This is an update from the previous version posted to include low-titer group A FFP/FP24 and low ABO-titer group O whole blood.
Principle:
This policy outlines steps taken to minimize the risk of data entry errors and is based on a dualistic approach: review of results by a senior technologist and/or supervisor and various computer safeguards built into the Medinfo Hematos IIG blood bank computer HIIG system. This policy also discusses the verification (here called authorization) and purge processes of HIIG.
Policy:
- Review by senior technical, supervisory, or transfusion medical staff:
- Designated test procedures require review by a second technologist before authorization.
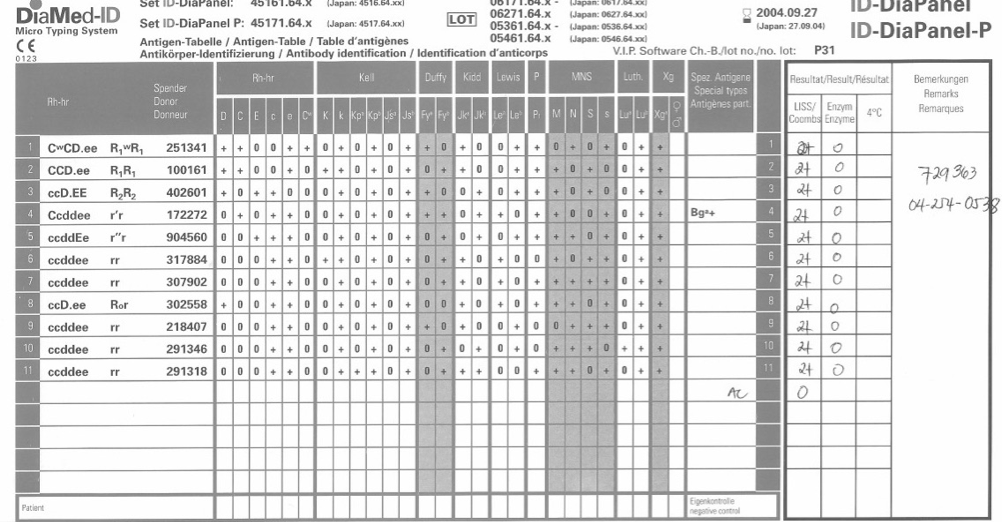
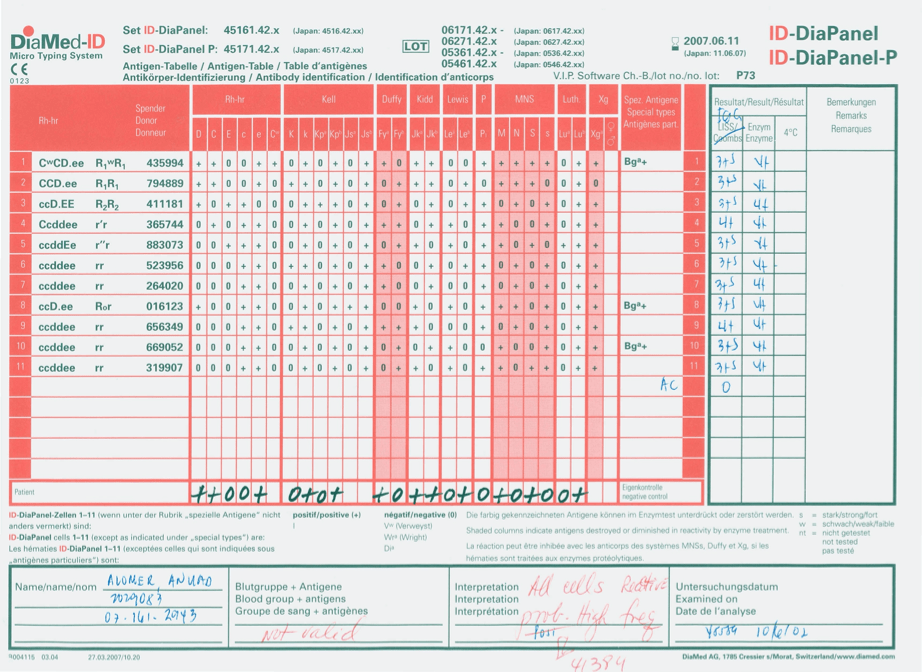
- Complex immunohematology testing and specimens showing aberrant results (e.g. ABO/D discrepancies) are reviewed by the supervisors or designates and ultimately a transfusion medicine physician before authorization.
- Computer system HIIG rules:
- Privileges:
- System restricts which staff can perform specific tests
- Patient/donor identity:
- System asks end-users to verify patient/donor identity before starting any access to the patient/donor record.
