This is part 2 of a lecture I gave for medical technology students while I was at National Guard Health Affairs in Riyadh.












Transfusion- and donor-service-related processes for all types of transplantation
This is part 2 of a lecture I gave for medical technology students while I was at National Guard Health Affairs in Riyadh.











2011
Established automated component production using Atreus technology, plasma and platelet pathogen inactivation (Mirasol)—made HMC component production Good Manufacturing System GMP compliant
2011
Qatar is the first to adopt non-PCR-based NAT technology (Grifols/Novartis Tigress) and becomes world reference site for this
2011
Based on the above, Qatar can now completely process all whole blood into blood components (red cells, platelets, and plasma) in as little as 5 hours from collection!
2011-2020:
I established policies and procedures for the hospital blood banks/transfusion services, blood donor center, therapeutic apheresis, and laboratory information systems to bring HMC in compliance with the Council of Europe, international AABB, and other standards. I customized our own standards for our local needs based on them.
2012-2013
Implemented custom build of the multilingual blood bank computer system (Medinfo) for both patient and donor services, including development of interfaces to all production equipment including Atreus and Mirasol (world’s first) and a direct link to Ministry of the Interior to obtain patient demographics in English and Arabic—Qatar became the world’s first site to combine fully-interfaced, automated component production with pathogen inactivation: Qatar becomes world reference site for this.
2013-2014
Built, validated, and implemented laboratory build of hospital information system, Cerner Millennium
2015
Replaced and updated Atreus with Reveos automated component production to allow faster throughput and capacity with a full bidirectional interface (world’s first), introduced platelet
additive solution PAS with pathogen inactivation (Mirasol)—Medinfo interfaces updated to Reveos for all equipment: this doubles the capacity to process whole blood into components using the same physical space
2015-2019
Updated dedicated blood bank software Medinfo Hematos IIG by several versions using Division Head, LIS, and internally trained Super Users—at great cost savings to HMC by not using outside consultants (e.g. Dell Consulting)
2019
Established column absorption technology using Terumo Optia therapeutic apheresis machine for treatment of ABO-incompatible renal transplants: I validated using the Ortho Vision MAX to perform ABO antibody titers for this system and correlated it with the reference method at Karolinska Institutet in Stockholm (manual gel) to bring rapid throughput and labor savings—Qatar being the first-site in the world to do this. We saved money by using the same apheresis machine to use this column absorption technology (no need for second machine to use the columns)
2020
Expedited setup (two weeks total) of COVID-19 convalescent plasma production, initially manual and then fully integrated into the Medinfo computer system as a customized module with separate quarantine collection, production, and transfusion service functions
Other:
I was awarded two HMC Star of Excellence Awards:
2013—Liver Transplantation Transfusion Support
2019—ABO-Incompatible Renal Transplantation Support














Everyone is excited at the potential of using stem cells for research and therapy. Below is my presentation of the logistics necessary to get those stem collected in an orderly manner, especially in this time of the COVID-19 pandemic. It will also consider blood bank software logistics.




















Principle:
Kidneys have strong expression of ABO type and must be matched the same way as RBC components. In the case of ABO-incompatible renal transplants, we must not give significant amounts of plasma incompatible to the ABO type of the donor kidney. Plasma must also be compatible with the patient’s ABO type for RBC transfusions. The amount of residual plasma in PRBCs is limited since we use an additive solution SAGM. Likewise, platelet components are suspended in platelet additive solution with only minimal residual plasma. Cryoprecipitate has only minimal plasma and is given without regard to the patient’s ABO type.
All of the following rules can be built into the blood bank computer system Medinfo without hard coding.
Policy:
References:
Setting up ABO-incompatible renal transplants is a major undertaking and requires close coordination between Transfusion Medicine and the clinical team. This post addresses my experience in setting up this program in 2019 at HMC in Qatar.
Like any process involving titration, it is best to automate it to minimize inter-technologist variability. Unfortunately, doing both IgG and IgM titers takes up to 1 hour per machine and totally monopolizes the machine during that interval. I did not have sufficient staff to even consider doing the titrations manually. Performing automated titers disrupted my workflow so I encouraged the clinicians to send the specimens for off-peak processing.
