This Powerpoint file summarizes the past previous posts about the use of automated components, pathogen inactivation, and their synergism with the blood bank computer software Medinfo Hematos IIG












































Automated and manual component processing including Reveos, Mirasol, PAS
This Powerpoint file summarizes the past previous posts about the use of automated components, pathogen inactivation, and their synergism with the blood bank computer software Medinfo Hematos IIG

















































Enforcing Good Manufacturing Process Through A Dedicated Blood Bank Software
Blood components are a drug and like medications must be consistently produced and follow Good Manufacturing Practices GMP. The following system that I set up for HMC Doha Qatar blood collection and processing is an example of the impact of Medinfo donor software on enhancing our safety and GMP compliance. In earlier posts, I provided the Medinfo flowcharts for these processes.
This is an outline of the processes I built in conjunction with the Medinfo software engineers:
I emphasize that only if all criteria across all areas pass is the final ISBT label printed. Medinfo is not a label printing program. It enforces the rules ruthlessly. My technical staff tell me that it is merciless—as it should be for patient safety.
Attachments: None—please refer to earlier posts regarding collection, processing, donor testing, and inter-depot transfer.
28/6/20
Automated Component Processing: Reveos and Mirasol Pathogen-Inactivation
The production instruments have more complicated interfaces than the testing equipment discussed in the previous post:
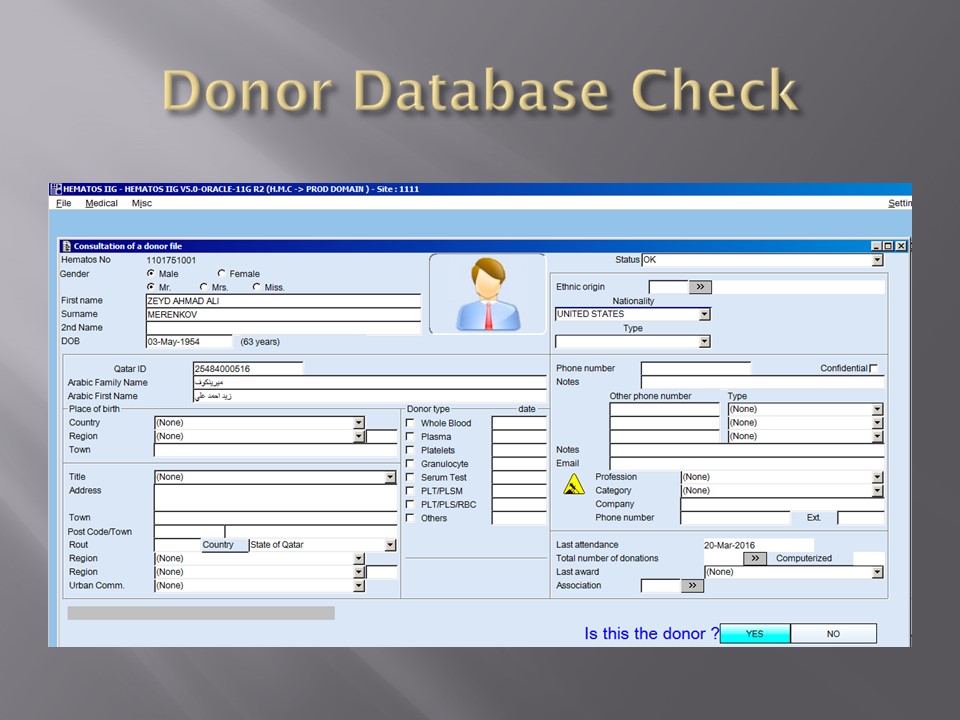
In the collection area (on-site or remote), the cvolume of the whole blood and collection time are recorded in Medinfo and based on the rules, production may only occur within specified volume and collection time. Otherwise, Medinfo will block further processing.
The ISBT unit number of the whole blood units are read by the Reveos. Only those units passing the collection criteria will proceed to separation.
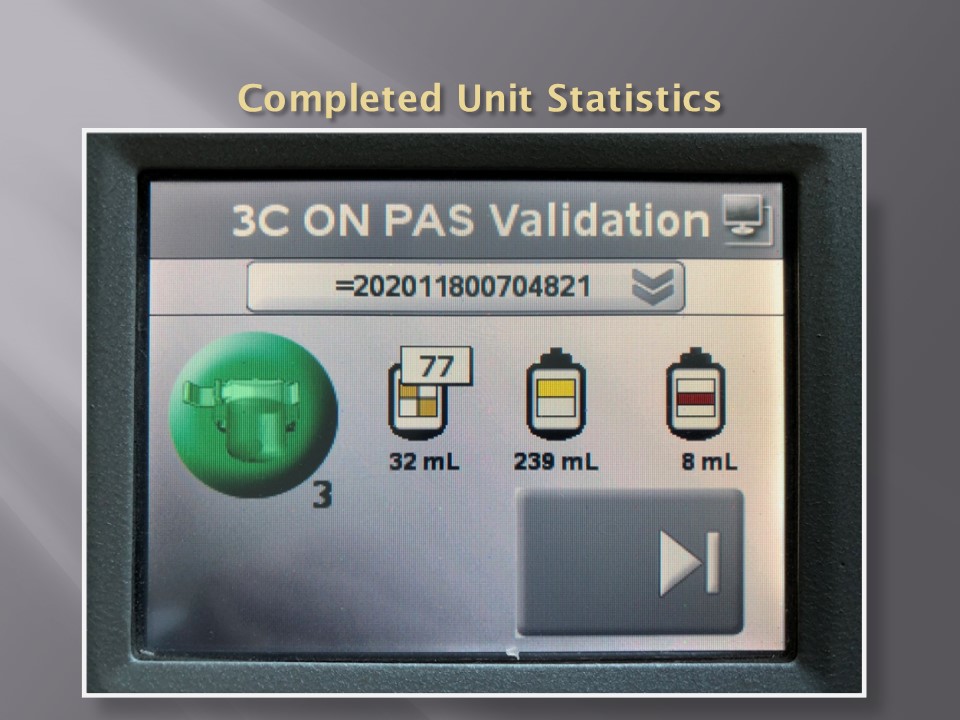
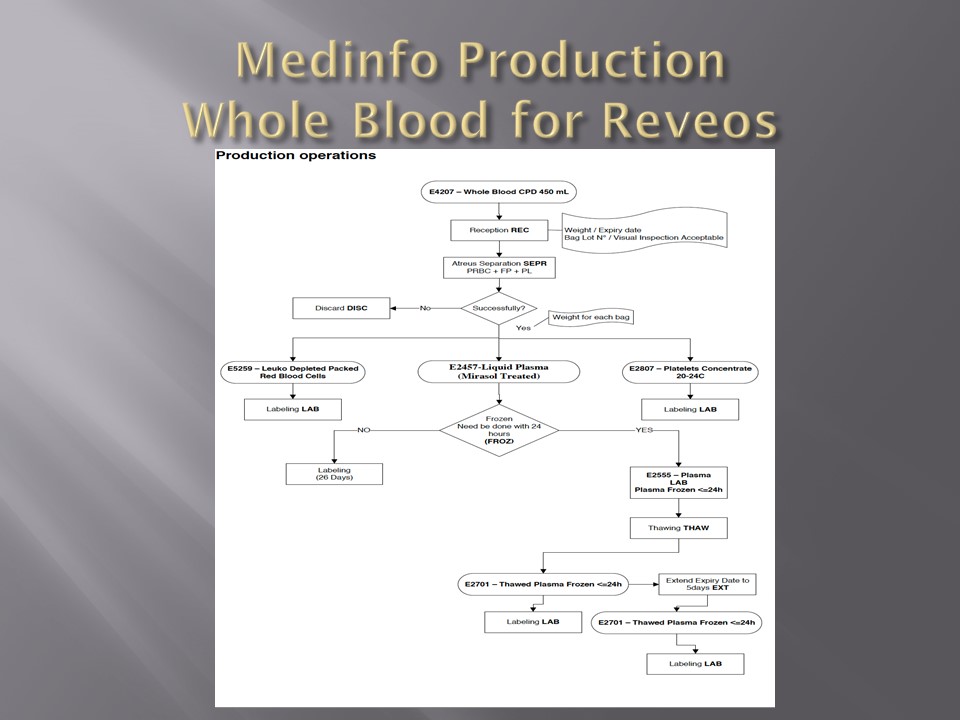
In about 20 minutes, the Reveos machine will simultaneously process four units of whole blood into packed RBCs, leukodepleted plasma, buffy coat platelets, and residual buffy coat. The volumes of the RBCs, plasma, and platelets are recorded in Reveos. For the platelets, the platelet yield index is also provided.

Within Medinfo, these parameters are compared to criteria of acceptability according to the manufacturer. Volumes for the platelets and plasma must be within certain ranges to permit pooling and pathogen inactivation and additive solution. Medinfo will not permit these subsequent procedures if the values are out of range and the intermediate components will be discarded.
Here is a sample of Reveos acceptable ranges for component volumes:
E4207 – Whole Blood CPD 450 mL
Volume Consequences
< 400 mL Discard
400 – 500 mL OK
> 500 mL Discard
E5259 – Leukodepleted Packed Red Blood Cells
Volume Consequences
< 230 mL Discard
230 – 330 mL OK
> 330 mL Discard
E2807 – Platelets Concentrate 20-24°C
Volume Consequences
< 20 mL Discard
20 – 55 mL OK
> 55 mL Manual decision
E2555 – FP24: Plasma Frozen <= 24h
Volume Consequences
< 170 mL Discard
170 – 360 mL OK
> 360 mL Manual decision
All these production parameters are permanently stored in Medinfo as part of the production record of that unit. The actual location (bucket) of the whole blood unit in the Reveos is also available.
