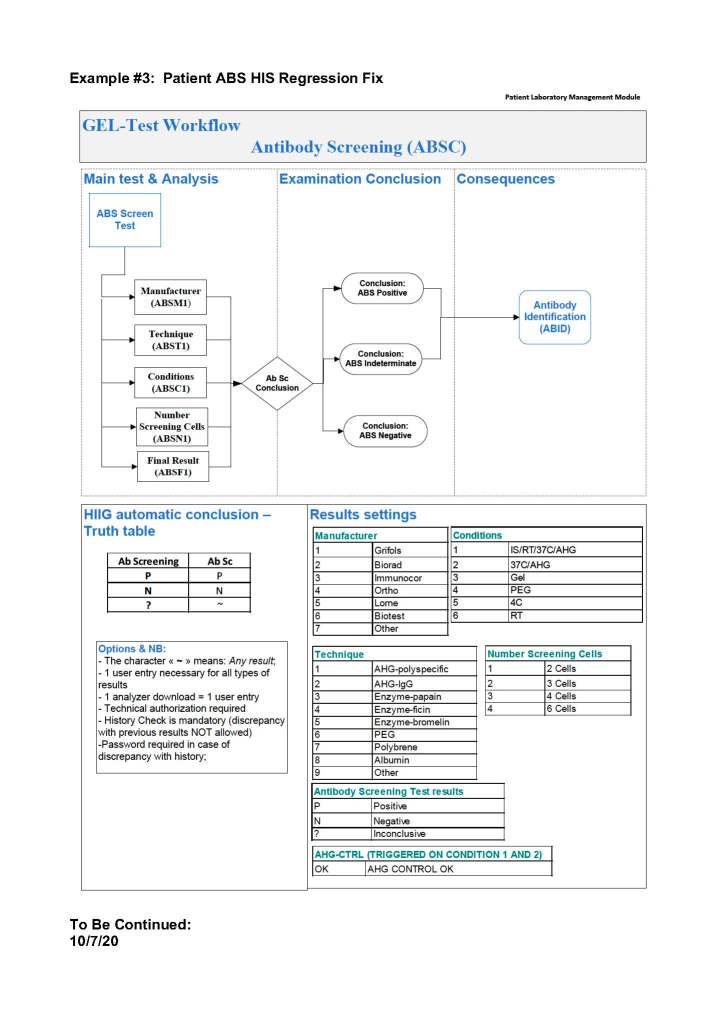
During my career, the role of the transfusion medicine physician in handling RBC antibodies has evolved. Depending on your location, either a specific transfusion medicine physician or a hematologist covering the specialty handled this. In the United States, most hematologists nowadays do not do this.
During my US residency training in pathology, most trainees did not have an interest in blood banking and often used the rotation to take vacations. For their board certification in clinical pathology, they crammed for the examination and afterwards did not engage in it.
When I started my career in the United States, hospitals had a hospital blood bank/transfusion service and handled most of their antibody problems themselves. In Chicago where I practiced, many blood banks had Specialist in Blood Banks SBB graduates or SBB students working, and of course, they had a dedicated blood bank supervisor.
Currently in the United States, the hospital blood banks may be staffed by generalists and there is no one with specific antibody experience. SBB graduates are expensive and usually work in blood centers or academic hospitals.
If you have a regional blood center with a reference laboratory, you can send your antibody workups there and let the blood center select appropriately antigen-matched units. The physician covering the blood bank does not have to get very involved.
In the Middle East in the systems that I have run, there have been no reference laboratories and often no regional blood center. The hospital system or the free-standing blood bank is an independent entity and must rely on itself. The physician responsible for the blood bank must review the antibody panels and make his own decision how to select RBC components.
In the Middle East, especially in the larger centers with academic hospitals, there is usually a transfusion medicine physician who reviews the antibody workups and makes the final decision of what RBC types to dispense. He does not have a reference laboratory to rely on. He is responsible for the choices and has no one to refer cases on a 24/7 basis.
In the Middle East, the transfusion medicine physician must be proactive. He must also select antigen-matched, fully or partially, and understand the trade-offs in cases with multiple antibodies. He must know how to deal with nonspecific antibodies and antibodies to high-incidence antigens, especially if he works in a region where the antibody panels are not optimized to the local population or there are many different ethnic groups in the local area.
Thus, training someone to practice in the Middle East requires spending considerable time in technical matters that might not be necessary if he/she practiced in the West. He may not only have to serve as a physician but he may be the equivalent of the reference laboratory supervisor. Until such time that reference laboratories are available, this model is essential for safe practice. Training programs for the region must reflect this and in particular, teach about specific antibodies common to the region. He must be technically oriented and he must take ultimate responsibility for the interpretation of the antibody workups and selection of the appropriately matched units.
This is a revised version of a previous post.
As much as 90% of the RBC component allocation can be performed without an actual crossmatch test (AHG or immediate-spin) provided that certain criteria are met.
Enforcing these rules, however, can be cumbersome unless one has blood bank software that verifies that each rule is met. In the Medinfo patient module, the transfusion history database is checked automatically. If the rules are met, then Medinfo allows the selection (allocation) of RBC units without performing a crossmatch test. Otherwise, it will check to see if the AHG crossmatch has been done within the past 3 days. If not, it will prompt for new crossmatch testing with a new specimen. If the situation is urgent, one can go to Emergency Mode and release components without the crossmatch.
Principle:
In selected patient categories, no classical crossmatch may be required for release of RBC components. The criteria are specified here as applicable in my build of the Medinfo Hematos IIG computer system HIIG.
Policy:
References: