This process was originally done in the first phase of CCP collection. I have updated it to include SARS-CoV-2 antibody testing.
Principle:
Due to the pandemic, we will initially MANUALLY collect an experimental, investigational-use-only plasma product from apheresis donors and treat it with Mirasol. THIS IS A EMERGENCY INTERIM PROCESS UNTIL THE MEDINFO HEMATOS IIG PROCESSES ARE PREPARED AND VALIDATED.
Policy:
- Good Manufacturing Practice applies:
- Manufacturers’ recommended processes for equipment and materials usage applies.
- All staff engaged in these processes must be competency assessed successfully.
- Pre-Screening:
- Clinical staff will use the prescreening document to select donors for pre-donation screening.
- Quarantine:
- All processes (day 0, day 1, day 2, and product modification and release) will be done in quarantine areas SEPARATE and DISTINCT from regular Transfusion Medicine activities. This includes:
- Separate space and equipment must be provided.
- Equipment for this project may NOT be used for regular, non-quarantine processes
- Separate space and equipment must be provided.
- Non-Transfusion Medicine staff will not be permitted in operational areas.
- Prospective donors will not be permitted in the processing, testing, storage, or blood bank work areas.
- All processes (day 0, day 1, day 2, and product modification and release) will be done in quarantine areas SEPARATE and DISTINCT from regular Transfusion Medicine activities. This includes:
- Donation Process:
- Day 0: Registration, check donor deferral database, questionnaire, physical exam including arm check, and specimen collection using ISBT specimen labels
- Use latest manual donor questionnaire.
- Day 1: Donor marker and immunohematology testing, review of results, accept or reject donor for actual plasmapheresis
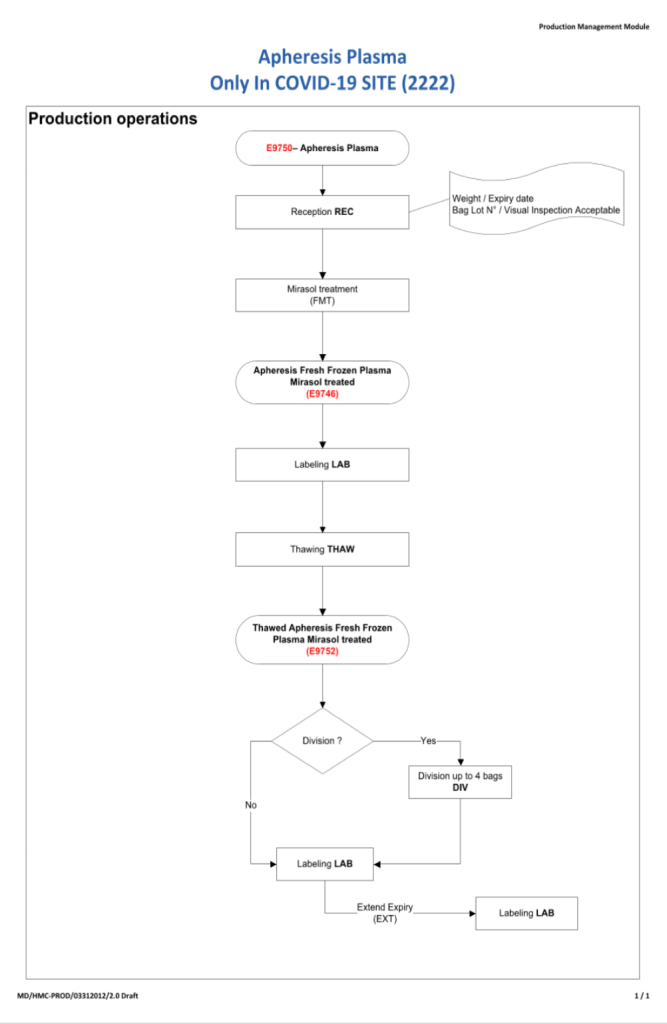
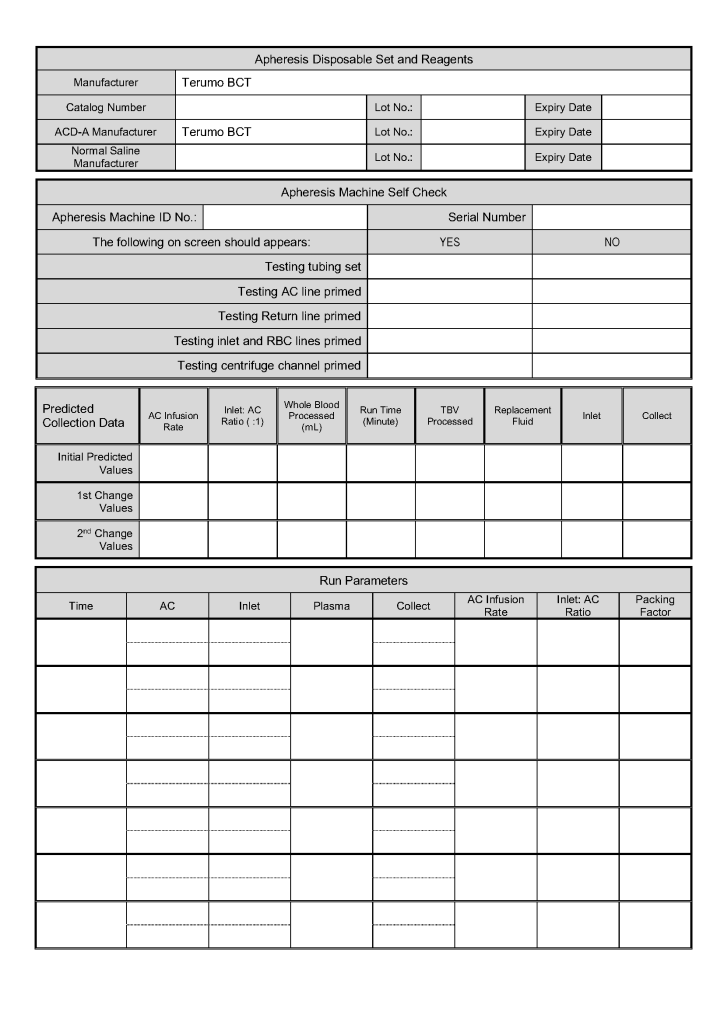
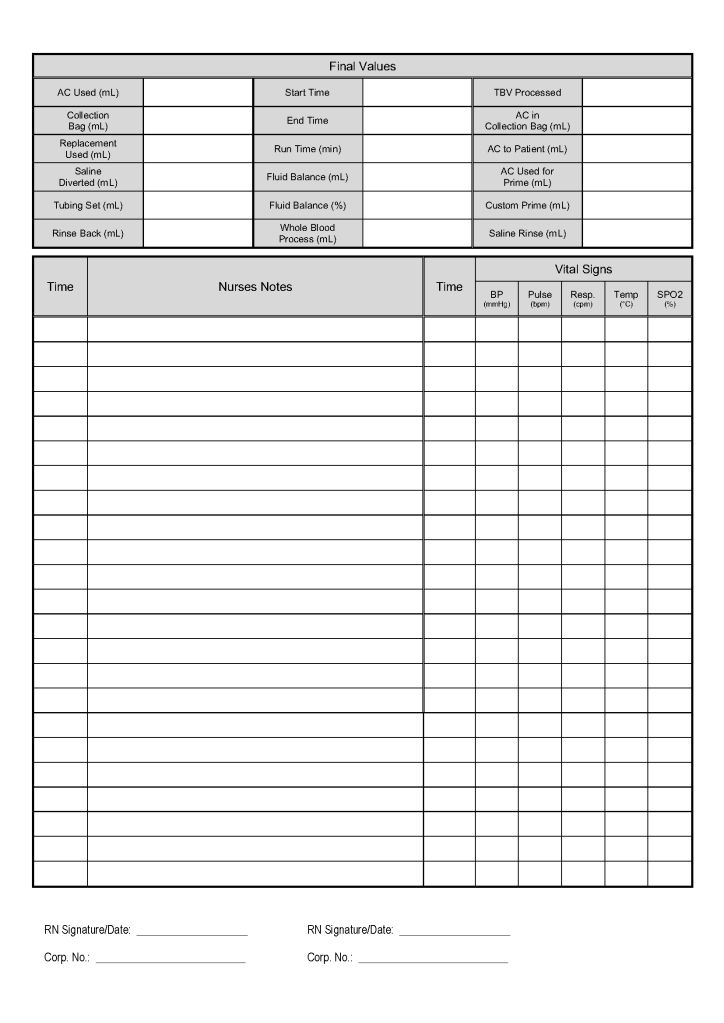
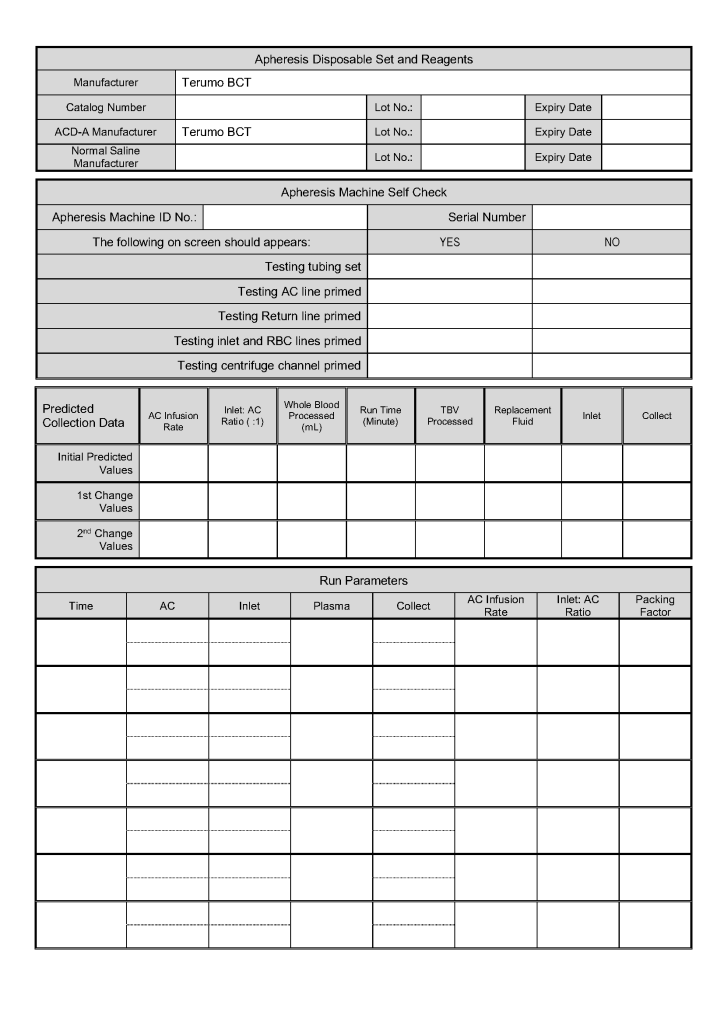
- Day 2: Collect manufacturer’s recommended volume of plasma (500 ml if < 80 kg, 600 ml if >= 80 kg), aliquot, pathogen-inactivate (Mirasol), freeze at minus 80C
- Testing:
- Testing will be performed with regular blood donor specimens using ISBT specimen labels
- Testing must be done by donor-specific processes (not those for clinical patients)
- Exclude malaria and HTLV testing.
- Testing must be directly interfaced to Medinfo Hematos IIG donor module
- CCP COVID antibody testing:
- SARS-CoV-2 antibody testing to be performed to determine cut-off for donor eligibility for CCP collection.
- Use of donors with antibody levels below threshold is at the discretion of the treating clinician.
- Processing:
- Aliquoting, pathogen-inactivation, and labelling may proceed if the pre-donation screening results are acceptable.
- Storage:
- Long-term in minus 80C quarantine freezer
- Short-term at 1-6 C just after thawing in quarantine refrigerator
- Standard temperature monitoring and alarms apply
- Labelling:
- The backup manual labelling process applies
- The ISBT specimen label will the donor unit number
- Outdate will be 6 years if the product is stored at -65C, 1 year if stored at -18C
- Product Release:
- Orders must be on the PAPER requisition (old Blood Bank Order Form) with a patient prescription and signed by a physician designated to treat COVID patients.
- No orders in Cerner
- Thawing plasma at 37C upon receipt of order by Transfusion Medicine staff
- Signing out component to clinical unit by Transfusion Medicine Staff to locations treating COVID-19 patients.
- Orders must be on the PAPER requisition (old Blood Bank Order Form) with a patient prescription and signed by a physician designated to treat COVID patients.
- Information Technology: Medinfo Hematos IIG customized software to be implemented as soon as possible for all processes
- Not covered: Transfusion Medicine is NOT responsible for:
- Triage of request for convalescent plasma
- Pickup and transport of components
References:
- Level 1-4 documents for donation, testing, processing, and release of blood components
- COVID-19 Plasma Donor Prescreening Document, 8/4/20