RBCs are manually leukodepleted and the final volumes recorded in Medinfo based on weight. Based on the platelet yield index, platelets are pooled and the final volume recorded. Those permissible volumes are next treated with platelet additive solution PAS and then pathogen inactivated. The acceptable volumes are based on the process used, e.g. platelets in plasma versus platelets in PAS.
How a sophisticated blood bank software like Medinfo enforces good manufacturing process at all stage of production will be a future topic.
To Be Continued:
26/6/20
Principle:
Due to the pandemic, we will initially MANUALLY collect an experimental, investigational-use-only plasma product from apheresis donors and treat it with Mirasol. THIS IS A EMERGENCY INTERIM PROCESS UNTIL THE MEDINFO HEMATOS IIG PROCESSES ARE PREPARED AND VALIDATED.
Policy:
8/4/20
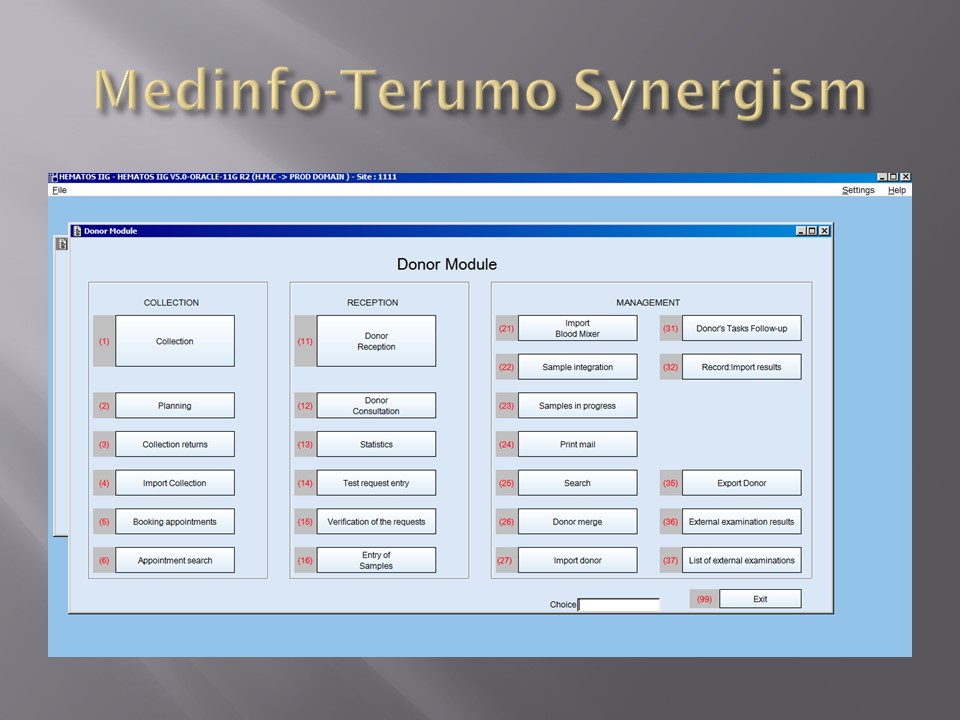
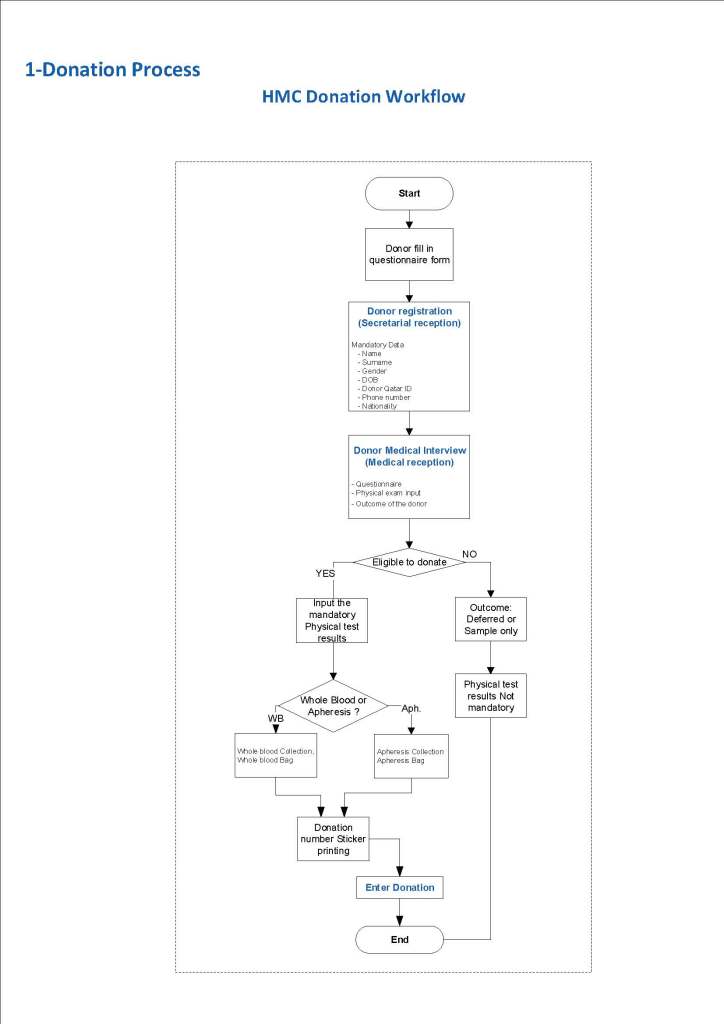
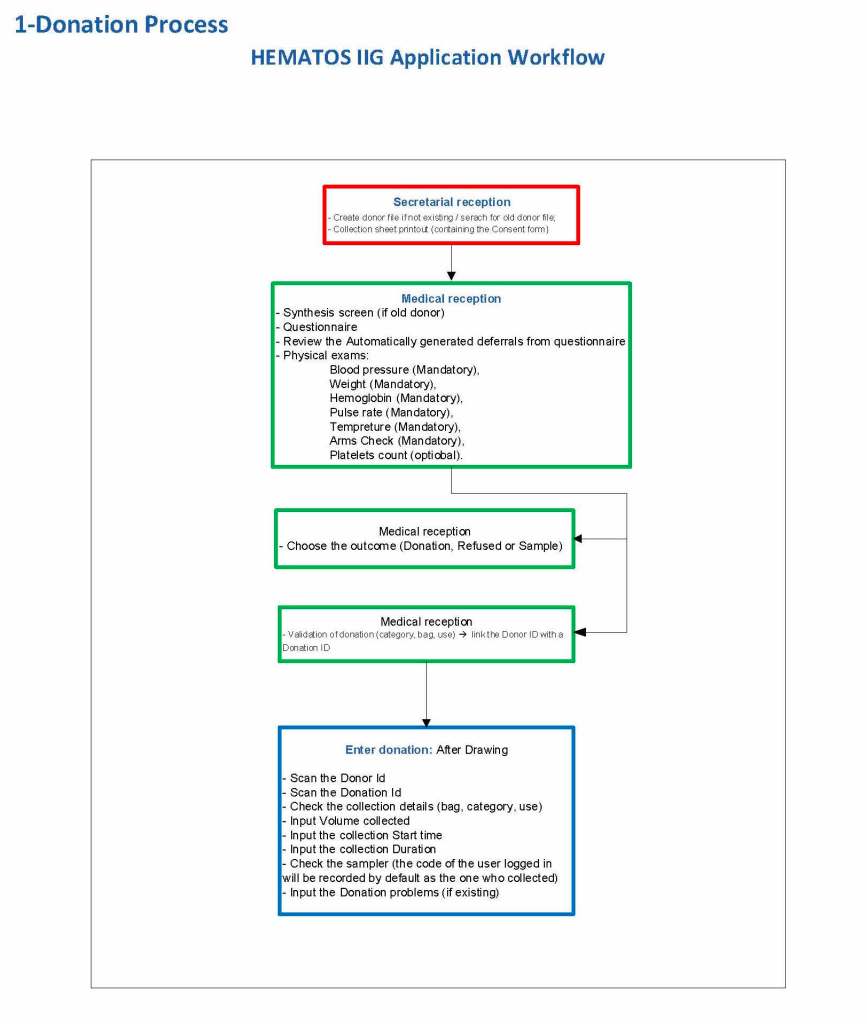
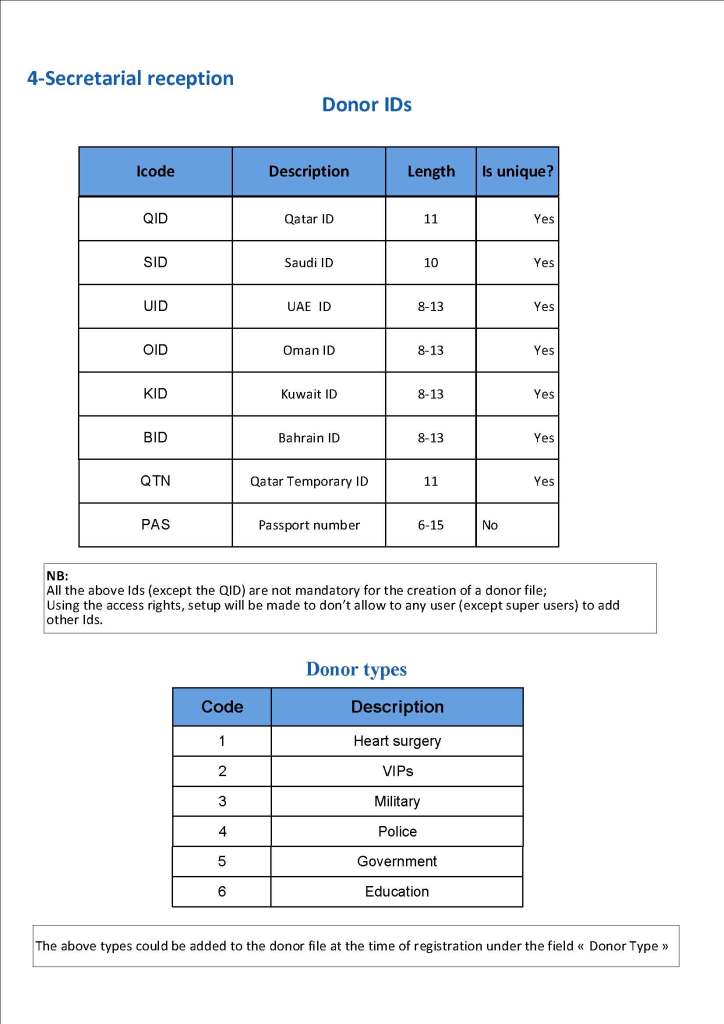
This is a sample Medinfo overview document for the blood collection process for HMC Doha that I designed in conjunction with Medinfo France and Medinfo Doha. This includes, registration, donor consent, questionnaire, physical examination, and collection.