- System performs historical database checking and flags any inconsistencies (e.g. historical ABO/D typing differences, etc.)
- Testing:
- Only selected staff have privileges to authorize or purge.
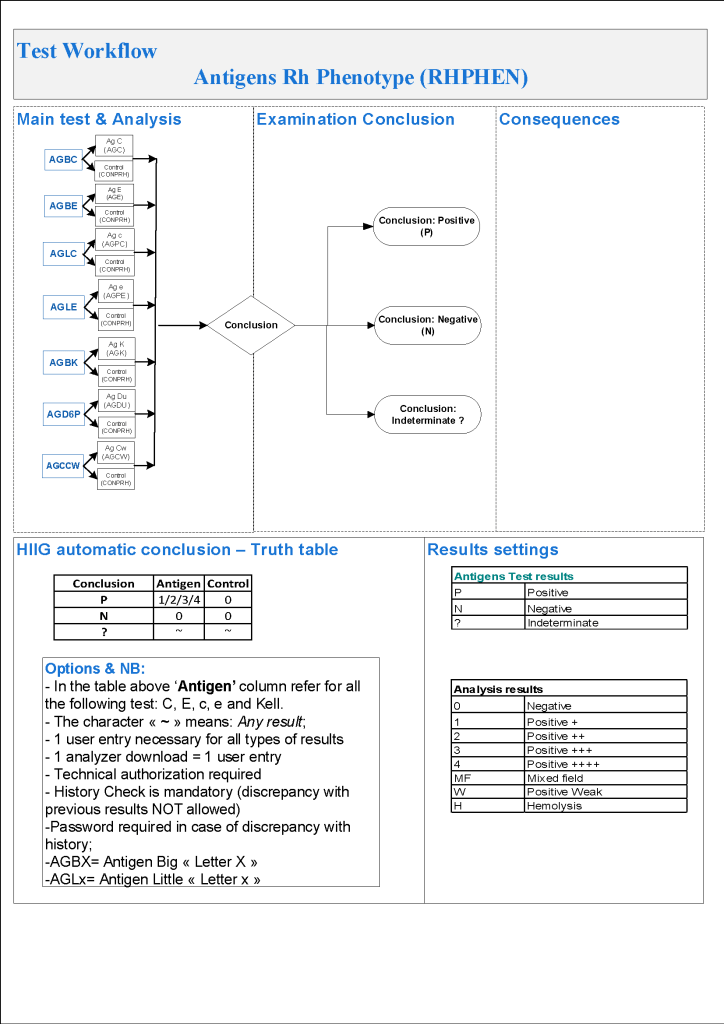
- ABO/D testing algorithms require entry of reactions, not interpretation of results and are compared to a truth table.
- Aberrant results require special review before ABO/D typing results can be authorized/purged.
- D-controls must be negative to allow D typing results to be authorized for liquid D-typing reagents.
- DAT results require appropriate controls to meet truth-table criteria.
- Eluates require last wash to be negative before authorization
- Blood components:
- Selection of RBC or plasma units requires two independent sample determinations within 72 hours of each other.
- ABO-incompatible RBC or FFP/FP24 transfusions are not allowed.
- Titer-based ABO blood group selection:
- Low titer group A FFP may be used as universal plasma like group AB.
- Group O whole blood with low anti-A and anti-B titers may be used for all ABO types.
- Acceptable titer threshold is specifically defined as parameters in Medinfo.
- Donors with any detectable clinically significant antibodies are permanently deferred.
- Depending on the patient’s antibody history, release of RBC units may require antigen-matched units. Examples:
- Mandatory matching (only antigen negative matched units allowed—no antigen positive or antigen-untyped units): Antibodies against H, D, c, K, k, Kpa, Kpb, Jsa, Jsb, Jka, Jkb antigens, anti-PP1Pk
- Priority matching (incompatible or untested can be approved by a transfusion medicine physician): C,E, e, Fya, Fyb, M, S, s
- Antigen matching not required: Lea, Leb, N
- Least-incompatible crossmatch require special authorization to release
- Protocols to force irradiation or other modified components can be setup in HIIG.
- Donors:
- Donor tests have same criteria as the same test used in patient testing for controls, etc.
- Donor demographics are read directly from the Ministry of Interior database—no manual entry (bar code only used).
- Privileges:
References:
- Workflows for Hematos IIG (1001 through 1005), 2013-2020
- Standards for Blood Banks and Transfusion Services, Current Edition, AABB, Bethesda, MD, USA
- Guidelines to the Preparation, Use, and Quality Assurance of Blood Components, European Committee (Partial Agreement) on Blood Transfusion (CD-P-TS), Current Edition