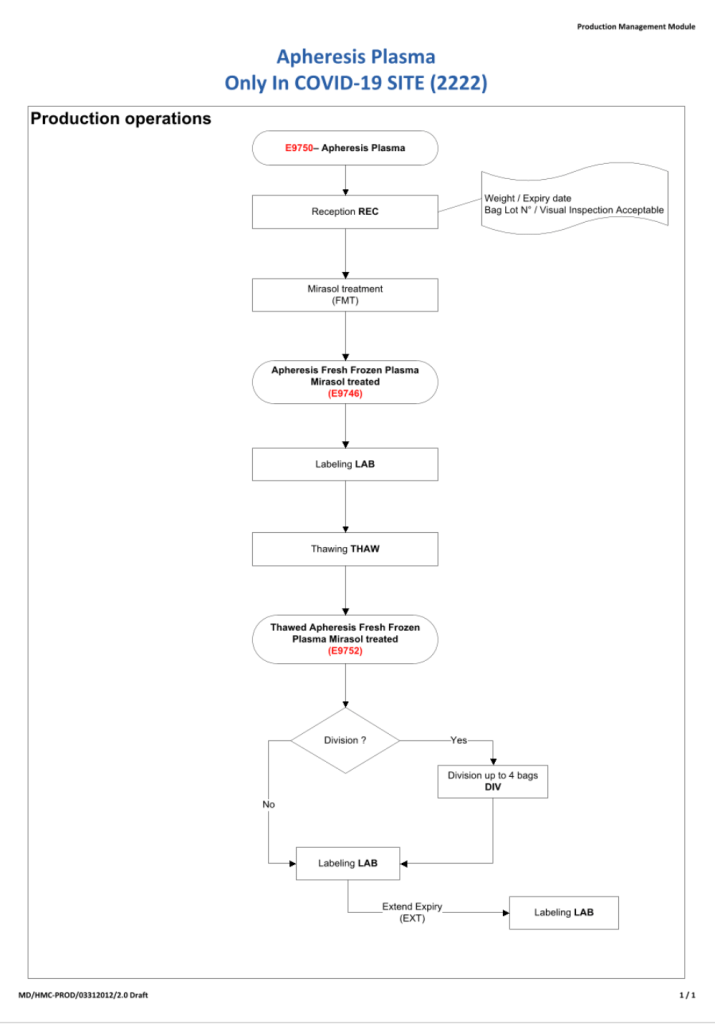
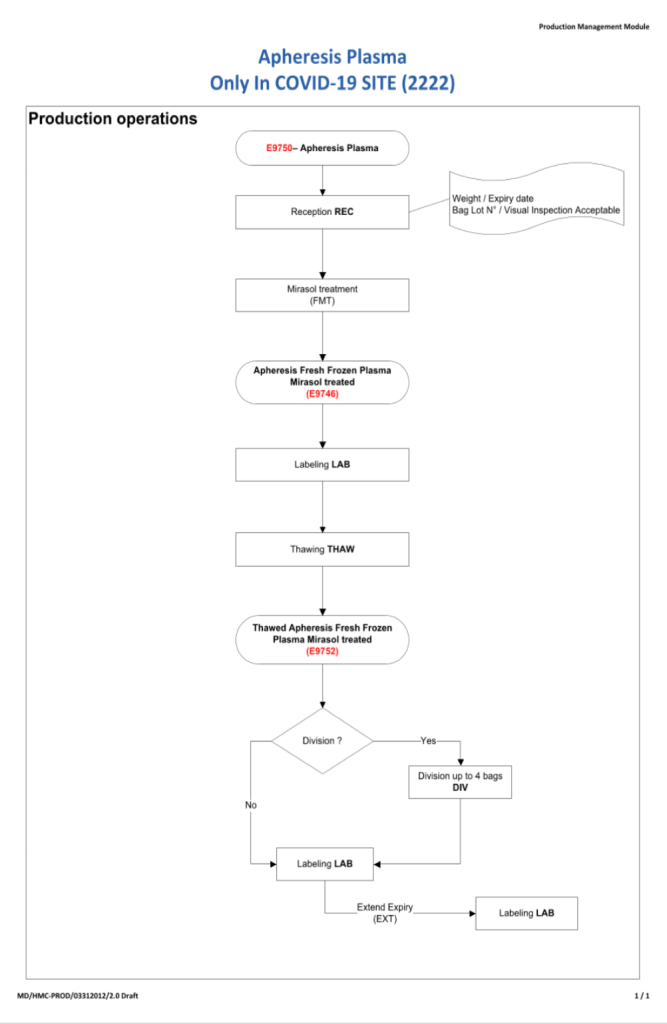
Principle:
For the purpose of typing blood donors, we want to detect weak and partial D types and consider them as D-positive since even a portion of the D molecule is immunogenic and sensitization to it may cause anti-D hemolytic disease of the newborn.