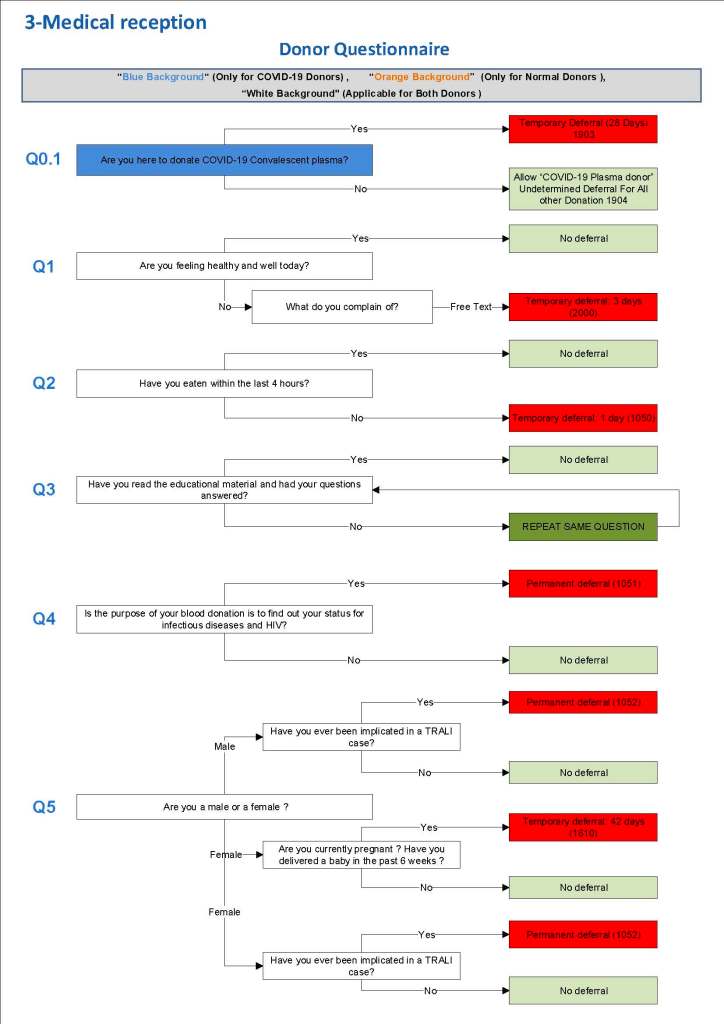


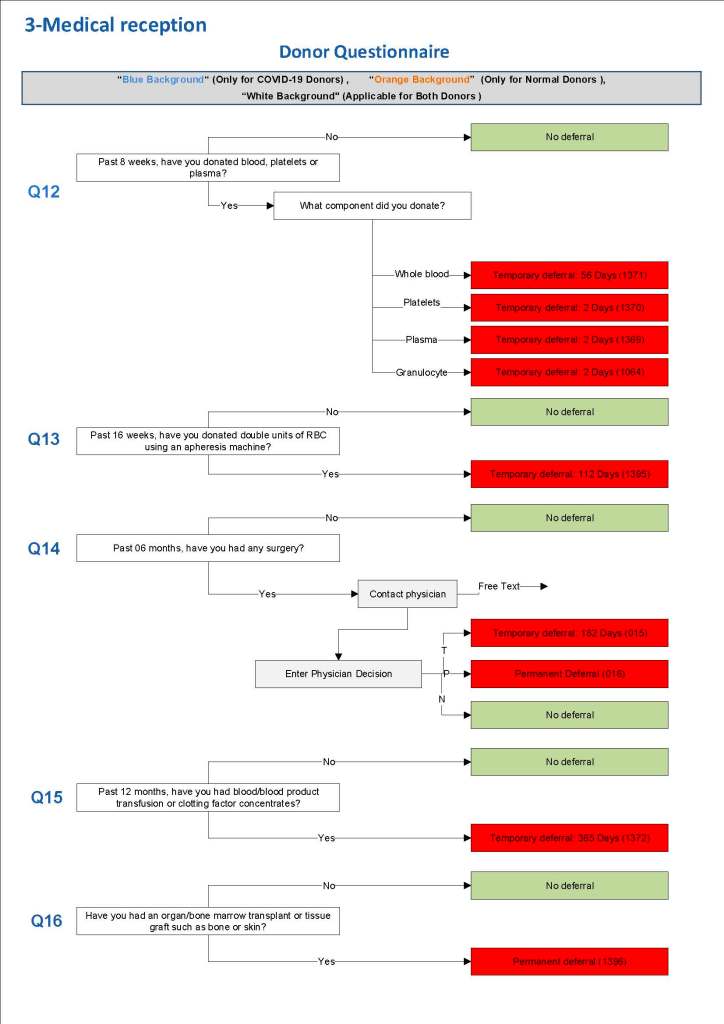
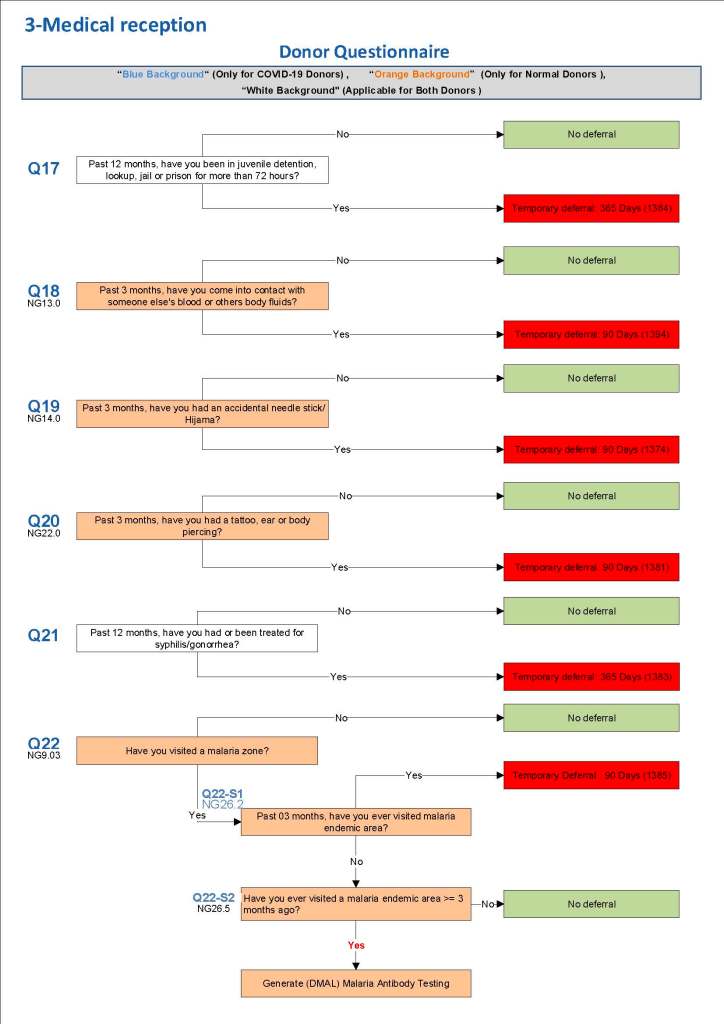
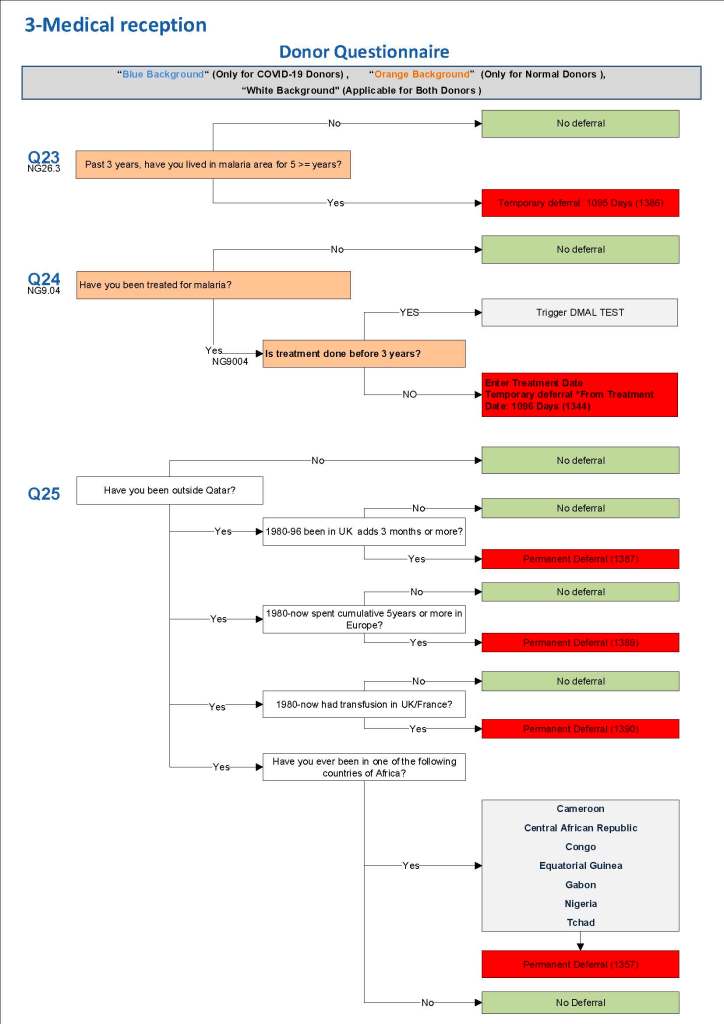

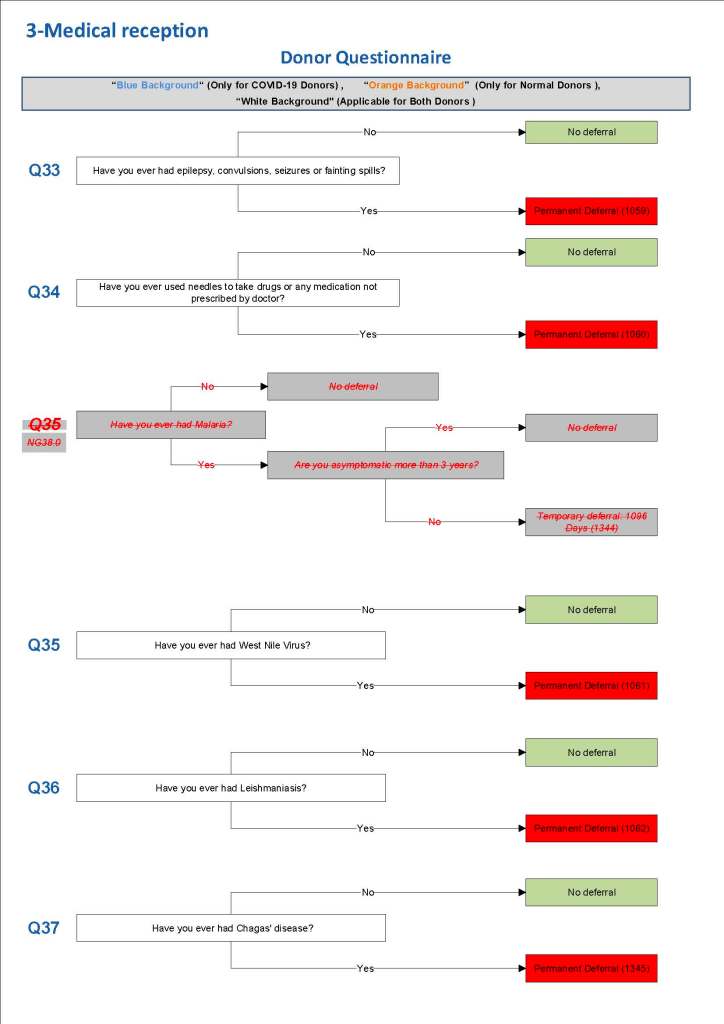
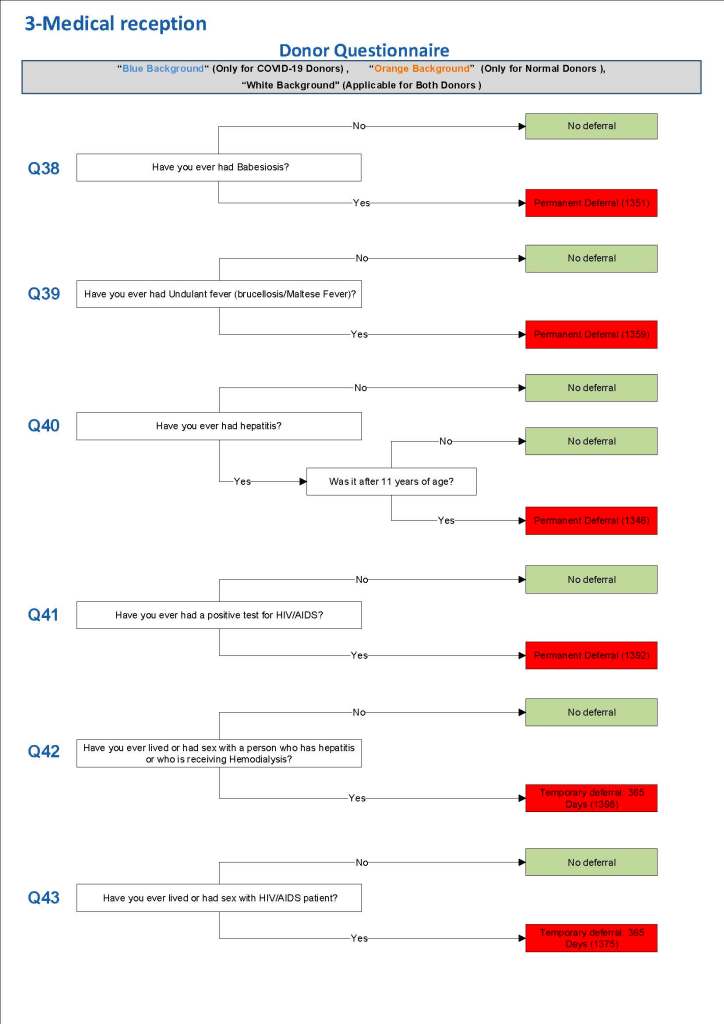

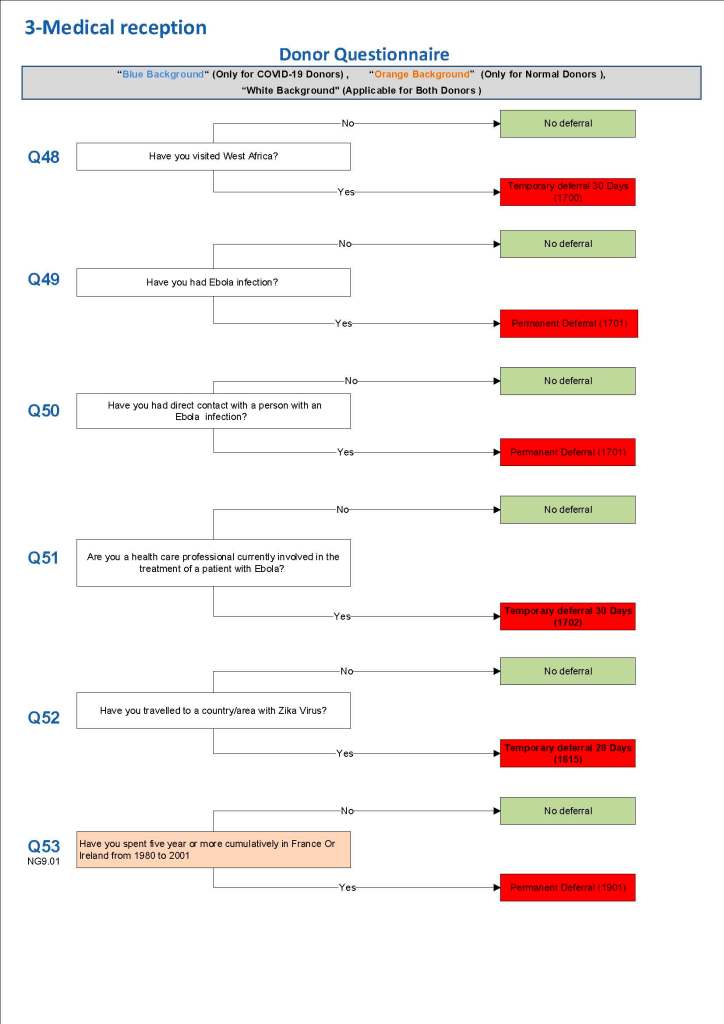
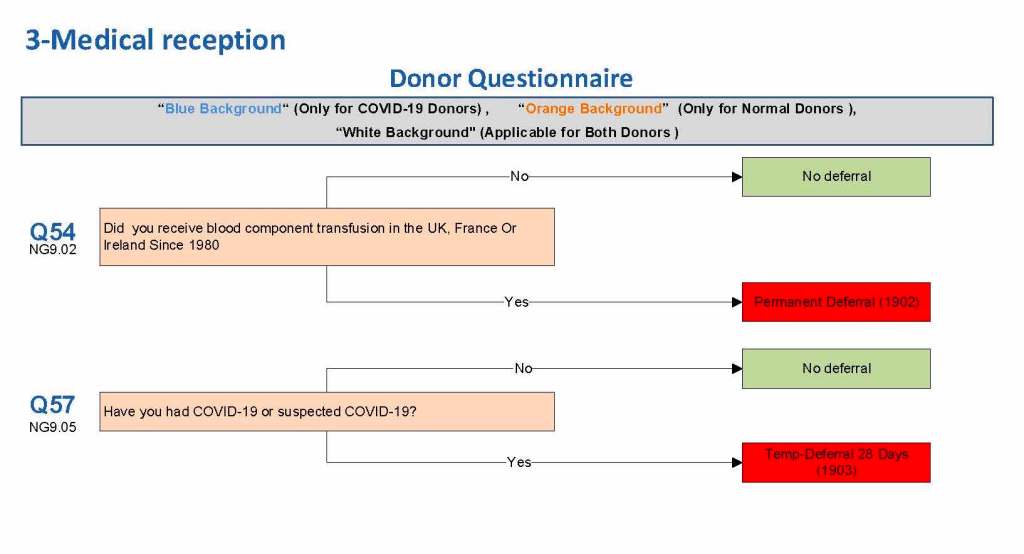