Titration:
Columns:
Apheresis Equipment:
Staffing:
Specimen Collection:
Table of Permissible ABO Types:
Software:
Selection of blood component for ABO-incompatible renal transplantation is discussed in a separate post that will follow.
25/12/20
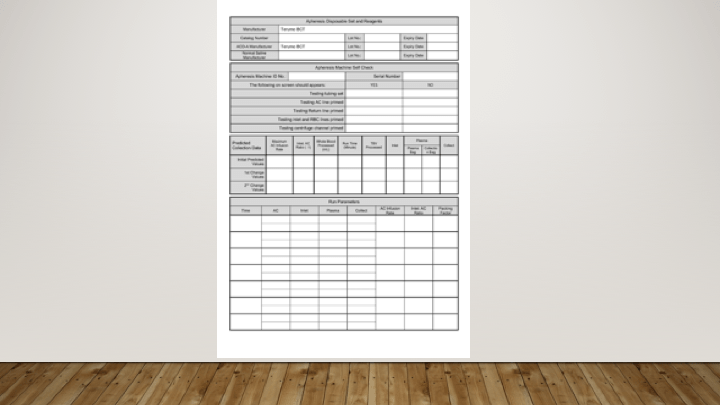
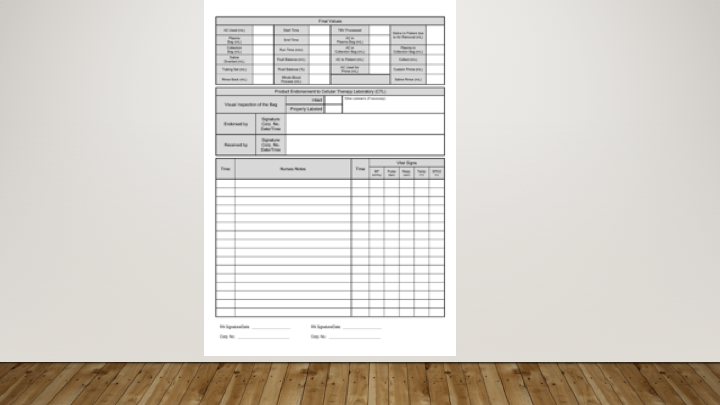
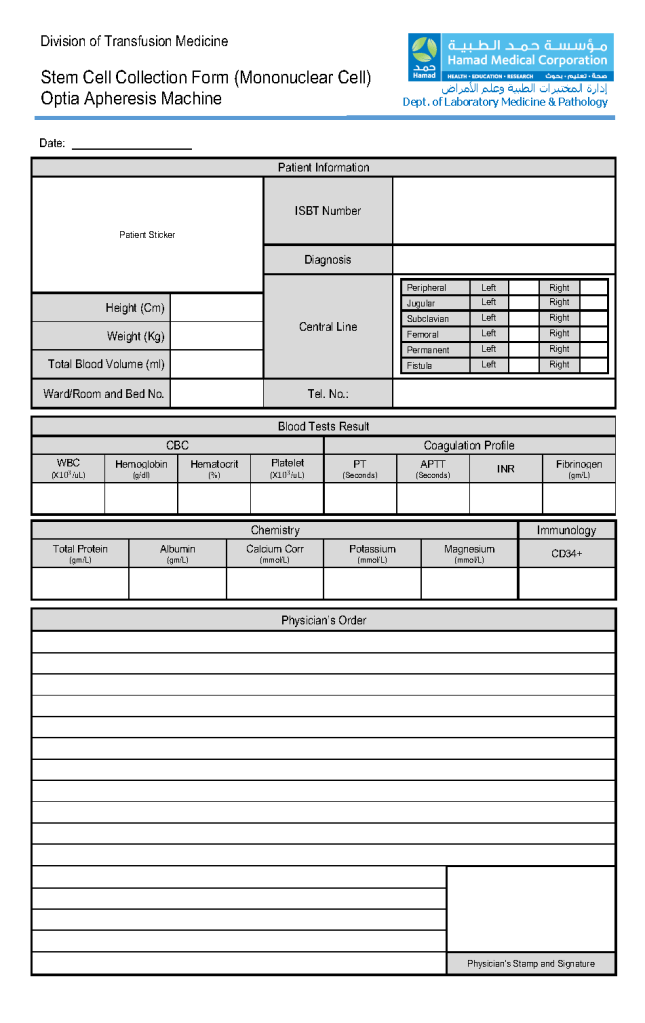
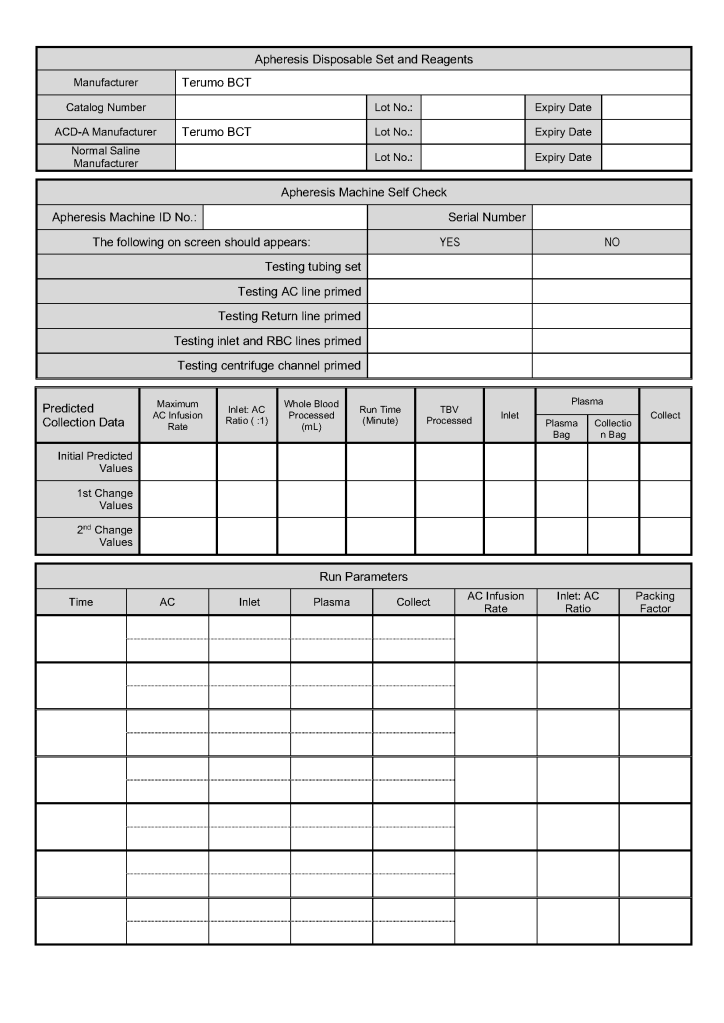
This is a sample of the stem cell therapeutic apheresis form that my apheresis team and I developed. It can be readily made into an electronic form. I want to thank Dr. Saloua Al Hmissi, Apheresis Consultant, and Ms. Mini Paul, Head Apheresis Nurse, for their efforts in making this form a success.
Principle:
Donor lymphocytes in an organ transplant may make antibodies and cause a clinically significant hemolytic anemia, i.e. a graft vs. host hemolytic anemia GVHHA. Optimal handling in these cases should include antibody screening/identification for all potential donors and recipients. The transfusion medicine physician should review the results for possible issues of antibody/antigen incompatibilities to proactively select matched blood components and avoid GVHHA.
In the Medinfo blood bank computer system, we can make custom rules to ensure release of only antigen-matched units as needed.
Policy:
Example: Donor has anti-Kell (K1) and patient is K1-positive. Use only K1-negative RBCs post-transplant.
References:
Principle:
Liver transplantation requires coordination of the Transfusion Service TS, Liver Transplant LTS , and the Blood Donor Center BDC to prepare blood components for this massive transfusion setting.
Policy:
Principle:
Organ donors with a history of RBC transfusion within the past three months must have ABO subgroup (weak A as detected by A2 cells and anti-A1 lectin) if the transfusion included group A RBCs.
Policy:
Reference:
Standards for Blood Banks and Transfusion Services, AABB, Current Edition, Bethesda, MD, USA