Background:
Ortho Diagnostics Reagents use three different monoclonal antibody cocktails that react variably with the antigen D (Rh1)—these are found on TWO (2) cards: Anti-A/B/A,B/D/CDE and Anti-DVI:
Anti-D/Anti-RH1—IgM monoclonal antibody clone D7B8 can detect most examples of weak and partial D including weak D types 1, 2, 3, 4.0, and D categories II, III, IV, V, VII, DBT, and R0Har. It does NOT detect category VI. Retest positive reactions of 2+ or less by an alternate method. It may show different serologic activity compared to other D typing reagents.
Anti-CDE/Anti-RH1,2,3—IgM monoclonal blend of clone MS24 (anti-C), clone MAD2 (Anti-D), and clone C2 (Anti-E) can detect most cells expressing C, D, or E antigens. Most examples of partial D including DVI and weak D express C or E antigens and will be detected directly by the included anti-D or indirectly by the anti-C or anti-E in the cocktail. It does NOT detect Rh:33 (R0Har).
Anti-DVI/Anti-RH1 will agglutinate cells with a DVI phenotype, analogous to our previous DVI+ reagents.
Policy:
- Follow the manufacturer’s instruction for storage, handling, and usage of all reagents.
- If the D-control is positive, the reactions are indeterminate, repeat by another method.
- For donors, run all three anti-D reagents listed—do not use the patient typing algorithm or reagents.
- Use the following table for interpretation and further actions if needed:
| Pattern # | Anti-D/D7B8 | Anti-CDE | Anti-DVI | D-Interpretation |
| 1 | Positive | Positive | Positive | D-positive |
| 2 | Positive | Positive | Negative | Do additional testing |
| 3 | Positive | Negative | Negative | Do additional testing |
| 4 | Negative | Negative | Positive | Do additional testing |
| 5 | Positive | Negative | Positive | Do additional testing |
| 6 | Negative | Positive | Positive | D-positive, probable DVI variant |
| 7 | Negative | Positive | Negative | D-negative, probable rare genotypes r’ and/or r’’ |
| 8 | Negative | Negative | Negative | D-Negative |
If the reaction is 2+ or less with the Ortho anti-D/D7B8 reagent or 1+ with either the CDE or DVI reagents is patterns 2, 3, 4, or 5 above, repeat by another manufacturer’s reagents, including DVI+ and DVI- sensitivities.
Medinfo-Ortho interface settings for Blood Donor Center:
| Anti-D/D7B8 | Anti-CDE | D-Interpretation |
| 3,4 | 1,2,3,4 | D-positive |
| 3,4 | 0 | Indeterminate |
| 0 | 0 | Indeterminate |
| 0 | 1,2,3,4 | Do DVI REFLEX |
| REFLEX DVI | If DVI-pos, then D-positive | |
| REFLEX DVI | If DVI-neg, then D-negative | |
| 0 | 0 | D-negative |
| ~ | ~ | Indeterminate |
~ means any other result
Note all of the following:
- No reagents may be able to detect all D variants.
- ~ means any other value for that reagent (e.g. anti-D/D7B8 reactions 1, 2, mf, hemolyzed)
- Note that this new algorithm makes a 2+ reactivity with Anti-D/D7B8 as indeterminate.
References:
- Product Insert, Anti-A/B/A,B/D/CDE/Control Card, Revised January, 2015, Ortho Clinical Diagnostics, High Wycombe, Buckinghamshire/UK
- Publication J55650_EN, Instructions for Use, Blood Grouping Reagent Ortho Sera Anti-D(DVI) (Anti-RH1), Version 2.0, 2015-07-30, Alba Bioscience, Edinburgh, UK
- Standards for Blood Banks and Transfusion Services, 29th Edition, AABB, Bethesda, MD, USA