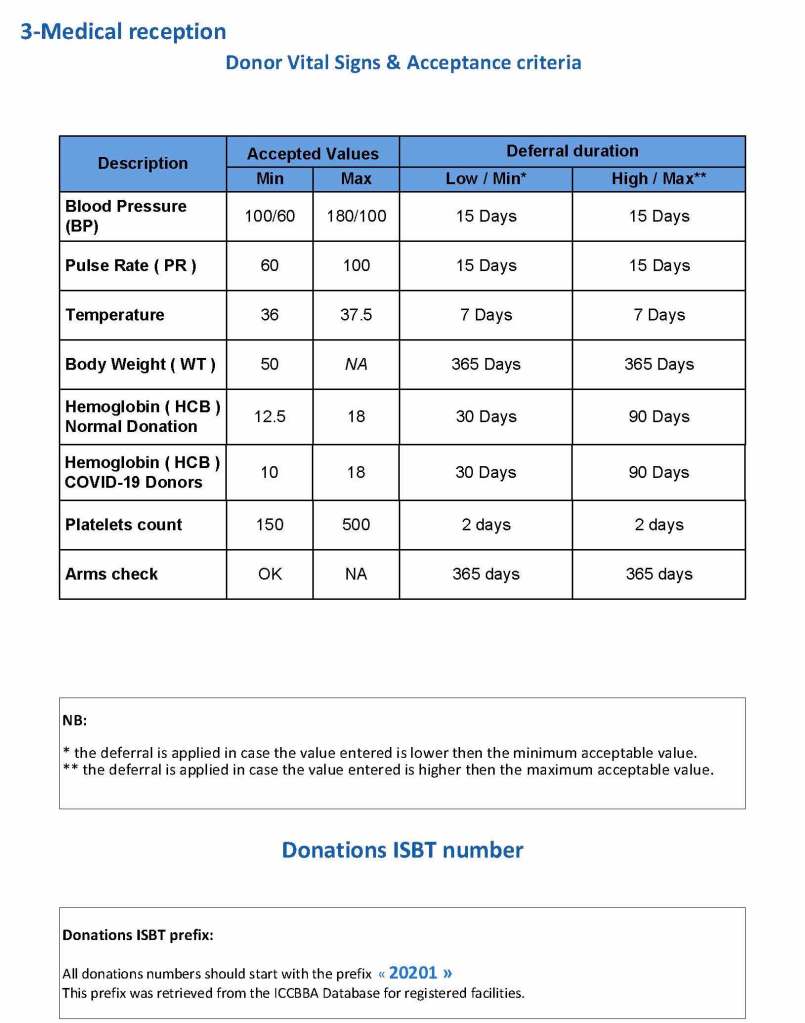
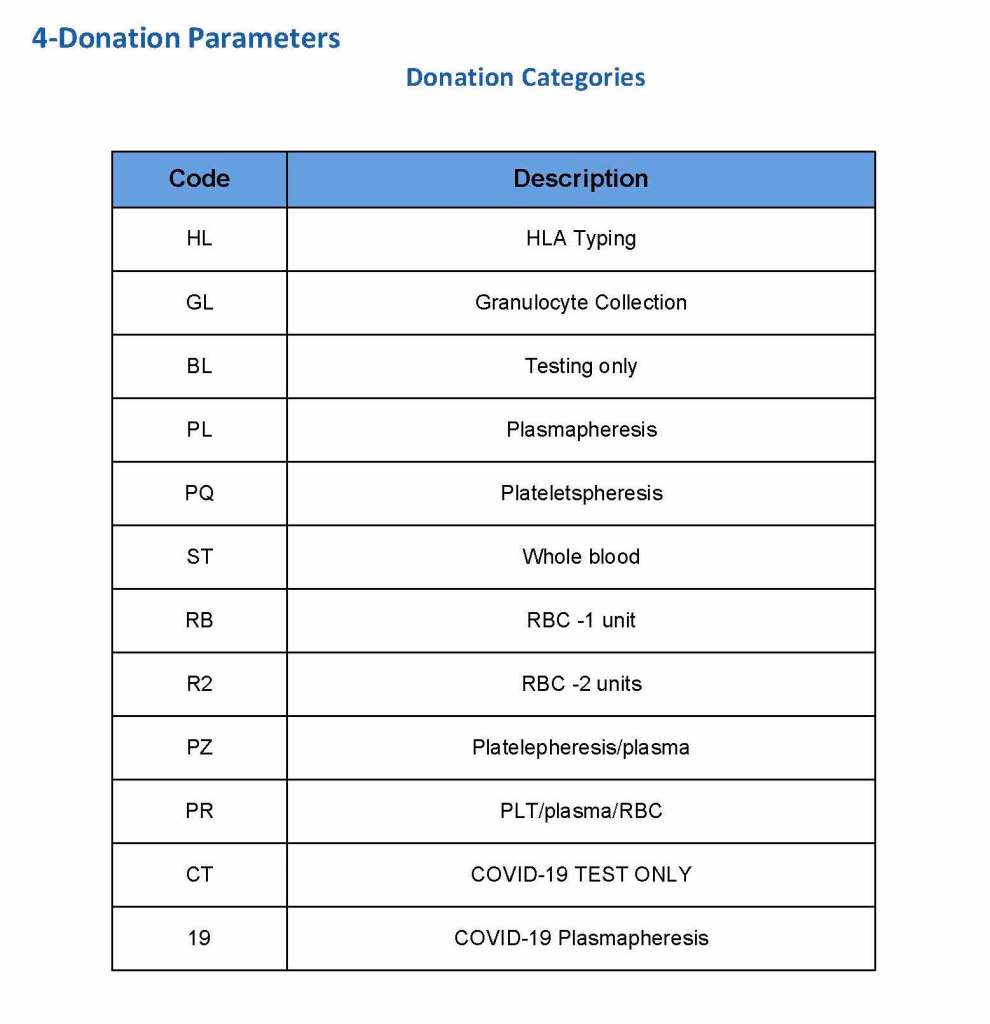
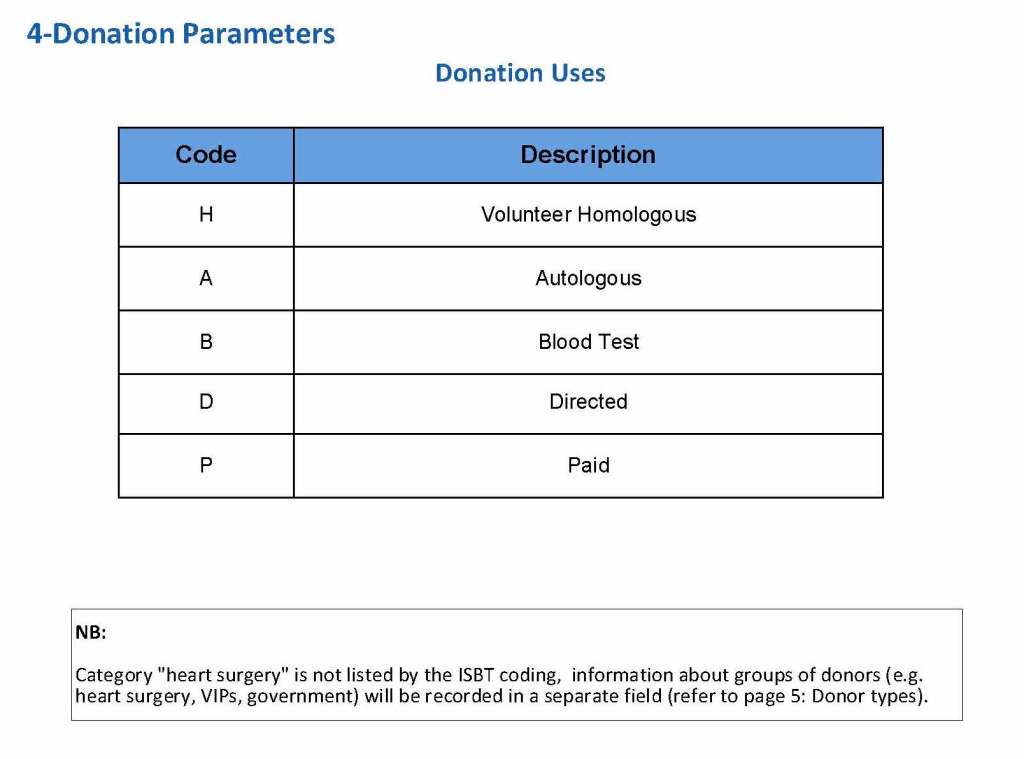
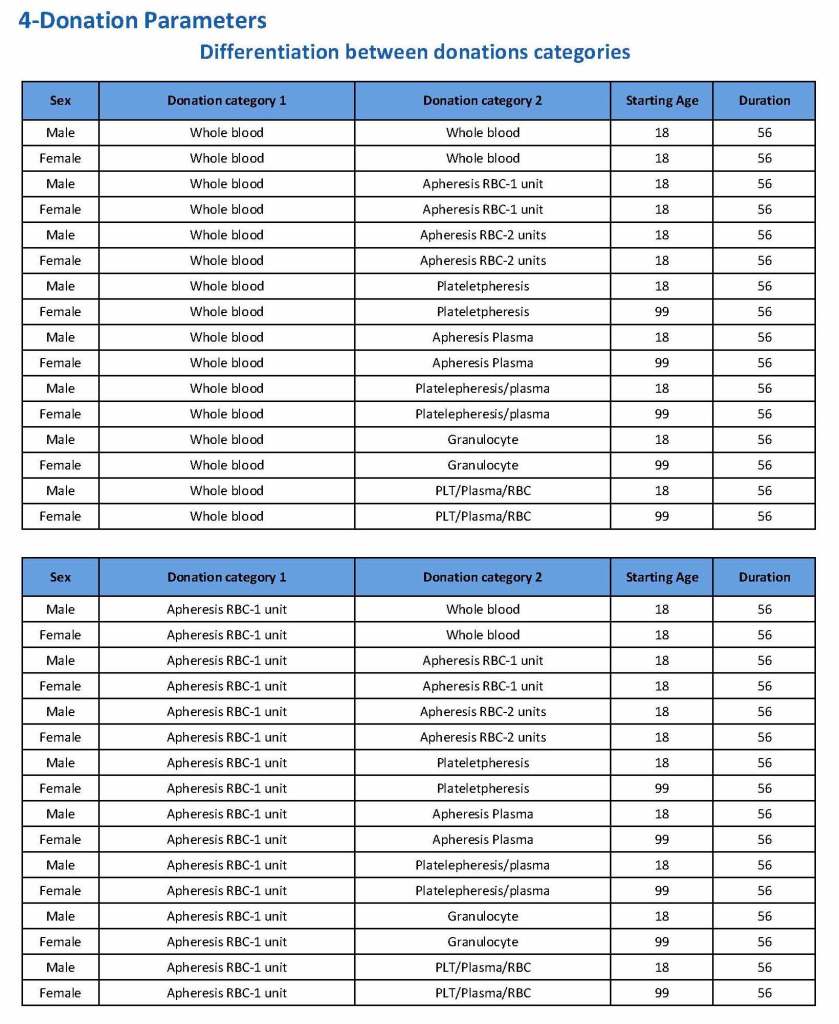
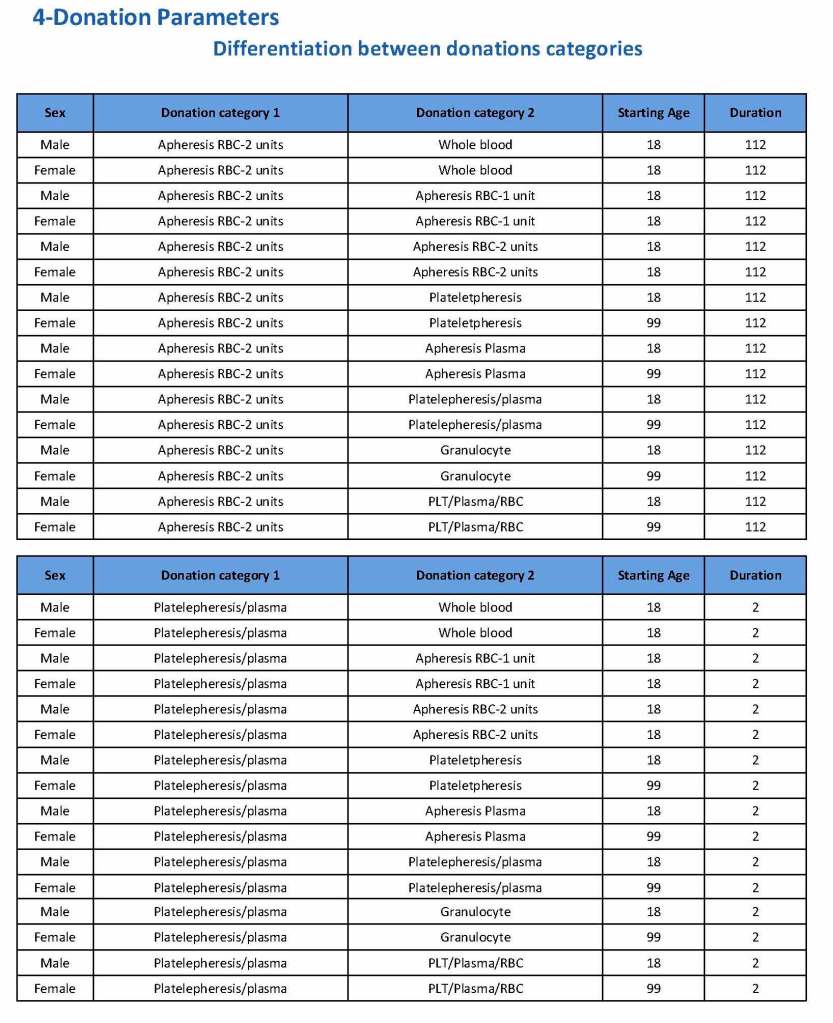
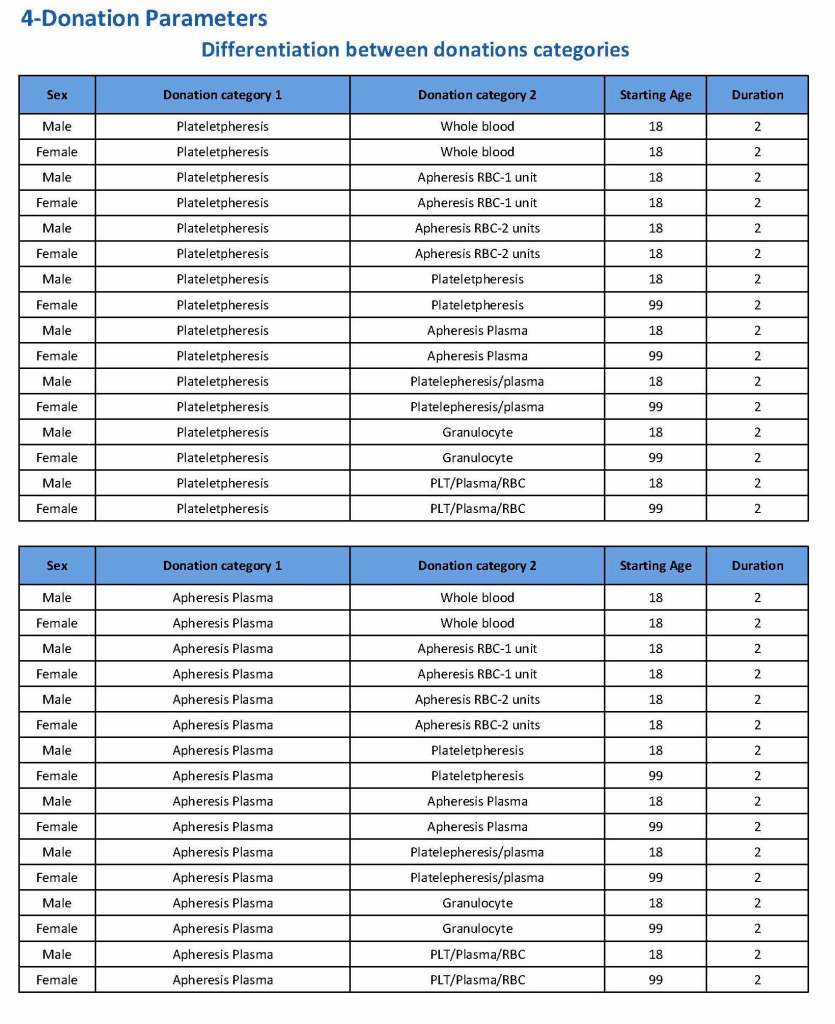
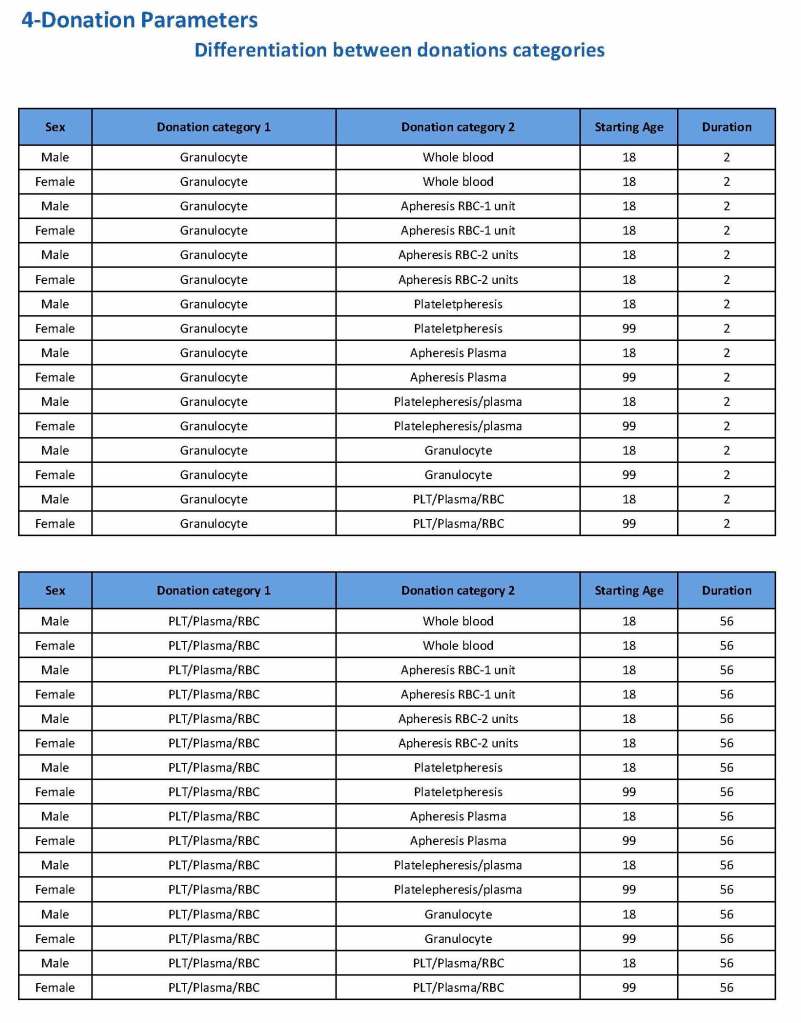
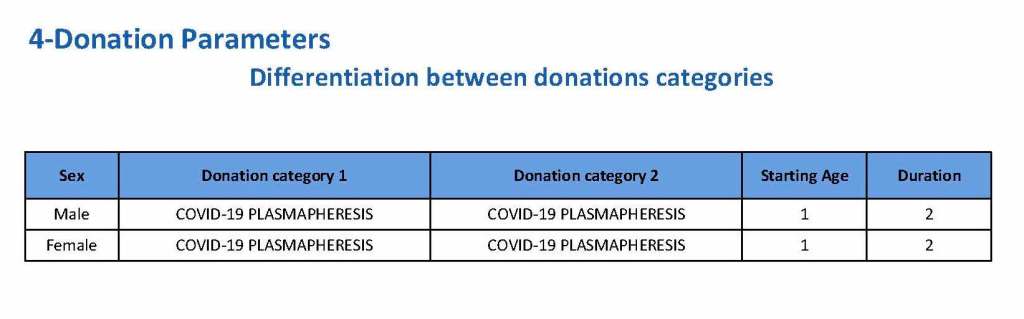
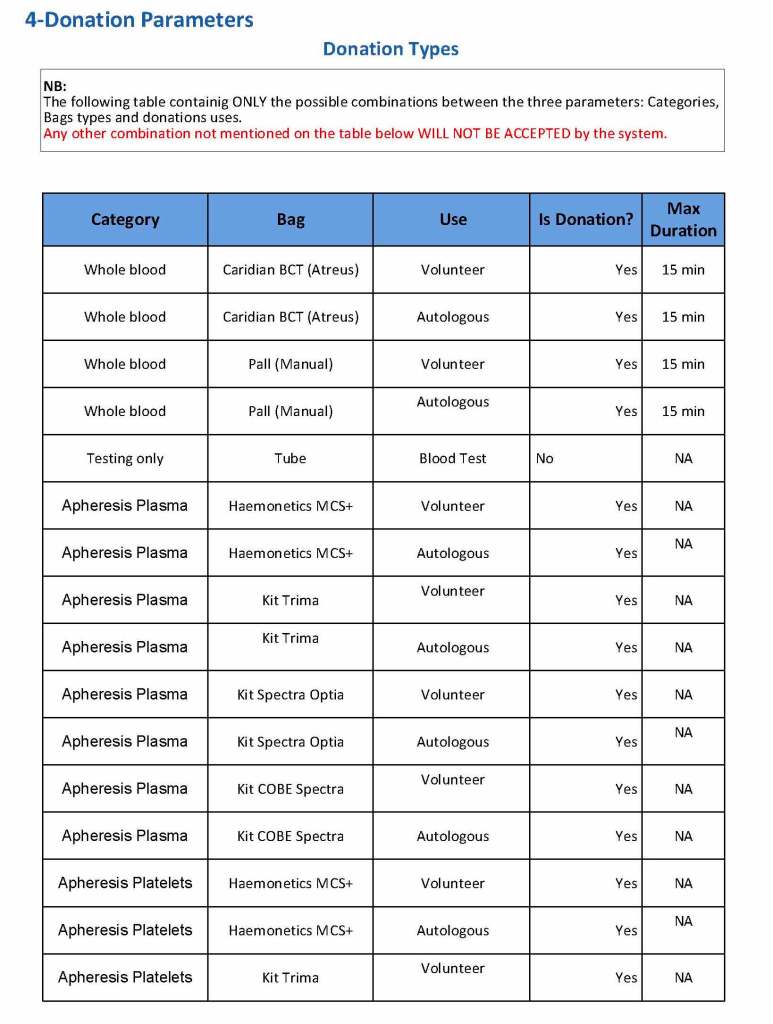
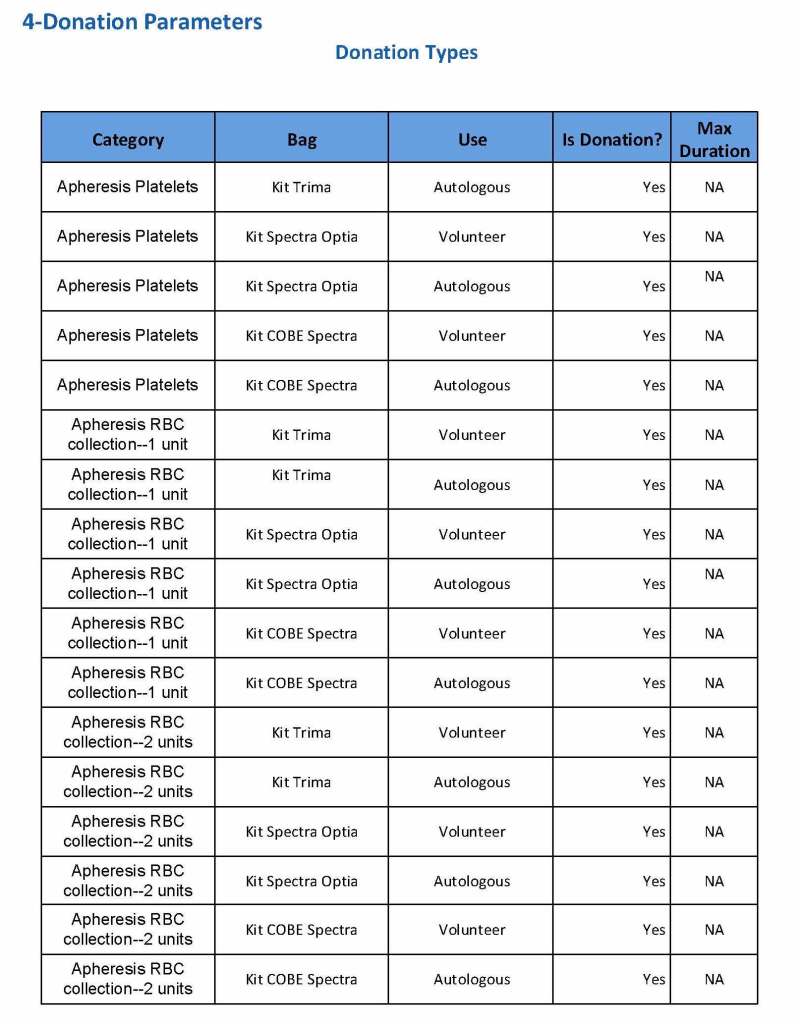
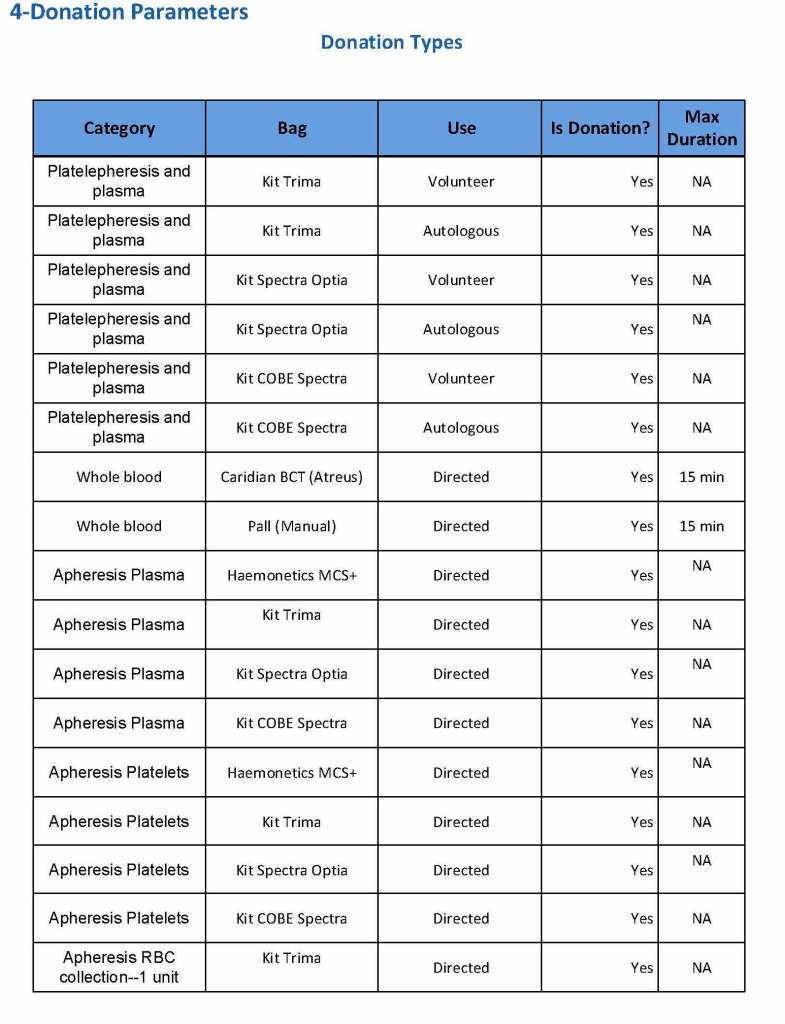
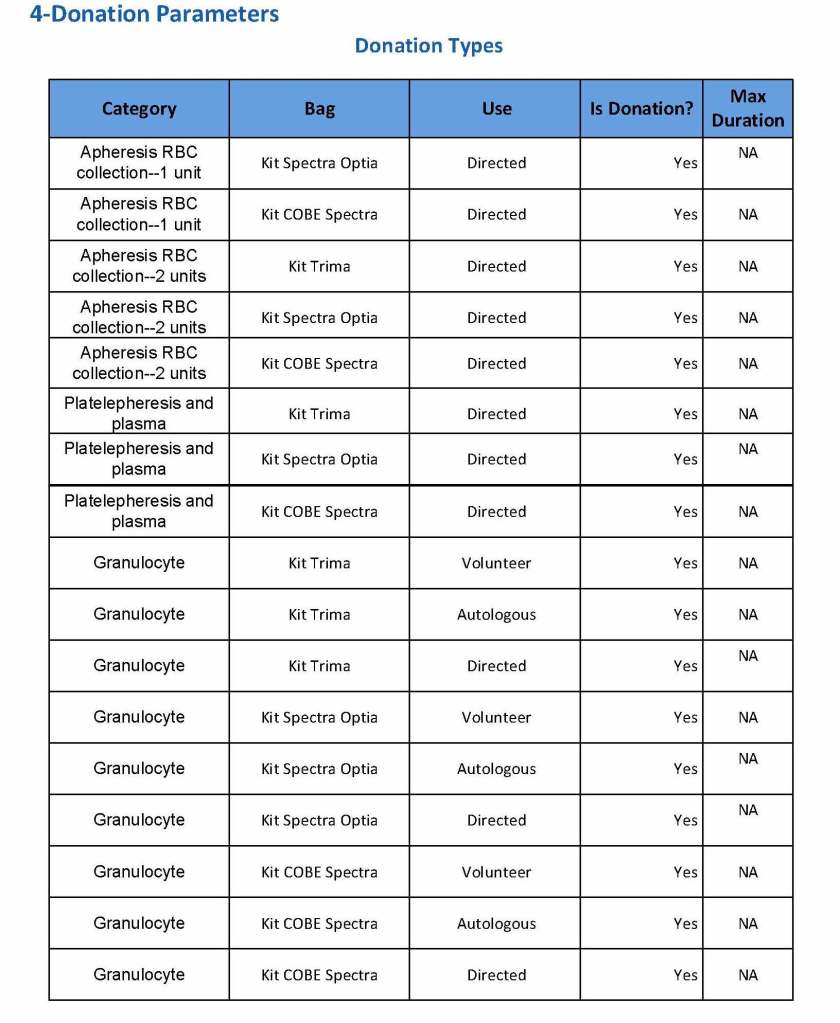
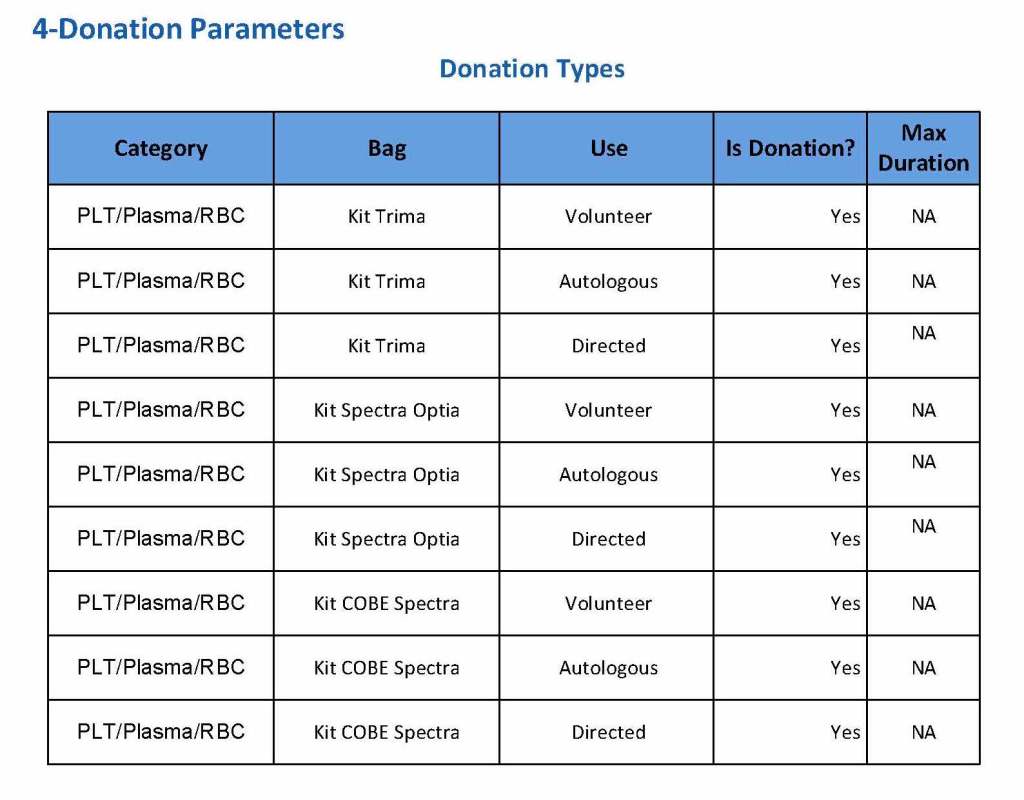
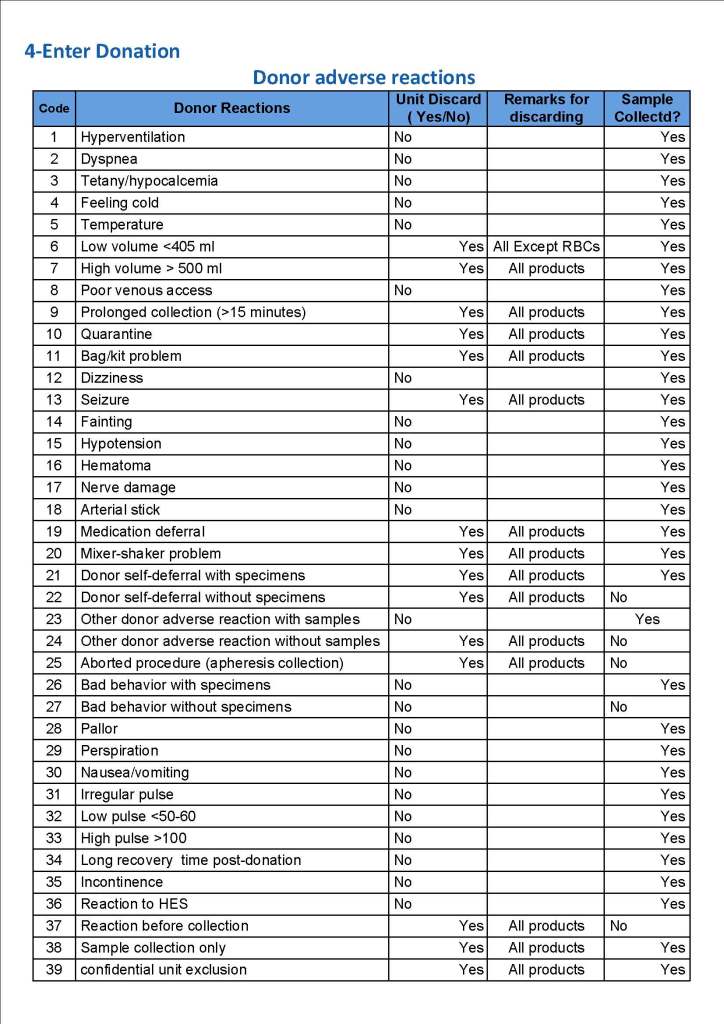
This is the basic COVID-19 convalescent plasma processing workflow specified by me and designed in Medinfo. I wish to thank Medinfo France and Medinfo Qatar for their fantastic support in completing the entire parallel but quarantined workflow in less than two weeks!! This was an extraordinary collaboration.
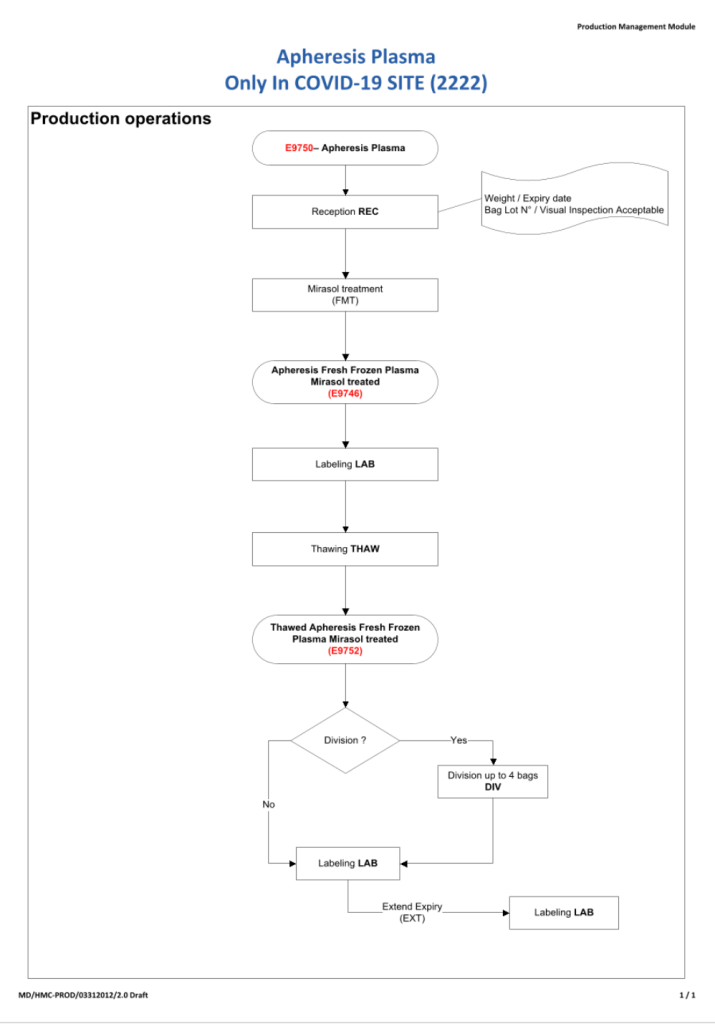
While I was still associated with HMC Doha, I developed and set up an expedited setup for COVID-19 convalescent plasma production, initially manual and then fully integrated into the Medinfo blood bank computer system.
Specifically, I built a customized version of our Medinfo blood bank system to replace the manual system and increase safety the safety and production throughput while maintaining good manufacturing practices GMP. The full system (manual first, then computerized) was implemented within two weeks including a completely separate quarantine convalescent COVID donor screening, collections, processing, and release.
Subsequent posts will detail my processes.